Abstract
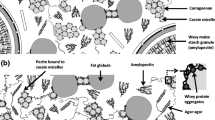
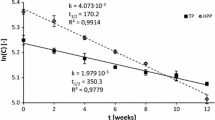
The objective of the present study was to reproduce in 4 months the quality changes that occur in shelf-stable strawberry pudding after 1 year of storage. A full factorial experiment 23 was used to investigate the effect of storage temperature (16 and 30 °C), headspace volume (9.5 and 18 cm3), and agitation (no/yes—pouches turned upside down twice a week) on aging. Sensory, rheological, pH, and fluorescence measurements were performed on samples subjected to accelerated aging tests (up to 4 months) or stored in standard conditions (up to 1 year). Although storage at 30 °C induced a slight decrease in apparent viscosity and thickness—in contrast with samples stored at 16 °C—it showed the best results for most of the studied parameters, regardless of the headspace volume and agitation. Hence, storage at 30 °C could be used for accelerated shelf-life or stability studies of sterilized pudding.






Similar content being viewed by others
References
Al-Attabi, Z., D’Arcy, B. R., & Deeth, H. C. (2009). Volatile sulphur compounds in UHT milk. Critical Reviews in Food Science and Nutrition, 49(1), 28–47. https://doi.org/10.1080/10408390701764187.
Birlouez-Aragon, I., Nicolas, M., Metais, A., Marchond, N., Grenier, J., & Calvo, D. (1998). A rapid fluorimetric method to estimate the heat treatment of liquid milk. International Dairy Journal, 8(9), 771–777. https://doi.org/10.1016/S0958-6946(98)00119-8.
Cardelli, C., & Labuza, T. P. (2001). Application of Weibull hazard analysis to the determination of the shelf life of roasted and ground coffee. LWT - Food Science and Technology, 34(5), 273–278. https://doi.org/10.1006/fstl.2000.0732.
Catauro, P. M., & Perchonok, M. H. (2012). Assessment of the long-term stability of retort pouch foods to support extended duration spaceflight. Journal of Food Science, 77(1), S29–S39. https://doi.org/10.1111/j.1750-3841.2011.02445.x.
Clare, D. A., Bang, W. S., Cartwright, G., Drake, M. A., Coronel, P., & Simunovic, J. (2005). Comparison of sensory, microbiological, and biochemical parameters of microwave versus indirect UHT fluid skim milk during storage. Journal of Dairy Science, 88(12), 4172–4182. https://doi.org/10.3168/jds.S0022-0302(05)73103-9.
Depypere, F., Verbeken, D., Torres, J. D., & Dewettinck, K. (2009). Rheological properties of dairy desserts prepared in an indirect UHT pilot plant. Journal of Food Engineering, 91(1), 140–145. https://doi.org/10.1016/j.jfoodeng.2008.08.017.
Diaz, J. V., Anthon, G. E., & Barrett, D. M. (2007). Nonenzymatic degradation of citrus pectin and pectate during prolonged heating: effects of pH, temperature, and degree of methyl esterification. Journal of Agricultural and Food Chemistry, 55(13), 5131–5136. https://doi.org/10.1021/jf0701483.
Dufour, E., Mazerolles, G., Devaux, M. F., Duboz, G., Duployer, M. H., & Mouhous Riou, N. (2000). Phase transition of triglycerides during semi-hard cheese ripening. International Dairy Journal, 10(1–2), 81–93. https://doi.org/10.1016/S0958-6946(00)00025-X.
Gaucher, I., Mollé, D., Gagnaire, V., & Gaucheron, F. (2008). Effects of storage temperature on physico-chemical characteristics of semi-skimmed UHT milk. Food Hydrocolloids, 22(1), 130–143. https://doi.org/10.1016/j.foodhyd.2007.04.007.
Grewal, M. K., Chandrapala, J., Donkor, O., Apostolopoulos, V., Stojanovska, L., & Vasiljevic, T. (2017). Fourier transform infrared spectroscopy analysis of physicochemical changes in UHT milk during accelerated storage. International Dairy Journal, 66, 99–107. https://doi.org/10.1016/j.idairyj.2016.11.014.
Hammami, M., Rouissi, H., Salah, N., Selmi, H., Al-Otaibi, M., Blecker, C., & Karoui, R. (2010). Fluorescence spectroscopy coupled with factorial discriminant analysis technique to identify sheep milk from different feeding systems. Food Chemistry, 122, 1344–1350.
Hansen, E., & Skibsted, L. H. (2000). Light-induced oxidative changes in a model dairy spread. Wavelength dependence of quantum yields. https://doi.org/10.1021/JF991232O.
Herbert, S., Riaublanc, A., Bouchet, B., Gallant, D. J., & Dufour, E. (1999). Fluorescence spectroscopy investigation of acid-or rennet-induced coagulation of milk. Journal of Dairy Science, 82(10), 2056–2062. https://doi.org/10.3168/jds.S0022-0302(99)75446-9.
Herbert, S., Riou, N. M., Devaux, M. F., Riaublanc, A., Bouchet, B., Gallant, D. J., & Dufour, E. (2000). Monitoring the identity and the structure of soft cheeses by fluorescence spectroscopy. Le Lait, 80(6), 621–634. https://doi.org/10.1051/lait:2000149.
Huang, R., Choe, E., & Min, D. b. (2004). Kinetics for singlet oxygen formation by riboflavin photosensitization and the reaction between riboflavin and singlet oxygen. Journal of Food Science, 69(9), C726–C732. https://doi.org/10.1111/j.1365-2621.2004.tb09924.x.
Intawiwat, N., Pettersen, M. K., Rukke, E. O., Meier, M. A., Vogt, G., Dahl, A. V., Skaret, J., Keller, D., & Wold, J. P. (2010). Effect of different colored filters on photooxidation in pasteurized milk. Journal of Dairy Science, 93(4), 1372–1382. https://doi.org/10.3168/jds.2009-2542.
Jha, A., Murli, Patel, A. A., Gopal, T. K. S., & Ravishankar, C. N. (2012). Development of a process for shelf stable dairy dessert dalia and its physico-chemical properties. LWT - Food Science and Technology, 49(1), 80–88. https://doi.org/10.1016/j.lwt.2012.05.004.
Kamal, M., & Karoui, R. (2017). Monitoring of mild heat treatment of camel milk by front-face fluorescence spectroscopy. LWT - Food Science and Technology, 79, 586–593. https://doi.org/10.1016/j.lwt.2016.11.013.
Karoui, R., & Dufour, E. (2003). Dynamic testing rheology and fluorescence spectroscopy investigations of surface to centre differences in ripened soft cheeses. International Dairy Journal, 13(12), 973–985. https://doi.org/10.1016/S0958-6946(03)00121-3.
Karoui, R., Laguet, A., & Dufour, E. (2003). Fluorescence spectroscopy: a tool for the investigation of cheese melting—correlation with rheological characteristics. Le Lait, 83(3), 251–264. https://doi.org/10.1051/lait:2003014.
Karoui, R., Dufour, E., & De Baerdemaeker, J. (2006a). Front face fluorescence spectroscopy coupled with chemometric tools for monitoring the oxidation of semi-hard cheeses throughout ripening. Food Chemistry, 101(3), 1305–1314. https://doi.org/10.1016/j.foodchem.2006.01.028.
Karoui, R., Mouazen, A. M., Dufour, E., Pillonel, L., Picque, D., Bosset, J.-O., & De Baerdemaeker, J. (2006b). Mid-infrared spectrometry: a tool for the determination of chemical parameters in Emmental cheeses produced during winter. Le Lait, 86(1), 83–97.
Karoui, R., Dufour, E., & De Baerdemaeker, J. (2007). Monitoring the molecular changes by front face fluorescence spectroscopy throughout ripening of a semi-hard cheese. Food Chemistry, 104(1), 409–420. https://doi.org/10.1016/j.foodchem.2006.09.020.
Karoui, R., De Baerdemaeker, J., & Dufour, E. (2008). A comparison and joint use of mid infrared and fluorescence spectroscopic methods for differentiating between manufacturing processes and sampling zones of ripened soft cheeses. European Food Research and Technology, 226(4), 861–870. https://doi.org/10.1007/s00217-007-0608-x.
Kokkinidou, S., & Peterson, D. G. (2014). Control of Maillard-type off-flavor development in ultrahigh-temperature- processed bovine milk by phenolic chemistry. Journal of Agricultural and Food Chemistry, 62(32), 8023–8033. https://doi.org/10.1021/jf501919y.
Kozak, W., & Samotyja, U. (2013). The use of oxygen content determination method based on fluorescence quenching for rapeseed oil shelf-life assessment. Food Control, 33(1), 162–165. https://doi.org/10.1016/j.foodcont.2013.02.028.
Kulmyrzaev, A. A., Levieux, D., & Dufour, É. (2005). Front-face fluorescence spectroscopy allows the characterization of mild heat treatments applied to milk. Relations with the denaturation of milk proteins. Journal of Agricultural and Food Chemistry, 53(3), 502–507. https://doi.org/10.1021/jf049224h.
Lakowicz, J. R. (2006). Principles of fluorescence spectroscopy. Berlin: Springer.
Leriche, F., Bordessoules, A., Fayolle, K., Karoui, R., Laval, K., Leblanc, L., & Dufour, E. (2004). Alteration of raw-milk cheese by Pseudomonas spp.: Monitoring the sources of contamination using fluorescence spectroscopy and metabolic profiling. Journal of Microbiological Methods, 59(1), 33–41. https://doi.org/10.1016/j.mimet.2004.05.009.
Maldonado, J. A., Bruins, R. B., Yang, T., Wright, A., Dunne, C. P., & Karwe, M. V. (2015). Browning and ascorbic acid degradation in meals ready-to-eat pear rations in accelerated shelf life. Journal of Food Processing and Preservation, 39(6), 2035–2042. https://doi.org/10.1111/jfpp.12446.
Mestdagh, F., De Meulenaer, B., De Clippeleer, J., Devlieghere, F., & Huyghebaert, A. (2005). Protective influence of several packaging materials on light oxidation of milk. Journal of Dairy Science, 88(2), 499–510. https://doi.org/10.3168/jds.S0022-0302(05)72712-0.
Miquel Becker, E., Christensen, J., Frederiksen, C. S., Haugaard, V. K., Riou, N. M., Dufour, E., & Dufour, E. (2003). Front-face fluorescence spectroscopy and chemometrics in analysis of yogurt: rapid analysis of riboflavin. Journal of Dairy Science, 86(8), 2508–2515. https://doi.org/10.3168/jds.S0022-0302(03)73845-4.
Moufle, A.-L., Jamet, J., & Karoui, R. (2017). Impact of temperature cycling and isothermal storage on the quality of acidic and neutral shelf-stable dairy desserts packaged in flexible pouches. Food and Bioprocess Technology, 11(2), 1–19. https://doi.org/10.1007/s11947-017-2018-9.
Nicoli, M. C. (2012). An introduction to food shelf life: definitions, basic concepts, and regulatory aspects. In M. C. Nicoli (Ed.), Shelf life assessment of food (pp. 1–16). Boca Raton: CRC Press.
Ramírez-Sucre, M. O., & Vélez-Ruiz, J. F. (2014). Effect of formulation and storage on physicochemical and flow properties of custard flavored with caramel jam. Journal of Food Engineering, 142, 221–227. https://doi.org/10.1016/j.jfoodeng.2014.06.013.
Richards, M., De Kock, H. L., & Buys, E. M. (2014). Multivariate accelerated shelf-life test of low fat UHT milk. International Dairy Journal, 36(1), 38–45. https://doi.org/10.1016/j.idairyj.2013.12.012.
Semagoto, H. M., Liu, D., Koboyatau, K., Hu, J., Lu, N., Liu, X., Regenstein, J. M., & Zhou, P. (2014). Effects of UV induced photo-oxidation on the physicochemical properties of milk protein concentrate. Food Research International, 62, 580–588. https://doi.org/10.1016/j.foodres.2014.04.012.
Sila, D. N., Van Buggenhout, S., Duvetter, T., Fraeye, I., De Roeck, A., Van Loey, A., & Hendrickx, M. (2009). Pectins in processed fruits and vegetables: part II-structure function relationships. Comprehensive Reviews in Food Science and Food Safety, 8(2), 105–117. https://doi.org/10.1111/j.1541-4337.2009.00072.x.
Simpson, B. K. (2012). Food biochemistry and food processing. Hoboken: Wiley-Blackwell.
Smet, K., Raes, K., De Block, J., Herman, L., Dewettinck, K., & Coudijzer, K. (2008). A change in antioxidative capacity as a measure of onset to oxidation in pasteurized milk. International Dairy Journal, 18(5), 520–530. https://doi.org/10.1016/j.idairyj.2007.11.012.
Smet, K., De Block, J., De Campeneere, S., De Brabander, D., Herman, L., Raes, K., et al. (2009). Oxidative stability of UHT milk as influenced by fatty acid composition and packaging. International Dairy Journal, 19(6–7), 372–379. https://doi.org/10.1016/j.idairyj.2009.02.006.
Verbeken, D., Bael, K., Thas, O., & Dewettinck, K. (2006). Interactions between κ-carrageenan, milk proteins and modified starch in sterilized dairy desserts. International Dairy Journal, 16(5), 482–488. https://doi.org/10.1016/j.idairyj.2005.06.006.
Weerawatanakorn, M., Wu, J. C., Pan, M. H., & Ho, C. T. (2015). Reactivity and stability of selected flavor compounds. Journal of Food and Drug Analysis, 23(2), 176–190. https://doi.org/10.1016/j.jfda.2015.02.001.
Wold, J. P., Jørgensen, K., & Lundby, F. (2002). Nondestructive measurement of light-induced oxidation in dairy products by fluorescence spectroscopy and imaging. Journal of Dairy Science, 85(7), 1693–1704. https://doi.org/10.3168/jds.S0022-0302(02)74242-2.
Zhang, W., Xiao, S., & Ahn, D. U. (2013). Protein oxidation: basic principles and implications for meat quality. Critical Reviews in Food Science and Nutrition, 53(11), 1191–1201. https://doi.org/10.1080/10408398.2011.577540.
Acknowledgements
This work has been carried out in the framework of Alibiotech project which is financed by the European Union, French State, and the French Region of Hauts-de-France.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moufle, AL., François, PA., Jamet, J. et al. Accelerated Aging Test of Sterilized Acidic Pudding: Combined Effects of Temperature, Headspace Volume, and Agitation. Food Bioprocess Technol 11, 1286–1299 (2018). https://doi.org/10.1007/s11947-018-2100-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-018-2100-y