Abstract
Background
Osteomyelitis caused by Methicillin-resistant Staphylococcus aureus (MRSA) often requires surgery and prolonged systemic antibiotic treatment. Local antibiotic delivery systems of bioceramics or polymers have been developed to treat osteomyelitis. A disadvantage of biodegradable polymers is the initial burst of antibiotics into the environment; one advantage of bioceramics is its osteoconductivity. We therefore developed a vancomycin-containing poly-l-lactic acid/β-tricalcium phosphate (PLLA/β-TCP) composite to control antibiotic release and stimulate bone formation.
Questions/purposes
We (1) characterized these composites, (2) assessed vancomycin release in inhibitory doses, and (3) determined whether they would permit cell adhesion, proliferation, and mineralization in vitro.
Methods
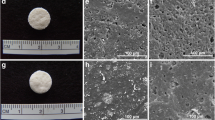
We molded 250 vancomycin-containing (VC) and 125 vancomycin-free (VUC) composites using PLLA, β-TCP, and chloroform. One hundred twenty-five VC composites were further dip-coated with PLLA (CVC) to delay antibiotic release. Composites were characterized according to their pore structure, size, volume, density, and surface area. Vancomycin release and bioactivity were determined. Adhesion, proliferation, and mineralization were assessed for two and three replicates on Days 3 and 7 with mesenchymal stem (MSC) and Saos type 2 cells.
Results
Pore size, volume, apparent density, and surface area of the CVC were 3.5 ± 1.9 μm, 0.005 ± 0.002 cm3/g, 1.18 g/cm3 and 3.68 m2/g, respectively. CVC released 1.71 ± 0.13 mg (63.1%) and 2.49 ± 0.64 mg (91.9%) of its vancomycin on Day 1 and Week 6, respectively. MSC and Saos type 2 cells attached and proliferated on composites on Days 3 and 7.
Conclusions
Vancomycin-containing PLLA/β-TCP composites release antibiotics in inhibitory doses after dip coating and appeared biocompatible based on adhesion, proliferation, and mineralization.
Clinical Relevance
Vancomycin-containing PLLA/β-TCP composites may be useful for controlling MRSA but will require in vivo confirmation.






Similar content being viewed by others
References
Adams K, Couch L, Cierny G, Calhoun J, Mader JT. In vitro an in vivo evaluation of antibiotic diffusion from antibiotic-impregnated polymethylmethacrylate beads. Clin Orthop Relat Res. 1992;278:244–252.
Antoci V Jr, Adams CS, Parvizi J, Davidson HM, Composto RJ, Freeman TA, Wickstrome E, Ducheyne P, Jungkind D, Shapiro IM, Hickok NJ. The inhibition of Staphylococcus epidermidis biofilm formation by vancomycin modified titanium alloy and implications for the treatment of periprosthetic infection. Biomaterials. 2008;29:4684–4690.
Aslam S, Darouiche RO. Antimicrobial therapy for bone and joint infections. Curr Infect Dis Rep. 2009;11:7–13.
Aunoble S, Clement D, Frayssinet P, Harmand MF, Le Huec JC. Biological performance of a new β-TCP/PLLA composite material for applications in spine surgery: in vitro and in vivo studies. J Biomed Mater Res A. 2006;78:416–422.
Broz A, Baresova V, Kromka A, Rezek B, Kalbacova M. Strong influence of hierarchically structured diamond nanotopography on adhesion of human osteoblasts and mesenchymal cells. Physica Status Solidi A. 2009;206:2038–2041.
Bryers JD. Medical biofilms. Biotechnol Bioeng. 2008;100:1–18.
Cabanas MV, Pena J, Roman J, Vallet-Regi M. Tailoring vancomycin release from β-TCP/agarose scaffolds. Eur J Pharm Sci. 2009;37:249–256.
Cevher E, Orhan Z, Mulazimoglu L, Sensoy D, Alper M, Yildiz A, Ozsoy Y. Characterization of biodegradable chitosan microspheres containing vancomycin and treatment of experimental osteomyelitis caused by methicillin-resistant Staphylococcus aureus with prepared microspheres. Int J Pharm. 2006;317: 127–135.
Chen Y, Cho MR, Mak AFT, Li JS, Wang M, Sun S. Morphology and adhesion of mesenchymal stem cells on PLLA, apatite and apatite/collagen surfaces. J Mater Sci Mater Med. 2008;19:2563–2567.
Cierny G 3rd, Mader JT, Penninck JJ. A clinical staging system for adult osteomyelitis. Clin Orthop Relat Res. 2003;414:7–24.
Dash AK, Cudworth GC. Therapeutic applications of implantable drug delivery systems. J Pharmacol Toxicol Methods. 1998;40:1–12.
Dion A, Langman M, Hall G, Filiaggi M. Vancomycin release behavior from amorphous calcium polyphosphate matrices intended for osteomyelitis treatment. Biomaterials. 2005;26:7276–7285.
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop DJ, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8:315–317.
Esposito S, Leone S. Prosthetic joint infections: microbiology, diagnosis, management and prevention. Int J Antimicrob Agents. 2008;32:287–293.
Garvin K, Feschuk C. Polylactide-polyglycolide antibiotic implants. Clin Orthop Relat Res. 2006;437:105–110.
Gbureck U, Vorndran E, Barralet JE. Modeling vancomycin release kinetics from microporous calcium phosphate ceramics comparing static and dynamic immersion conditions. Acta Biomater. 2008;4:1480–1486.
Gursel I, Yagmurlu F, Korkusuz F, Hasirci V. In vitro antibiotic release from poly(3-hydroxybutyrate-co-3-hydroxyvalerate) rods. J Microencapsul. 2002;19:153–164.
Hartemann-Heurtier A, Senneville E. Diabetic foot osteomyelitis. Diabetes Metab. 2008;34:87–95.
Hendriks JG, van Horn JR, van der Mei HC, Busscher HJ. Backgrounds of antibiotic-loaded bone cement and prosthesis-related infection. Biomaterials. 2004;25:545–556.
Jager M, Feser T, Denck H, Krauspe R. Proliferation and osteogenic differentiation of mesenchymal stem cells cultured onto three different polymers in vitro. Ann Biomed Eng. 2005;33:1319–1332.
Kanellakopoulou K, Giamerallos-Bourboulis EJ. Carrier systems for the local delivery of antibiotics in bone infections. Drugs. 2000;59:1223–1232.
Kluin OS, van der Mei HC, Busscher HJ, Neut D. A surface-eroding antibiotic delivery system based on poly-(trimethylene carbonate). Biomaterials. 2009;30:4738–4742.
Koç N, Timuçin M, Korkusuz F. Fabrication and characterization of porous tricalcium phosphate ceramics. Ceramics Int. 2004;30:205–211.
Korkusuz F, Uchida A, Shinto Y, Inoue K, Ono K. Biomaterial centered chronic osteomyelitis. Turk J Med Res. 1992;10:268–292.
Lamp KC, Friedrich LV, Mendez-Vigo L, Russo R. Clinical experience with daptomycin for the treatment of patients with osteomyelitis. Am J Med. 2007;120:13–20.
Lavery LA, Peters EJ, Armstrong DG, Wendel CS, Murdoch DP, Lipsky BA. Risk factors for developing osteomyelitis in patients with diabetic foot wounds. Diabetes Res Clin Pract. 2009;83:347–352.
Lew DP, Waldvogel FA. Osteomyelitis. Lancet. 2004;364: 369–379.
Li H, Ogle H, Jiang B, Hagar M, Li B. Cefazolin embedded biodegradable polypeptide nanofilms promising for infection prevention: a preliminary study on cell responses. J Orthop Res. 2010;28:992–999.
Liu SJ, Ueng SW, Lin S, Chan E. In vivo release of vancomycin from biodegradable beads. J Biomed Mater Res. 2002;63:807–813.
Lucke M, Schmidmaier G, Sadoni S, Wildemann B, Schiller R, Haas NP, Raschke M. 2003. Gentamicin coating of metallic implants reduces implant-related osteomyelitis in rats. Bone. 2003;32:521–531.
Mamidwar SS, Arena C, Kelly S, Alexander H, Ricci J. In vitro characterization of a calcium sulfate/PLLA composite for use as a bone graft material. J Biomed Mater Res B Appl Biomater. 2007;81:57–65.
Merten HA, Wiltfang J, Grohmann U, Hoenig JF. Intraindividual comparative animal study of α- and β-tricalcium phosphate degradation in conjunction with simultaneous insertion of dental implants. J Craniofac Surg. 2001;12:59–68.
Miyai T, Ito A, Tamazawa G, Matsuno T, Sogo Y, Nakamura C, Yamazaki A, Satoh T. Antibiotic-loaded poly-ε-caprolactone and porous β-tricalcium phosphate composite for treating osteomyelitis. Biomaterials. 2008;29:350–358.
Montjovent MO, Mark S, Mathieu L, Scaletta C, Scherberich A, Delabarde C, Zambelli PY, Bourban PE, Applegate LA, Pioletti DP. Human fetal bone cells associated with ceramic reinforced PLA scaffolds for tissue engineering. Bone. 2008;42:554–564.
Nandi KS, Kundu B, Ghosh SK, Mandal TK, Datta S, De DK, Basu D. Cefuroxime-impregnated calcium phosphates as an implantable delivery system in experimental osteomyelitis. Ceramics Int. 2009;35:1367–1376
Noel SP, Courtney H, Bumgardner JD, Haggard WO. Chitosan films: a potential local drug delivery system for antibiotics. Clin Orthop Relat Res. 2008;466:1377–1382.
Petrone C, Hall G, Langman M, Filiaggi MJ. Compaction strategies for modifying the drug delivery capabilities of gelled calcium polyphosphate matrices. Acta Biomater. 2008;4:403–413.
Rezwan K, Chen QZ, Blaker JJ, Boccaccini AR. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials. 2006;27:3413–3431.
Rybak M, Lomaestro B, Rotschafer JC, Moellering R Jr, Craig W, Billeter M, Dalovisio JR, Levine DP. Therapeutic monitoring of vancomycin in adult patients: a consensus review of the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, and the Society of Infectious Diseases Pharmacists. Am J Health Syst Pharm. 2009;66:82–98.
Schmidmaier G, Lucke M, Wildemann B, Haas NP, Raschke M. Prophylaxis and treatment of implant-related infections by antibiotic-coated implants: a review. Injury. 2006;37:105–112.
Schofield SC, Berno B, Langman M, Hall G, Filiaggi MJ. Gelled calcium polyphosphate matrices delay antibiotic release. J Dent Res. 2006;85:643–647.
Senthi S, Munro JT, Pitto RP. Infection in total hip replacement: meta-analysis. Int Orthop. 2011;35:253–260.
Shikhar Vohra S, Hennessy KM, Sawyer AA, Zhuo Y, Bellis SL. Comparison of mesenchymal stem cell and osteosarcoma cell adhesion to hydroxyapatite. J Mater Sci Mater Med. 2008;19:3567–3574.
Sia IG, Berbari EF. Osteomyelitis. Best Pract Res Clin Rheumatol. 2006;20:1065–1081.
Tanaka KS, Dietrich E, Ciblat S, Métayer C, Arhin FF, Sarmiento I, Moeck G, Parr TR Jr, Far AR. Synthesis and in vitro evaluation of bisphosphonated glycopeptide prodrugs for the treatment of osteomyelitis. Bioorg Med Chem Lett. 2010;20:1355–1359.
Tenover FC, Lancaster MV, Hill BC, Steward CD, Stocker SA, Hancock GA, O’Hara CM, McAllister SK, Clark NC, Hiramatsu K. Characterization of staphylococci with reduced susceptibilities to vancomycin and other glycopeptides. J Clin Microbiol. 1998;36:1020–1027.
Webb ND, McCanless JD, Courtney HS, Bumgardner JD, Haggard WO. Daptomycin eluted from calcium sulfate appears effective against Staphylococcus. Clin Orthop Relat Res. 2008;466:1383–1387.
Yagmurlu MF, Korkusuz F, Gursel I, Korkusuz P, Ors U, Hasirci V. Sulbactam-cefoperazone polyhydroxybutyrate-cohydroxyvalerate (PHBV) local antibiotic delivery system: in vivo effectiveness and biocompatibility in the treatment of implant-related experimental osteomyelitis. J Biomed Mater Res. 1999;46:494–503.
Zimmerli W. Infection and musculoskeletal conditions: prosthetic-joint-associated infections. Best Pract Res Clin Rheumatol. 2006;20:1045–1063.
Acknowledgments
We thank Muharrem Timuçin and Nursen Koç of Middle East Technical University, Department of Metallurgical and Material Engineering, Ankara, Turkey, who produced and provided the β-TCP. We also thank Duygu Uçkan, MD, PhD, of Hacettepe University, Faculty of Medicine, Department of Pediatrics Bone Marrow Transplantation Unit, Ankara, Turkey, for designing and interpreting the results of the in vitro study; Bülent Gümusel, PhD, of Hacettepe University, Faculty of Pharmacy, Department of Pharmacology, Ankara, Turkey, for directing the vancomycin release studies, and Kemal Behlulgil, PhD, of Middle East Technical University Central Laboratory for characterizing the composites.
Author information
Authors and Affiliations
Corresponding author
Additional information
Each author certifies that he or she has no commercial associations (eg, consultancies, stock ownership, equity interest, patent/licensing arrangements, etc) that might pose a conflict of interest in connection with the submitted article.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research editors and board members are on file with the publication and can be viewed on request.
Clinical Orthopaedics and Related Research neither advocates nor endorses the use of any treatment, drug or device. Readers are encouraged to always seek additional information, including FDA-approval status, of any drug or device prior to clinical use.
Each author certifies that his or her institution waived approval for the reporting of this investigation and that all investigations were conducted in conformity with ethical principles of research.
This study was performed at Middle East Technical and Hacettepe Universities of Ankara, Turkey.
About this article
Cite this article
Kankilic, B., Bayramli, E., Kilic, E. et al. Vancomycin Containing PLLA/β-TCP Controls MRSA In Vitro. Clin Orthop Relat Res 469, 3222–3228 (2011). https://doi.org/10.1007/s11999-011-2082-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-011-2082-9