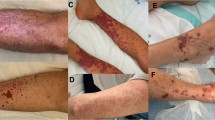
Abstract
Purpose of Review
The purpose of the study is to perform an update on the current knowledge on genetics, clinical manifestations, and therapy in immunoglobulin A vasculitis (IgAV) (Henoch-Schönlein purpura).
Recent Findings
A strong genetic predisposition in individuals with IgAV was confirmed. It was due to the association with the HLA class II region that in people of European background is mainly related to HLA-DRB1*01 allele. Recent reports support the claim that kidney disease is more common in adults than in children with IgAV. The clinical spectrum and outcome of adults with IgAV depends on the age of onset. Relapses are not uncommon in IgAV. The presence of renal impairment or proteinuria excretion exceeding 1 g/24 h at the time of disease diagnosis and the degree of renal damage on the kidney biopsy are the best predictors of end-stage renal failure in adults with IgAV. The levels of urinary IgA at the onset of the disease may predict a poor renal outcome. The use of prednisone does not seem to prevent persistent kidney disease in children with IgAV. No additional benefit of adding cyclophosphamide to glucocorticoids in adults with IgAV was found. Rituximab seems to be a promising therapy in the management of adults with IgAV.
Summary
In this overview, we focus on the genetics, clinical manifestations, and therapy of IgA vasculitis, emphasizing the main differences in the clinical expression of the disease between children and adults.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Jennette JC, Falk RJ, Bacon PA, et al. 2012 Revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013;65:1–11.
Gonzalez-Gay MA, Garcia-Porrua C, Pujol RM. Clinical approach to cutaneous vasculitis. Curr Opin Rheumatol. 2005;17:56–61.
González-Gay MA, García-Porrúa C. Epidemiology of the vasculitides. Rheum Dis Clin N Am. 2001;27:729–49.
Calviño MC, Llorca J, García-Porrúa C, Fernández-Iglesias JL, Rodríguez-Ledo P, Gonzalez-Gay MA. Henoch-Schönlein purpura in children from northwestern Spain: a 20-year epidemiologic and clinical study. Medicine (Baltimore). 2001;80:279–90.
García-Porrúa C, Calviño MC, Llorca J, Couselo JM, González-Gay MA. Henoch-Schönlein purpura in children and adults: clinical differences in a defined population. Semin Arthritis Rheum. 2002;32:149–56.
• Piram M, Maldini C, Biscardi S, et al. Incidence of IgA vasculitis in children estimated by four-source capture-recapture analysis: a population-based study. Rheumatology (Oxford). 2017;56:1358–66. According to these investigators, an important number of children with of IgAV remains unrecognized in epidemiological surveys.
•• López-Mejías R, Castañeda S, Genre F, et al. Genetics of immunoglobulin-A vasculitis (Henoch-Schönlein purpura): an updated review. Autoimmun Rev. 2018;S1568–9972(18):30012–0. https://doi.org/10.1016/j.autrev.2017.11.024. Comprehensive review of genetics in patients with IgA vasculitis. Besides an association with HLA-region, in particular with HLA-DRB1 alleles, other gene polymorphisms appear to be implicated in the susceptibility to IgAV as well as the risk of nephritis.
•• López-Mejías R, Genre F, Pérez BS, et al. HLA-DRB1 association with Henoch-Schonlein purpura. Arthritis Rheumatol. 2015;67:823–7. This study confirms the association of IgA with HLA-DRB1*01 in Caucasians. Data from this study indicate the presence of protective alleles against the development of IgA vasculitis, such as the HLA-DRB1*03, in Caucasians
• López-Mejías R, Genre F, Pérez BS, et al. Association of HLA-B*41:02 with Henoch-Schönlein Purpura (IgA Vasculitis) in Spanish individuals irrespective of the HLA-DRB1 status. Arthritis Res Ther. 2015;17:102. Association of IgA vasculitis in Spanish individuals with HLA-class I, in particular with HLA-B*41:02 independently of the previously reported associated with HLA-DRB1*01:03.
•• López-Mejías R, Carmona FD, Castañeda S, et al. A genome-wide association study suggests the HLA Class II region as the major susceptibility locus for IgA vasculitis. Sci Rep. 2017;7:5088. This is the first genome wide association study n IgA vasculitis. This study shows that IgA vasculitis is an archetypal HLA class II disease. Other gene polymorphisms located outside this region may also account for the susceptibility to IgA vasculitis.
Amoli MM, Thomson W, Hajeer AH, et al. Interleukin 1 receptor antagonist gene polymorphism is associated with severe renal involvement and renal sequelae in Henoch-Schönlein purpura. J Rheumatol. 2002;29:1404–7.
Amoli MM, Thomson W, Hajeer AH, et al. Interleukin 8 gene polymorphism is associated with increased risk of nephritis in cutaneous vasculitis. J Rheumatol. 2002;29:2367–70.
Rueda B, Perez-Armengol C, Lopez-Lopez S, Garcia-Porrua C, Martín J, Gonzalez-Gay MA. Association between functional haplotypes of vascular endothelial growth factor and renal complications in Henoch-Schönlein purpura. J Rheumatol. 2006;33:69–73.
Martin J, Paco L, Ruiz MP, et al. Inducible nitric oxide synthase polymorphism is associated with susceptibility to Henoch-Schönlein purpura in northwestern Spain. J Rheumatol. 2005;32:1081–5.
López-Mejías R, Sevilla Pérez B, Genre F, et al. Lack of association between IL6 gene and Henoch-Schönlein purpura. Clin Exp Rheumatol. 2014;32(3 Suppl 82):S141–2.
López-Mejías R, Genre F, Remuzgo-Martínez S, et al. Role of PTPN22 and CSK gene polymorphisms as predictors of susceptibility and clinical heterogeneity in patients with Henoch-Schönlein purpura (IgA vasculitis). Arthritis Res Ther. 2015;17:286.
• Calvo-Río V, Loricera J, Mata C, et al. Henoch-Schönlein purpura in northern Spain: clinical spectrum of the disease in 417 patients from a single center. Medicine (Baltimore). 2014;93:106–13. Study supporting the claim that IgA vasculitis is mainly a childhood disease. Data from this series of unselected patients showed that the outcome is good in most cases, depending largely on renal involvement.
• Kang Y, Park JS, Ha YJ, et al. Differences in clinical manifestations and outcomes between adult and child patients with Henoch-Schönlein purpura. J Korean Med Sci. 2014;29:198–203. Study in Korean patients that further supports the worse outcome and the higher frequency of chronic renal failure in adults when compared with children with IgA vasculitis
• Gupta V, Aggarwal A, Gupta R, et al. Differences between adult and pediatric onset Henoch-Schonlein purpura from North India. Int J Rheum Dis 2017. https://doi.org/10.1111/1756-185X.13221. Unlike most studies, in this study from Northern India no clear differences in the risk of nephritis and outcome were seen between children and adults with IgA vasculitis.
• González-Gay MA, Blanco R, Castañeda S. Henoch-Schönlein purpura (IgA vasculitis): the paradox of the different incidence and clinical spectrum in children and adults. Clin Exp Rheumatol. 2017;35(Suppl 103):3–4. Article addressing the paradox of the lower frequency and worse prognosis of IgA vasculitis in adulthood when compared with that of childhood
•• Chan H, Tang YL, Lv XH, et al. Risk factors associated with renal involvement in childhood Henoch-Schönlein Purpura: a meta-analysis. PLoS One. 2016;11:e0167346. A meta-analysis that assessed the predictors of nephritis in 2398 children with IgA vasculitis. Male gender, an age older than 10 years old, presence of severe manifestations or persistent purpura were associated with higher risk of nephritis. It was also the case for those who had relapses, leukocytosis, thrombocytosis; elevated antistreptolysin O titer and low C3.
•• Hong S, Ahn SM, Lim DH, et al. Late-onset IgA vasculitis in adult patients exhibits distinct clinical characteristics and outcomes. Clin Exp Rheumatol. 2016;34(3 Suppl 97):S77–83. The clinical spectrum and outcome of adults with IgA vasculitis depends on the age of onset. Individuals aged 60 years and older age have more commonly nephritis, diabetes and hypertension at the onset of the vasculitis and are more commonly associated with malignancy. Late-onset adults with IgA vasculitis have an increased risk of chronic kidney disease, end-stage renal failure and death than the patients with IgA vasculitis older than 20 and younger than 60 years.
Byun JW, Song HJ, Kim L, Shin JH, Choi GS. Predictive factors of relapse in adults with Henoch-Schönlein purpura. Am J Dermatopathol. 2012;34:139–44.
Prais D, Amir J, Nussinovitch M. Recurrent Henoch-Schönlein purpura in children. J Clin Rheumatol. 2007;13:25–8.
• Calvo-Río V, Hernández JL, Ortiz-Sanjuán F, et al. Relapses in patients with Henoch-Schönlein purpura: analysis of 417 patients from a single center. Medicine (Baltimore). 2016;95:e4217. Relapses occurred in almost one-third of 417 unselected patients with IgA vascultis. The clinical features at the time of the relapses were similar to those found at the time of disease diagnosis. Patients presenting with joint or gastrointestinal manifestations at the time of diagnosis of IgA vasculitis had more commonly relapses.
• St John J, Vedak P, Garza-Mayers AC, Hoang MP, Nigwekar SU, Kroshinsky D. Location of skin lesions in Henoch-Schönlein purpura and its association with significant renal involvement. J Am Acad Dermatol. 2018;78:115–20. A retrospective study suggesting that the skin lesion distribution on the extremities may be a predictor of long-term renal involvement in adults with IgA vasculitis.
García-Porrúa C, González-Louzao C, Llorca J, González-Gay MA. Predictive factors for renal sequelae in adults with Henoch-Schönlein purpura. J Rheumatol. 2001;28:1019–24.
•• Audemard-Verger A, Pillebout E, Guillevin L, Thervet E, Terrier B. IgA vasculitis (Henoch-Shönlein purpura) in adults: diagnostic and therapeutic aspects. Autoimmun Rev. 2015;14:579–85. The presence of renal impairment or proteinuria excretion exceeding 1 g/24 hours at the time of disease diagnosis and the degree of renal damage on the kidney biopsy are the best predictors of end-stage renal failure in adults with IgA vasculitis.
• He X, Yin W, Ding Y, et al. Higher serum angiotensinogen is an indicator of IgA vasculitis with nephritis revealed by comparative proteomes analysis. PLoS One. 2015;10:e0130536. The authors performed a comparative analysis of serum proteomes and they found that angiotensinogen concentration correlates with nephritis in patients with IgA vasculitis and that it may be used as a potential marker for the progression of IgA vasculitis.
•• Pillebout E, Jamin A, Ayari H, et al. Biomarkers of IgA vasculitis nephritis in children. PLoS One. 2017;12:e0188718. Study aimed to identify serum and urinary biomarkers of nephritis in children with IgA vasculitis.
•• Berthelot L, Jamin A, Viglietti D, et al. Value of biomarkers for predicting immunoglobulin A vasculitis nephritis outcome in an adult prospective cohort. Nephrol Dial Transplant 2017. https://doi.org/10.1093/ndt/gfx300. Interesting study focused on the search of biomarkers of nephritis in adults with IgA vasculitis. The levels of urinary IgA at the onset of the disease predicted a poor renal outcome after 1 year of follow-up.
• Nagy GR, Kemény L. Bata-Csörgő Z. Neutrophil-to-lymphocyte ratio: a biomarker for predicting systemic involvement in adult IgA vasculitis patients. J Eur Acad Dermatol Venereol. 2017;31:1033–7. The neutrophil-to-lymphocyte ratio may be a potential prognostic factor of systemic involvement in adults with IgA vasculitis.
Ronkainen J, Koskimies O, Ala-Houhala M, et al. Early prednisone therapy in Henoch-Schönlein purpura: a randomized, double-blind, placebo-controlled trial. J Pediatr. 2006;149:241–7.
Weiss PF, Klink AJ, Localio R, et al. Corticosteroids may improve clinical outcomes during hospitalization for Henoch-Schönlein purpura. Pediatrics. 2010;126:674–81.
•• Hahn D, Hodson EM, Willis NS, Craig JC. Interventions for preventing and treating kidney disease in Henoch-Schönlein Purpura (HSP). Cochrane Database Syst Rev 2015; (8):CD005128. A review article showing that the use of prednisone and antiplatelet agents does not seem to prevent persistent kidney disease in children with IgA vasculitis. No evidence of benefit for cyclophosphamide therapy in children or adults with IgA vasculitis and severe kidney disease was found. The authors of this review concluded that data on mycophenolate mofetil or cyclosporine potential efficacy in IgA vasculitis nephritis are still scarce to draw strong conclusions.
Du Y, Hou L, Zhao C, Han M, Wu Y. Treatment of children with Henoch-Schönlein purpura nephritis with mycophenolate mofetil. Pediatr Nephrol. 2012;27:765–71.
• Hackl A, Becker JU, Körner LM, et al. Mycophenolate mofetil following glucocorticoid treatment in Henoch-Schönlein purpura nephritis: the role of early initiation and therapeutic drug monitoring. Pediatr Nephrol 2017. https://doi.org/10.1007/s00467-017-3846-6. According to this study based on 18 patients seen at a single center, mycophenolate mofetil may be safe and potentially effective as a secondary therapeutic option for children with nephritis in the setting of IgA vasculitis.
•• Audemard-Verger A, Pillebout E, Guillevin L, Thervet E, Terrier B. IgA vasculitis (Henoch-Schönlein purpura) in adults: diagnostic and therapeutic aspects. Autoimmun Rev. 2015;14:579–85. Interesting review focused on diagnostic and therapeutic aspects of IgA vasculitis in adults. The outcome of adults with IgA vasculitis is sometimes unexpected. There is an absence of correlation between the initial presentation and the long-term renal outcome. Treatment of severe involvement, including severe gastrointestinal complications or proliferative glomerulonephritis, remains controversial.
•• Audemard-Verger A, Terrier B, Dechartres A, et al. Characteristics and management of IgA vasculitis (Henoch-Schönlein) in adults: data From 260 patients included in a French multicenter retrospective survey. Arthritis Rheumatol. 2017;69:1862–70. Retrospective survey on 260 adults with IgA vasculitis included in a French multicenter study. Glucocorticoids alone constitute the first-line therapy in patients with IgA vasculitis. The benefit of adding cyclophosphamide to glucocorticoids in adults with IgA vasculitis remains unclear.
•• Maritati F, Fenoglio R, Pillebout E, et al. Rituximab for the treatment of adult-onset IgA vasculitis (Henoch-Schönlein). Arthritis Rheumatol. 2018;70:109–14. In a series of 22 adults with IgA vasculitis, the remission rate following rituximab therapy was 91%. Most patients had remission within six months of this therapy.
Rostoker G, Desvaux-Belghiti D, Pilatte Y, et al. Immunomodulation with low-dose immunoglobulins for moderate IgA nephropathy and Henoch-Schönlein purpura. Preliminary results of a prospective uncontrolled trial. Nephron. 1995;69:327–34.
Augusto JF, Sayegh J, Delapierre L, et al. Addition of plasma exchange to glucocorticosteroids for the treatment of severe Henoch-Schönlein purpura in adults: a case series. Am J Kidney Dis. 2012;59:663–9.
• Kawabe M, Yamamoto I, Komatsuzaki Y, et al. Recurrence and graft loss after renal transplantation in adults with IgA vasculitis. Clin Exp Nephrol. 2017;21:714–20. Recurrences occurred in almost a third of the adults with nephritis associated to IgA vasculitis who required renal transplantation. None of them experienced graft loss
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Vasculitis
Rights and permissions
About this article
Cite this article
González-Gay, M.A., López-Mejías, R., Pina, T. et al. IgA Vasculitis: Genetics and Clinical and Therapeutic Management. Curr Rheumatol Rep 20, 24 (2018). https://doi.org/10.1007/s11926-018-0735-3
Published:
DOI: https://doi.org/10.1007/s11926-018-0735-3