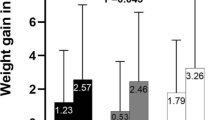
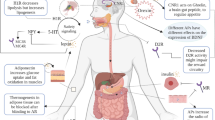
Abstract
Antipsychotic-induced weight gain (AIWG) is a prevalent side effect of antipsychotic treatment, particularly with second generation antipsychotics, such as clozapine and olanzapine. At this point, there is virtually nothing that can be done to predict who will be affected by AIWG. However, hope for the future of prediction lies with genetic risk factors. Many genes have been studied for their association with AIWG with a variety of promising findings. This review will focus on genetic findings in the last year and will discuss the first epigenetic and biomarker findings as well. Although there are significant findings in many other genes, the most consistently replicated findings are in the melanocortin 4 receptor (MC4R), the serotonin 2C receptor (HTR2C), the leptin, the neuropeptide Y (NPY) and the cannabinoid receptor 1 (CNR1) genes. The study of genetic risk variants poses great promise in creating predictive tools for side effects such as AIWG.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Ucok A, Gaebel W. Side effects of atypical antipsychotics: a brief overview. World Psychiatry. 2008;7(1):58–62.
Gebhardt S et al. Body weight gain induced by atypical antipsychotics: an extension of the monozygotic twin and sib pair study. J Clin Pharm Ther. 2010;35(2):207–11.
Altar CA et al. Clinical validity of cytochrome P450 metabolism and serotonin gene variants in psychiatric pharmacotherapy. Int Rev Psychiatry. 2013;25(5):509–33.
Lett TA et al. Pharmacogenetics of antipsychotic-induced weight gain: review and clinical implications. Mol Psychiatry. 2012;17(3):242–66.
Kao AC, Müller DJ. Genetics of antipsychotic-induced weight gain: update and current perspectives. Pharmacogenomics. 2013;14(16):2067–83. Comprehensive, detailed, and most recent review on AIWG.
Müller DJ, Chowdhury NI, Zai CC. The pharmacogenetics of antipsychotic-induced adverse events. Curr Opin Psychiatry. 2013;26(2):144–50.
Müller DJ et al. Pharmacogenetics of antipsychotic-induced weight gain. Pharmacol Res. 2004;49(4):309–29.
Müller DJ, Kennedy JL. Genetics of antipsychotic treatment emergent weight gain in schizophrenia. Pharmacogenomics. 2006;7(6):863–87.
Malhotra AK et al. Association between common variants near the melanocortin 4 receptor gene and severe antipsychotic drug-induced weight gain. Arch Gen Psychiatry. 2012;69(9):904–12.
Chowdhury NI et al. Genetic association study between antipsychotic-induced weight gain and the melanocortin-4 receptor gene. Pharmacogenomics J. 2013;13(3):272–9.
Nurmi EL et al. Moderation of antipsychotic-induced weight gain by energy balance gene variants in the RUPP autism network risperidone studies. Transl Psychiatry. 2013;3:e274.
Roffeei SN et al. Association of FTO, LEPR and MTHFR gene polymorphisms with metabolic syndrome in schizophrenia patients receiving antipsychotics. Pharmacogenomics. 2014;15(4):477–85.
Czerwensky F, Leucht S, Steimer W. Association of the common MC4R rs17782313 polymorphism with antipsychotic-related weight gain. J Clin Psychopharmacol. 2013;33(1):74–9.
Czerwensky F, Leucht S, Steimer W. MC4R rs489693: a clinical risk factor for second generation antipsychotic-related weight gain? Int J Neuropsychopharmacol. 2013;16(9):2103–9. Independent replication of the rs489693 marker of MC4R in AIWG.
Brandl EJ et al. Association study of polymorphisms in leptin and leptin receptor genes with antipsychotic-induced body weight gain. Prog Neuropsychopharmacol Biol Psychiatry. 2012;38(2):134–41.
Srisawat U et al. Methylenetetrahydrofolate reductase (MTHFR) 677C/T polymorphism is associated with antipsychotic-induced weight gain in first-episode schizophrenia. Int J Neuropsychopharmacol. 2014;17(3):485–90.
Klerk M et al. MTHFR 677C– > T polymorphism and risk of coronary heart disease: a meta-analysis. JAMA. 2002;288(16):2023–31.
Refsum H et al. Homocysteine and cardiovascular disease. Annu Rev Med. 1998;49:31–62.
Garcia-Miss Mdel R et al. Folate, homocysteine, interleukin-6, and tumor necrosis factor alfa levels, but not the methylenetetrahydrofolate reductase C677T polymorphism, are risk factors for schizophrenia. J Psychiatr Res. 2010;44(7):441–6.
Levine J et al. Homocysteine-reducing strategies improve symptoms in chronic schizophrenic patients with hyperhomocysteinemia. Biol Psychiatry. 2006;60(3):265–9.
Muntjewerff JW et al. Homocysteine, methylenetetrahydrofolate reductase and risk of schizophrenia: a meta-analysis. Mol Psychiatry. 2006;11(2):143–9.
Roffeei SN et al. Association of ADRA2A and MTHFR gene polymorphisms with weight loss following antipsychotic switching to aripiprazole or ziprasidone. Hum Psychopharmacol. 2014;29(1):38–45.
Kao, A.C., et al., Methylenetetrahydrofolate reductase gene variants and antipsychotic-induced weight gain and metabolic disturbances. J Psychiatr Res, 2014.
Nowrouzi B et al. Finite mixture regression model analysis on antipsychotics induced weight gain: investigation of the role of the serotonergic genes. Eur Neuropsychopharmacol. 2013;23(3):224–8.
del Castillo N, B.Z.M., Tyler B, Ellingrod VL, Calarge C, 759C/T Variants of the Serotonin (5-HT2C) Receptor Gene and Weight Gain in Children and Adolescents in Long-Term Risperidone Treatment. Clinic Pharmacol Biopharmaceut 2013. 2(110).
Sicard MN et al. Polymorphisms of the HTR2C gene and antipsychotic-induced weight gain: an update and meta-analysis. Pharmacogenomics. 2010;11(11):1561–71.
Wallace TJ et al. Role of 5-HT(2C) receptor gene variants in antipsychotic-induced weight gain. Pharmgenomics Pers Med. 2011;4:83–93.
SLC6A4 solute carrier family 6 (neurotransmitter transporter), member 4 [ Homo sapiens (human) ]. April 8 2014 [cited 2014 April 9]; Available from: http://www.ncbi.nlm.nih.gov.ezproxy.lib.ryerson.ca/gene/6532.
Almoguera B et al. Association of common genetic variants with risperidone adverse events in a Spanish schizophrenic population. Pharmacogenomics J. 2013;13(2):197–204.
Bah J et al. Further exploration of the possible influence of polymorphisms in HTR2C and 5HTT on body weight. Metabolism. 2010;59(8):1156–63.
HRH1 histamine receptor H1 [ Homo sapiens (human) ]. April 8 2014 [cited 2014 April 9]; Available from: http://www.ncbi.nlm.nih.gov.ezproxy.lib.ryerson.ca/gene/?term=h1hr.
Wirshing DA et al. Novel antipsychotics: comparison of weight gain liabilities. J Clin Psychiatry. 1999;60(6):358–63.
Godlewska BR et al. Polymorphisms of the histamine receptor (H1HR) gene are not associated with olanzapine-induced weight gain. J Clin Psychopharmacol. 2013;33(3):436–7.
Jia G et al. N6-methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nat Chem Biol. 2011;7(12):885–7.
Frayling TM et al. A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science. 2007;316(5826):889–94.
Reynolds GP et al. The obesity risk gene FTO influences body mass in chronic schizophrenia but not initial antipsychotic drug-induced weight gain in first-episode patients. Int J Neuropsychopharmacol. 2013;16(6):1421–5.
Shing EC et al. Fat mass- and obesity-associated (FTO) gene and antipsychotic-induced weight gain: an association study. Neuropsychobiology. 2014;69(1):59–63.
Hu X et al. Association study of NRXN3 polymorphisms with schizophrenia and risperidone-induced bodyweight gain in Chinese Han population. Prog Neuropsychopharmacol Biol Psychiatry. 2013;43:197–202.
Bogan JS. Regulation of glucose transporter translocation in health and diabetes. Annu Rev Biochem. 2012;81:507–32.
Brandl EJ et al. Exploratory study on association of genetic variation in TBC1D1 with antipsychotic-induced weight gain. Hum Psychopharmacol. 2013;28(2):183–7. One of the first studies implicating genes from glucose metabolism, particularly TBC1D1.
Tiwari AK et al. A common polymorphism in the cannabinoid receptor 1 (CNR1) gene is associated with antipsychotic-induced weight gain in Schizophrenia. Neuropsychopharmacology. 2010;35(6):1315–24.
Tiwari AK et al. Association of a functional polymorphism in neuropeptide Y with antipsychotic-induced weight gain in schizophrenia patients. J Clin Psychopharmacol. 2013;33(1):11–7.
Gamber KM, Macarthur H, Westfall TC. Cannabinoids augment the release of neuropeptide Y in the rat hypothalamus. Neuropharmacology. 2005;49(5):646–52.
Cota D et al. The endogenous cannabinoid system affects energy balance via central orexigenic drive and peripheral lipogenesis. J Clin Invest. 2003;112(3):423–31.
Osei-Hyiaman D et al. Endocannabinoid activation at hepatic CB1 receptors stimulates fatty acid synthesis and contributes to diet-induced obesity. J Clin Invest. 2005;115(5):1298–305.
NDUFS1 NADH dehydrogenase (ubiquinone) Fe-S protein 1, 75 kDa (NADH-coenzyme Q reductase) [ Homo sapiens (human) ]. April 8 2014 [cited 2014 April 9]; Available from: http://www.ncbi.nlm.nih.gov.ezproxy.lib.ryerson.ca/gene/4719.
COX18 COX18 cytochrome C oxidase assembly factor [Homo sapiens (human)]. April 8 2014 [cited 2014 April 9]; Available from: http://www.ncbi.nlm.nih.gov.ezproxy.lib.ryerson.ca/gene/285521.
Gonçalves, V.F., et al., A Hypothesis-Driven Association Study of 28 Nuclear-Encoded Mitochondrial Genes with Antipsychotic-Induced Weight Gain in Schizophrenia. Neuropsychopharmacology, 2013.
Ishoy PL et al. Treatment of antipsychotic-associated obesity with a GLP-1 receptor agonist–protocol for an investigator-initiated prospective, randomised, placebo-controlled, double-blinded intervention study: the TAO study protocol. BMJ Open. 2014;4(1):e004158.
Brandl EJ et al. Genetic variation in the GCG and in the GLP1R genes and antipsychotic-induced weight gain. Pharmacogenomics. 2014;15(4):423–31. One of the first studies implicating genes from glucose metabolism, particularly GLP1 and GCG.
Petronis A et al. Psychiatric epigenetics: a new focus for the new century. Mol Psychiatry. 2000;5(4):342–6.
Riddihough G, Zahn LM. Epigenetics. What is epigenetics? Introduction. Science. 2010;330(6004):611.
Rutten BP, Mill J. Epigenetic mediation of environmental influences in major psychotic disorders. Schizophr Bull. 2009;35(6):1045–56.
Minet-Ringuet J et al. Alterations of lipid metabolism and gene expression in rat adipocytes during chronic olanzapine treatment. Mol Psychiatry. 2007;12(6):562–71.
Melka MG et al. The effects of olanzapine on genome-wide DNA methylation in the hippocampus and cerebellum. Clin Epigenetics. 2014;6(1):1.
Rawlings JS, Rosler KM, Harrison DA. The JAK/STAT signaling pathway. J Cell Sci. 2004;117(Pt 8):1281–3.
Pijet M et al. Leptin impairs myogenesis in C2C12 cells through JAK/STAT and MEK signaling pathways. Cytokine. 2013;61(2):445–54.
Burghardt KJ et al. DNA methylation in schizophrenia subjects: gender and MTHFR 677C/T genotype differences. Epigenomics. 2012;4(3):261–8.
Lott SA et al. The influence of metabolic syndrome, physical activity and genotype on catechol-O-methyl transferase promoter-region methylation in schizophrenia. Pharmacogenomics J. 2013;13(3):264–71.
Burghardt KJ, Ellingrod VL. Detection of metabolic syndrome in schizophrenia and implications for antipsychotic therapy : is there a role for folate? Mol Diagn Ther. 2013;17(1):21–30. Comprehensive review on epigenetic mechanisms potentially involved in AIWG, implicating modifications of the COMT and MTHFR genes, as well as discussing the role of supplementary folate.
Schwarz, E., et al., Investigation of molecular serum profiles associated with predisposition to antipsychotic-induced weight gain. World J Biol Psychiatry, 2013. Sep 3. [Epub ahead of print]. One of the first studies implicating specific molecular serum profiles as method for predicting AIWG.
Vozarova B et al. Circulating interleukin-6 in relation to adiposity, insulin action, and insulin secretion. Obes Res. 2001;9(7):414–7.
Lazar MA. How obesity causes diabetes: not a tall tale. Science. 2005;307(5708):373–5.
Febbraio MA, Rose-John S, Pedersen BK. Is interleukin-6 receptor blockade the Holy Grail for inflammatory diseases? Clin Pharmacol Ther. 2010;87(4):396–8.
Baron DN. Hypothyroidism; its aetiology and relation to hypometabolism, hypercholesterolaemia, and increase in body-weight. Lancet. 1956;271(6937):277–81.
Howland RH. A "glucose eater" drug as a therapeutic agent in psychiatry. J Psychosoc Nurs Ment Health Serv. 2013;51(9):13–6.
Ganguli R et al. Weight loss intervention for individuals with diabetes and serious mental illness: a pragmatic experiment in community settings. Can J Diabetes. 2013;37(6):434.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Tahireh A. Shams declares no conflict of interest.
Daniel J. Müller has two patents submitted: "Compositions and Methods for the Treatment and Prevention of Antipsychotic Drug–Induced Weight Gain" and "Genetic markers for antipsychotics weight gain and methods for use thereof".
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Funding
Brain & Behaviour Research Foundation Independent Investigator Award to DJM, Canadian Institutes of Health Research Operating Funds (MOP 89853) to DJM, Canadian Institutes of Health Research Michael Smith New Investigator Salary Prize for Research in Schizophrenia to DJM, and an Early Researcher Award by the Ministry of Research and Innovation of Ontario to DJM.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Genetic Disorders
Rights and permissions
About this article
Cite this article
Shams, T.A., Müller, D.J. Antipsychotic Induced Weight Gain: Genetics, Epigenetics, and Biomarkers Reviewed. Curr Psychiatry Rep 16, 473 (2014). https://doi.org/10.1007/s11920-014-0473-9
Published:
DOI: https://doi.org/10.1007/s11920-014-0473-9