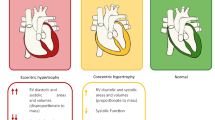
Opinion statement
Medical care of heart failure (HF) begins with the determination of the cause of the heart failure and diagnosing potential reversible causes (i.e., coronary heart disease, hyperthyroidism, etc.). Medical therapy includes pharmacological and nonpharmacological strategies that limit and/or reverse the signs and symptoms of HF. Initial behavior modification includes dietary sodium and fluid restriction to avoid weight gain; and encouraging physical activity when appropriate. Optimization of medical therapy is the first line of treatment that includes the use of diuretics, vasodilators (i.e., ACE inhibitors or ARBs), beta blockers, and potentially inotropic agents and anticoagulation depending on the patient’s severity of heart failure and LV dysfunction. As heart failure advances despite optimized medical management, cardiac resynchronization therapy (CRT), and implantable cardioverter defibrillators (ICDs) are appropriate device therapies. The development of progressive end-stage HF, despite maximal medical therapy, necessitates the consideration of mechanical circulatory devices such as ventricular assist devices (VADs) either as a bridge to heart transplantation or as destination therapy. Despite the advances in the treatment of heart failure, there is still a large morbidity and mortality associated with HF, thus the need to develop newer strategies for the treatment of HF.
Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Mozaffarian D et al. Executive summary: heart disease and stroke statistics-2016 update: a report from the American Heart Association. Circulation. 2016;133(4):447–54.
Kemp CD, Conte JV. The pathophysiology of heart failure. Cardiovasc Pathol. 2012;21(5):365–71.
von Lueder TG, Krum H. New medical therapies for heart failure. Nat Rev Cardiol. 2015;12(12):730–40.
Bigger Jr JT. Prophylactic use of implanted cardiac defibrillators in patients at high risk for ventricular arrhythmias after coronary-artery bypass graft surgery. Coronary Artery Bypass Graft (CABG) Patch Trial Investigators. N Engl J Med. 1997;337(22):1569–75.
Buxton AE et al. A randomized study of the prevention of sudden death in patients with coronary artery disease. Multicenter Unsustained Tachycardia Trial Investigators. N Engl J Med. 1999;341(25):1882–90.
Moss AJ et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002;346(12):877–83. Key trial demonstrating the utility of IDCs as primary prevention of preventing SCD in patients coronary artery disease and LV dysfunction.
Kadish A et al. Prophylactic defibrillator implantation in patients with nonischemic dilated cardiomyopathy. N Engl J Med. 2004;350(21):2151–8. Key trial demonstrating the utility of IDCs as primary prevention of preventing SCD in patients nonischemic dilated cardiomyopathy.
Hohnloser SH et al. Prophylactic use of an implantable cardioverter-defibrillator after acute myocardial infarction. N Engl J Med. 2004;351(24):2481–8.
Bardy GH et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med. 2005;352(3):225–37.
Steinbeck G et al. Defibrillator implantation early after myocardial infarction. N Engl J Med. 2009;361(15):1427–36.
A comparison of antiarrhythmic-drug therapy with implantable defibrillators in patients resuscitated from near-fatal ventricular arrhythmias. The Antiarrhythmics versus Implantable Defibrillators (AVID) Investigators. N Engl J Med. 1997; 337(22): 1576–83.
Kuck KH et al. Randomized comparison of antiarrhythmic drug therapy with implantable defibrillators in patients resuscitated from cardiac arrest: the Cardiac Arrest Study Hamburg (CASH). Circulation. 2000;102(7):748–54.
Connolly SJ et al. Canadian implantable defibrillator study (CIDS): a randomized trial of the implantable cardioverter defibrillator against amiodarone. Circulation. 2000;101(11):1297–302.
Epstein AE et al. 2012 ACCF/AHA/HRS focused update incorporated into the ACCF/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2013;61(3):e6–75.
Moss AJ et al. Improved survival with an implanted defibrillator in patients with coronary disease at high risk for ventricular arrhythmia. Multicenter Automatic Defibrillator Implantation Trial Investigators. N Engl J Med. 1996;335(26):1933–40. Key trial demonstrating the benefits of ICD for secondary prevention of SCD.
Abraham WT, Hayes DL. Cardiac resynchronization therapy for heart failure. Circulation. 2003;108(21):2596–603.
Ukkonen H et al. Effect of cardiac resynchronization on myocardial efficiency and regional oxidative metabolism. Circulation. 2003;107(1):28–31.
Nelson GS et al. Left ventricular or biventricular pacing improves cardiac function at diminished energy cost in patients with dilated cardiomyopathy and left bundle-branch block. Circulation. 2000;102(25):3053–9.
Rose EA et al. Long-term use of a left ventricular assist device for end-stage heart failure. N Engl J Med. 2001;345(20):1435–43.
Slaughter MS et al. Advanced heart failure treated with continuous-flow left ventricular assist device. N Engl J Med. 2009;361(23):2241–51.
Xie A, Phan K, Yan TD. Durability of continuous-flow left ventricular assist devices: a systematic review. Ann Cardiothorac Surg. 2014;3(6):547–56.
McIlvennan CK et al. Clinical outcomes after continuous-flow left ventricular assist device: a systematic review. Circ Heart Fail. 2014;7(6):1003–13.
Park SJ et al. Outcomes in advanced heart failure patients with left ventricular assist devices for destination therapy. Circ Heart Fail. 2012;5(2):241–8.
Yamaguchi A et al. Left ventricular reconstruction benefits patients with dilated ischemic cardiomyopathy. Ann Thorac Surg. 2005;79(2):456–61.
Athanasuleas CL et al. Surgical ventricular restoration in the treatment of congestive heart failure due to post-infarction ventricular dilation. J Am Coll Cardiol. 2004;44(7):1439–45.
Jones RH et al. Coronary bypass surgery with or without surgical ventricular reconstruction. N Engl J Med. 2009;360(17):1705–17.
Isomura T et al. Volume reduction rate by surgical ventricular restoration determines late outcome in ischaemic cardiomyopathy. Eur J Heart Fail. 2011;13(4):423–31.
Di Donato M, Castelvecchio S, Menicanti L. End-systolic volume following surgical ventricular reconstruction impacts survival in patients with ischaemic dilated cardiomyopathy. Eur J Heart Fail. 2010;12(4):375–81.
Buckberg G, Athanasuleas C, Conte J. Surgical ventricular restoration for the treatment of heart failure. Nat Rev Cardiol. 2012;9(12):703–16.
Fukamachi K, McCarthy PM. Initial safety and feasibility clinical trial of the myosplint device. J Card Surg. 2005;20(6):S43–7.
Mann DL et al. Clinical evaluation of the CorCap Cardiac Support Device in patients with dilated cardiomyopathy. Ann Thorac Surg. 2007;84(4):1226–35.
Sabbah HN et al. Reversal of chronic molecular and cellular abnormalities due to heart failure by passive mechanical ventricular containment. Circ Res. 2003;93(11):1095–101.
Saavedra WF et al. Reverse remodeling and enhanced adrenergic reserve from passive external support in experimental dilated heart failure. J Am Coll Cardiol. 2002;39(12):2069–76.
Starling RC et al. Sustained benefits of the CorCap Cardiac Support Device on left ventricular remodeling: three year follow-up results from the Acorn clinical trial. Ann Thorac Surg. 2007;84(4):1236–42.
Costanzo MR et al. Prospective evaluation of elastic restraint to lessen the effects of heart failure (PEERLESS-HF) trial. J Card Fail. 2012;18(6):446–58.
Singelyn JM et al. Catheter-deliverable hydrogel derived from decellularized ventricular extracellular matrix increases endogenous cardiomyocytes and preserves cardiac function post-myocardial infarction. J Am Coll Cardiol. 2012;59(8):751–63.
Yu J et al. Restoration of left ventricular geometry and improvement of left ventricular function in a rodent model of chronic ischemic cardiomyopathy. J Thorac Cardiovasc Surg. 2009;137(1):180–7.
Sabbah HN et al. Augmentation of left ventricular wall thickness with alginate hydrogel im- plants improves left ventricular function and prevents progressive remodeling in dogs with chronic heart failure. JACC Heart Fail. 2013;1:252–8.
Lee RJ et al. The feasibility and safety of Algisyl-LVR as a method of left ventricular augmentation in patients with dilated cardiomyopathy: initial first in man clinical results. Int J Cardiol. 2015;199:18–24.
Ankar SD et al. A prospective comparison of alginate-hydrogel with standard medical therapy to determine impact on functional capacity and clinical outcomes in patients with advanced heart failure (AUGMENT-HF trial). Eur Heart J. 2015;36(34):2297–309. First human trial to demonstrate the benefit of a tissue engineering approach for the treatment of HF.
Mann DL et al. One-year follow-up results from AUGMENT-HF: a multicentre randomized controlled clinical trial of the efficacy of left ventricular augmentation with Algisyl in the treatment of heart failure. Eur J Heart Fail. 2016;18(3):314–25. Demonstration that LV augmentation via a tissue engineering approach is durable and safe.
Cleland JG et al. The effect of cardiac resynchronization on morbidity and mortality in heart failure. N Engl J Med. 2005;352(15):1539–49.
Bristow MR et al. Cardiac-resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure. N Engl J Med. 2004;350(21):2140–50.
Young JB et al. Combined cardiac resynchronization and implantable cardioversion defibrillation in advanced chronic heart failure: the MIRACLE ICD Trial. Jama. 2003;289(20):2685–94. Key trial demonstrating the benefit of cardiac resynchonization therapy in patients with advanced heart failure.
Abraham WT et al. Cardiac resynchronization in chronic heart failure. N Engl J Med. 2002;346(24):1845–53.
Cazeau S et al. Effects of multisite biventricular pacing in patients with heart failure and intraventricular conduction delay. N Engl J Med. 2001;344(12):873–80.
Auricchio A et al. Long-term clinical effect of hemodynamically optimized cardiac resynchronization therapy in patients with heart failure and ventricular conduction delay. J Am Coll Cardiol. 2002;39(12):2026–33.
Tang AS et al. Cardiac-resynchronization therapy for mild-to-moderate heart failure. N Engl J Med. 2010;363(25):2385–95.
Zareba W et al. Effectiveness of Cardiac Resynchronization Therapy by QRS Morphology in the Multicenter Automatic Defibrillator Implantation Trial-Cardiac Resynchronization Therapy (MADIT-CRT). Circulation. 2011;123(10):1061–72.
Daubert C et al. Prevention of disease progression by cardiac resynchronization therapy in patients with asymptomatic or mildly symptomatic left ventricular dysfunction: insights from the European cohort of the REVERSE (Resynchronization Reverses Remodeling in Systolic Left Ventricular Dysfunction) trial. J Am Coll Cardiol. 2009;54(20):1837–46.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Praneeth Janaswamy, Tomos E. Walters, Babak Nazer, and Randall J. Lee each declare no potential conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Arrhythmia
Rights and permissions
About this article
Cite this article
Janaswamy, P., Walters, T.E., Nazer, B. et al. Current Treatment Strategies for Heart Failure: Role of Device Therapy and LV Reconstruction. Curr Treat Options Cardio Med 18, 57 (2016). https://doi.org/10.1007/s11936-016-0479-1
Published:
DOI: https://doi.org/10.1007/s11936-016-0479-1