Abstract
Purpose of Review
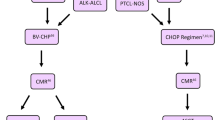
Given the rarity of anaplastic large cell lymphoma (ALCL), studies evaluating new therapies have typically grouped ALCL together with other peripheral T cell lymphomas (PTCL). Thus, the treatment paradigm for ALCL largely mirrors that of PTCL in general. In this review, we discuss the current standard of care as well as emerging therapies, including antibody-based drugs, in systemic ALCL as well as primary cutaneous and breast implant-associated ALCL.
Recent Findings
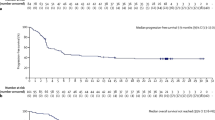
High CD30 expression in ALCL has allowed the use of brentuximab vedotin, an anti-CD30 antibody-drug conjugate, in both systemic and primary cutaneous ALCL. Recent clinical trials with brentuximab have shown substantial activity in the relapsed/refractory setting. A randomized phase III study is ongoing comparing brentuximab plus CHP (cyclophosphamide, doxorubicin, prednisone) with standard CHOP in the front-line setting.
Summary
The use of targeted therapies and other novel agents have improved outcomes for ALCL patients and in the future can complement or even replace the current standard of care and front-line treatment options.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Hapgood G, Savage KJ. Review article the biology and management of systemic anaplastic large cell lymphoma. Blood. 2015;126:17–26. Available from: http://www.bloodjournal.org/content/126/1/17.abstract%5Cn. http://www.ncbi.nlm.nih.gov/pubmed/25869285
Morris SW, Kirstein MN, Valentine MB, Dittmer KG, Shapiro DN, Saltman DL, et al. Fusion of a kinase gene, ALK, to a nucleolar protein gene, NPM, in non-Hodgkin’s lymphoma. Science. 1994;263:1281–4. [cited 2017 Sep 14], Available from: http://www.ncbi.nlm.nih.gov/pubmed/8122112
Al-Hamadani M, Habermann TM, Cerhan JR, Macon WR, Maurer MJ, Go RS. Non-Hodgkin lymphoma subtype distribution, geodemographic patterns, and survival in the US: a longitudinal analysis of the National Cancer Data Base from 1998 to 2011. Am J Hematol. 2015;90:790–5. https://doi.org/10.1002/ajh.24086.
Vose J, Armitage J, Weisenburger D, International T-Cell Lymphoma Project. International peripheral T-cell and natural killer/T-cell lymphoma study: pathology findings and clinical outcomes. J Clin Oncol. 2008;26:4124–30. https://doi.org/10.1200/JCO.2008.16.4558.
Savage KJ, Harris NL, Vose JM, Ullrich F, Jaffe ES, Connors JM, et al. ALK− anaplastic large-cell lymphoma is clinically and immunophenotypically different from both ALK+ ALCL and peripheral T-cell lymphoma, not otherwise specified: report from the International Peripheral T-Cell Lymphoma Project. Blood. 2008;111:5496–504. [cited 2017 Oct 1], Available from: http://www.ncbi.nlm.nih.gov/pubmed/18385450
Sibon D, Fournier M, Brière J, Lamant L, Haioun C, Coiffier B, et al. Long-term outcome of adults with systemic anaplastic large-cell lymphoma treated within the Groupe d’Etude des Lymphomes de l’Adulte trials. J Clin Oncol. 2012;30:3939–46. https://doi.org/10.1200/JCO.2012.42.2345.
Feldman AL, Dogan A, Smith DI, Law ME, Ansell SM, Johnson SH, et al. Discovery of recurrent t(6;7)(p25.3;q32.3) translocations in ALK-negative anaplastic large cell lymphomas by massively parallel genomic sequencing. Blood. 2011;117:915–9. https://doi.org/10.1182/blood-2010-08-303305.
Vasmatzis G, Johnson SH, Knudson RA, Ketterling RP, Braggio E, Fonseca R, et al. Genome-wide analysis reveals recurrent structural abnormalities of TP63 and other p53-related genes in peripheral T-cell lymphomas. Blood. 2012;120:2280–9. https://doi.org/10.1182/blood-2012-03-419937.
Parrilla Castellar ER, Jaffe ES, Said JW, Swerdlow SH, Ketterling RP, Knudson RA, et al. ALK-negative anaplastic large cell lymphoma is a genetically heterogeneous disease with widely disparate clinical outcomes. Blood [Internet] Am Soc Hematol. 2014;124:1473–80. [cited 2017 Oct 2], Available from: http://www.ncbi.nlm.nih.gov/pubmed/24894770
Brown RA, Fernandez-Pol S, Kim J. Primary cutaneous anaplastic large cell lymphoma. J Cutan Pathol. 2017;44:570–7. [cited 2017 Sep 14], Available from: http://www.ncbi.nlm.nih.gov/pubmed/28342276
Liu HL, Hoppe RT, Kohler S, Harvell JD, Reddy S, Kim YH. CD30+ cutaneous lymphoproliferative disorders: the Stanford experience in lymphomatoid papulosis and primary cutaneous anaplastic large cell lymphoma. J Am Acad Dermatol. 2003;49:1049–58. [cited 2017 Sep 14], Available from: http://www.ncbi.nlm.nih.gov/pubmed/14639383
Bekkenk MW, Geelen FA, van Voorst Vader PC, Heule F, Geerts ML, van Vloten WA, et al. Primary and secondary cutaneous CD30(+) lymphoproliferative disorders: a report from the Dutch Cutaneous Lymphoma Group on the long-term follow-up data of 219 patients and guidelines for diagnosis and treatment. Blood. 2000;95:3653–61. [cited 2017 Oct 8], Available from: http://www.ncbi.nlm.nih.gov/pubmed/10845893
Booken N, Goerdt S, Klemke C-D. Clinical spectrum of primary cutaneous CD30-positive anaplastic large cell lymphoma: an analysis of the Mannheim Cutaneous Lymphoma Registry. J Dtsch Dermatol. 2012;10:331–9. https://doi.org/10.1111/j.1610-0387.2011.07794.x.
Kempf W, Pfaltz K, Vermeer MH, Cozzio A, Ortiz-Romero PL, Bagot M, et al. EORTC, ISCL, and USCLC consensus recommendations for the treatment of primary cutaneous CD30-positive lymphoproliferative disorders: lymphomatoid papulosis and primary cutaneous anaplastic large-cell lymphoma. Blood. 2011;118:4024–35. https://doi.org/10.1182/blood-2011-05-351346.
•• Prince HM, Kim YH, Horwitz SM, Dummer R, Scarisbrick J, Quaglino P, et al. Brentuximab vedotin or physician’s choice in CD30-positive cutaneous T-cell lymphoma (ALCANZA): an international, open-label, randomised, phase 3, multicentre trial. Lancet (Lond Engl). 2017;390:555–66. [cited 2017 Oct 22], Available from: http://linkinghub.elsevier.com/retrieve/pii/S0140673617312667. Potentially practice-changing data in CD30-positive cutaneous T-cell lymphoma treated with brentuximab veodotin.
Leberfinger AN, Behar BJ, Williams NC, Rakszawski KL, Potochny JD, Mackay DR, et al. Breast implant–associated anaplastic large cell lymphoma. JAMA Surg. 2017;17033. https://doi.org/10.1001/jamasurg.2017.4026.
Johnson L, O’Donoghue JM, McLean N, Turton P, Khan AA, Turner SD, et al. Breast implant associated anaplastic large cell lymphoma: The UK experience. Recommendations on its management and implications for informed consent. Eur J Surg Oncol. 2017;43:1393–401. [cited 2017 Nov 24], Available from: http://linkinghub.elsevier.com/retrieve/pii/S074879831730481X
Miranda RN, Aladily TN, Prince HM, Kanagal-Shamanna R, de Jong D, Fayad LE, et al. Breast implant–associated anaplastic large-cell lymphoma: long-term follow-up of 60 patients. J Clin Oncol. 2014;32:114–20. [cited 2017 Sep 14], Available from: http://www.ncbi.nlm.nih.gov/pubmed/24323027
Schmitz N, Trümper L, Ziepert M, Nickelsen M, Ho AD, Metzner B, et al. Treatment and prognosis of mature T-cell and NK-cell lymphoma: an analysis of patients with T-cell lymphoma treated in studies of the German High-Grade Non-Hodgkin Lymphoma Study Group. Blood. 2010;116:3418–25. https://doi.org/10.1182/blood-2010-02-270785.
Simon A, Peoch M, Casassus P, Deconinck E, Colombat P, Desablens B, et al. Upfront VIP-reinforced-ABVD (VIP-rABVD) is not superior to CHOP/21 in newly diagnosed peripheral T cell lymphoma. Results of the randomized phase III trial GOELAMS-LTP95. Br J Haematol. 2010;151:159–66. https://doi.org/10.1111/j.1365-2141.2010.08329.x.
Escalón MP, Liu NS, Yang Y, Hess M, Walker PL, Smith TL, et al. Prognostic factors and treatment of patients with T-cell non-Hodgkin lymphoma: the M. D. Anderson Cancer Center experience. Cancer. 2005;103:2091–8. https://doi.org/10.1002/cncr.20999.
Abramson JS, Feldman T, Kroll-Desrosiers AR, Muffly LS, Winer E, Flowers CR, et al. Peripheral T-cell lymphomas in a large US multicenter cohort: prognostication in the modern era including impact of frontline therapy. Ann Oncol Off J Eur Soc Med Oncol. 2014;25:2211–7. https://doi.org/10.1093/annonc/mdu443.
d’Amore F, Relander T, Lauritzsen GF, Jantunen E, Hagberg H, Anderson H, et al. Up-front autologous stem-cell transplantation in peripheral T-cell lymphoma: NLG-T-01. J Clin Oncol. 2012;30:3093–9. https://doi.org/10.1200/JCO.2011.40.2719.
Wilhelm M, Smetak M, Reimer P, Geissinger E, Ruediger T, Metzner B, et al. First-line therapy of peripheral T-cell lymphoma: extension and long-term follow-up of a study investigating the role of autologous stem cell transplantation. Blood Cancer J. 2016;6:e452. [cited 2017 Oct 1], Available from: http://www.ncbi.nlm.nih.gov/pubmed/27471868
Zhang X-M, Li Y-X, Wang W-H, Jin J, Wang S-L, Liu Y-P, et al. Favorable outcome with doxorubicin-based chemotherapy and radiotherapy for adult patients with early stage primary systemic anaplastic large-cell lymphoma. Eur J Haematol. 2013;90:195–201. https://doi.org/10.1111/ejh.12060.
Gkotzamanidou M, Papadimitriou CA. Peripheral T-cell lymphoma: the role of hematopoietic stem cell transplantation. Crit Rev Oncol Hematol. 2014;89:248–61. [cited 2017 Sep 17], Available from: http://linkinghub.elsevier.com/retrieve/pii/S1040842813001935
Nademanee A, Palmer JM, Popplewell L, Tsai N-C, Delioukina M, Gaal K, et al. High-dose therapy and autologous hematopoietic cell transplantation in peripheral T cell lymphoma (PTCL): analysis of prognostic factors. Biol Blood Marrow Transplant. 2011;17:1481–9. [cited 2017 Sep 24], Available from: http://linkinghub.elsevier.com/retrieve/pii/S1083879111001029
Jacobsen ED, Kim HT, Ho VT, Cutler CS, Koreth J, Fisher DC, et al. A large single-center experience with allogeneic stem-cell transplantation for peripheral T-cell non-Hodgkin lymphoma and advanced mycosis fungoides/Sezary syndrome. Ann Oncol Off J Eur Soc Med Oncol. 2011;22:1608–13. https://doi.org/10.1093/annonc/mdq698.
Le Gouill S, Milpied N, Buzyn A, De Latour RP, Vernant J-P, Mohty M, et al. Graft-versus-lymphoma effect for aggressive T-cell lymphomas in adults: a study by the Société Francaise de Greffe de Moëlle et de Thérapie Cellulaire. J Clin Oncol. 2008;26:2264–71. https://doi.org/10.1200/JCO.2007.14.1366.
Smith SM, Burns LJ, van Besien K, LeRademacher J, He W, Fenske TS, et al. Hematopoietic cell transplantation for systemic mature T-cell non-Hodgkin lymphoma. J Clin Oncol. 2013;31:3100–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23897963
Corradini P, Dodero A, Zallio F, Caracciolo D, Casini M, Bregni M, et al. Graft-versus-lymphoma effect in relapsed peripheral T-cell non-Hodgkin’s lymphomas after reduced-intensity conditioning followed by allogeneic transplantation of hematopoietic cells. J Clin Oncol. 2004;22:2172–6. https://doi.org/10.1200/JCO.2004.12.050.
Younes A, Bartlett NL, Leonard JP, Kennedy DA, Lynch CM, Sievers EL, et al. Brentuximab vedotin (SGN-35) for relapsed CD30-positive lymphomas. N Engl J Med. 2010;363:1812–21. https://doi.org/10.1056/NEJMoa1002965.
Pro B, Advani R, Brice P, Bartlett NL, Rosenblatt JD, Illidge T, et al. Brentuximab vedotin (SGN-35) in patients with relapsed or refractory systemic anaplastic large-cell lymphoma: results of a phase II study. J Clin Oncol. 2012;30:2190–6. https://doi.org/10.1200/JCO.2011.38.0402.
Pro B, Advani R, Brice P, Bartlett NL, Rosenblatt JD, Illidge T, et al. Five-year results of brentuximab vedotin in patients with relapsed or refractory systemic anaplastic large cell lymphoma. Blood. 2017;blood-2017-05-780049. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28974506.
Bartlett NL, Chen R, Fanale MA, Brice P, Gopal A, Smith SE, et al. Retreatment with brentuximab vedotin in patients with CD30-positive hematologic malignancies. J Hematol Oncol. 2014;7:24. https://doi.org/10.1186/1756-8722-7-24.
• Fanale MA, Horwitz SM, Forero-Torres A, Bartlett NL, Advani RH, Pro B, et al. Brentuximab vedotin in the front-line treatment of patients with CD30+ peripheral T-cell lymphomas: results of a phase I study. J Clin Oncol. 2014;32:3137–43. https://doi.org/10.1200/JCO.2013.54.2456. Potentially practice-changing data in CD30-positive peripheral T-cell lymphomas which form the basis of the ongoing ECHELON-2 phase 3 trial comparing standard CHOP to combination CHP plus BV in newly diagnosed CD30+ PTCLs.
O’Connor OA, Pro B, Pinter-Brown L, Bartlett N, Popplewell L, Coiffier B, et al. Pralatrexate in patients with relapsed or refractory peripheral T-cell lymphoma: results from the pivotal PROPEL study. J Clin Oncol. 2011;29:1182–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21245435
Coiffier B, Pro B, Prince HM, Foss F, Sokol L, Greenwood M, et al. Results from a pivotal, open-label, phase II study of romidepsin in relapsed or refractory peripheral T-cell lymphoma after prior systemic therapy. J Clin Oncol. 2012;30:631–6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22271479
O’Connor OA, Horwitz S, Masszi T, Van Hoof A, Brown P, Doorduijn J, et al. Belinostat in patients with relapsed or refractory peripheral T-cell lymphoma: results of the pivotal phase II BELIEF (CLN-19) study. J Clin Oncol. 2015;33:2492–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26101246
Gambacorti-Passerini C, Messa C, Pogliani EM. Crizotinib in anaplastic large-cell lymphoma. N Engl J Med. 2011;364:775–6. https://doi.org/10.1056/NEJMc1013224.
Gambacorti Passerini C, Farina F, Stasia A, Redaelli S, Ceccon M, Mologni L, et al. Crizotinib in advanced, chemoresistant anaplastic lymphoma kinase-positive lymphoma patients. J Natl Cancer Inst. 2014;106:djt378. https://doi.org/10.1093/jnci/djt378.
Lesokhin AM, Ansell SM, Armand P, Scott EC, Halwani A, Gutierrez M, et al. Nivolumab in patients with relapsed or refractory hematologic malignancy: preliminary results of a phase Ib study. J Clin Oncol. 2016;34:2698–704. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27269947
Ansell S, Gutierrez ME, Shipp MA, Gladstone D, Moskowitz A, Borello I, Popa-Mckiver M, Farsaci B, Zhu L, Lesokhin AM, Arman P. A phase 1 study of nivolumab in combination with ipilimumab for relapsed or refractory hematologic malignancies (CheckMate 039). Blood. 2016;128. Available from: http://www.bloodjournal.org/content/128/22/183.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they do not have conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on T Cell and Other Lymphoproliferative Malignancies
Rights and permissions
About this article
Cite this article
Vu, K., Ai, W. Update on the Treatment of Anaplastic Large Cell Lymphoma. Curr Hematol Malig Rep 13, 135–141 (2018). https://doi.org/10.1007/s11899-018-0436-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-018-0436-z