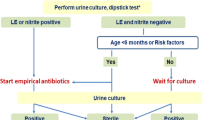
Abstract
Purpose of Review
The goal of this review is to summarize the current recommendations and evidence for the use of antibiotic prophylaxis in pelvic floor surgery. We provide a historical perspective on antimicrobial prophylaxis, review bacteria associated with pelvic floor surgery by site, and current antimicrobial prophylaxis recommendations from the American Urological Association (AUA) and American College of Obstetrics and Gynecology (ACOG).
Recent Findings
ACOG published updated recommendations for perioperative prophylaxis in June 2018 and the AUA in June 2019. There has been a complete de-emphasis on the use of fluoroquinolones; the preferred antimicrobials, when indicated, are first and second generation Cephalosporins alone for nearly all pelvic floor procedures via a vaginal or abdominal approach. Aside from allergy considerations, exceptions include reconstruction with use of large bowel, for which additional anaerobic coverage and mechanical and antibiotic bowel prep is recommended, and for select diagnostic and therapeutic procedures of the lower urinary tract, for which trimethoprim-sulfamethoxazole is recommended.
Summary
Pelvic floor surgery is unique due to its variable surgical approaches and the range of potential infectious sources to be considered (i.e., the skin, vagina, bladder, intestine). Urinary tract infection is the most common infectious complication across nearly all procedures, although its prevention is not simply related to antibiotic selection and duration. Antibiotic stewardship calls for judicious use, including non-use when risks are low. Although updated societal guidelines provide the foundation on which most perioperative antimicrobial recommendations should be based, surgeons must consider patient-specific factors (e.g., multidrug -resistant bacteriuria) and local antibiograms. Input from colleagues in pharmacy and infectious diseases may optimize antibiotic selection, dosing, and duration, particularly when non-guideline antibiosis appears to be indicated.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Surgical Site Infection (SSI) event. In: Network NHS, Prevention CfDCa, editors. Online2017.
•• Lightner DJ, Wymer K, Sanchez J, Kavoussi L. Best practice statement on urologic procedures and antimicrobial prophylaxis. J Urol. 2020;203(2):351–6. https://doi.org/10.1097/JU.0000000000000509This reference served as the primary resource for this review for antibiotic prophylaxis evidence and recommendations.
Lister J. On the antiseptic principle in the practice of surgery. Br Med J. 1867;2(351):246–8. https://doi.org/10.1136/bmj.2.351.246.
• Chodak GW, Plaut ME. Use of systemic antibiotics for prophylaxis in surgery: a critical review. Arch Surg. 1977;112(3):326–34. https://doi.org/10.1001/archsurg.1977.01370030098018This seminal reference provided the basis for current antibiotic prophylaxis rationale.
Black JD, de Haydu C, Fan L, Sheth SS. Surgical site infections in gynecology. Obstet Gynecol Surv. 2014;69(8):501–10. https://doi.org/10.1097/OGX.0000000000000102.
Pop-Vicas A, Musuuza JS, Schmitz M, Al-Niaimi A, Safdar N. Incidence and risk factors for surgical site infection post-hysterectomy in a tertiary care center. Am J Infect Control. 2017;45(3):284–7. https://doi.org/10.1016/j.ajic.2016.10.008.
Haraway AM, Clemens JQ, He C, Stroup C, Atiemo HO, Cameron AP. Differences in sacral neuromodulation device infection rates based on preoperative antibiotic selection. Int Urogynecol J. 2013;24(12):2081–5. https://doi.org/10.1007/s00192-013-2121-z.
Myer ENB, Petrikovets A, Slocum PD, Lee TG, Carter-Brooks CM, Noor N et al. Risk factors for explantation due to infection after sacral neuromodulation: a multicenter retrospective case-control study. Am J Obstet Gynecol. 2018;219(1):78 e1- e9. doi:https://doi.org/10.1016/j.ajog.2018.04.005.
Brueseke T, Livingston B, Warda H, Osann K, Noblett K. Risk factors for surgical site infection in patients undergoing sacral nerve modulation therapy. Female Pelvic Med Reconstr Surg. 2015;21(4):198–204. https://doi.org/10.1097/SPV.0000000000000183.
Uppal S, Harris J, Al-Niaimi A, Swenson CW, Pearlman MD, Reynolds RK, et al. Prophylactic antibiotic choice and risk of surgical site infection after hysterectomy. Obstet Gynecol. 2016;127(2):321–9. https://doi.org/10.1097/AOG.0000000000001245.
Sanaee MS, Hutcheon JA, Larouche M, Brown HL, Lee T, Geoffrion R. Urinary tract infection prevention after midurethral slings in pelvic floor reconstructive surgery: a systematic review and meta-analysis. Acta Obstet Gynecol Scand. 2019;98:1514–22. https://doi.org/10.1111/aogs.13661.
Vigil HR, Mallick R, Nitti VW, Lavallee LT, Breau RH, Hickling DR. Risk factors for urinary tract infection following mid urethral sling surgery. J Urol. 2017;197(5):1268–73. https://doi.org/10.1016/j.juro.2016.12.093.
Foxman B, Cronenwett AE, Spino C, Berger MB, Morgan DM. Cranberry juice capsules and urinary tract infection after surgery: results of a randomized trial. Am J Obstet Gynecol. 2015;213(2):194 e1–8. doi:https://doi.org/10.1016/j.ajog.2015.04.003.
Chung CP, Kuehl TJ, Harris SK, McBride MM, Larsen WI, Yandell PM, et al. Incidence and risk factors of postoperative urinary tract infection after uterosacral ligament suspension. Int Urogynecol J. 2012;23(7):947–50. https://doi.org/10.1007/s00192-012-1709-z.
Gehrich AP, Lustik MB, Mehr AA, Patzwald JR. Risk of postoperative urinary tract infections following midurethral sling operations in women undergoing hysterectomy. Int Urogynecol J. 2016;27(3):483–90. https://doi.org/10.1007/s00192-015-2861-z.
Fok CS, McKinley K, Mueller ER, Kenton K, Schreckenberger P, Wolfe A, et al. Day of surgery urine cultures identify urogynecologic patients at increased risk for postoperative urinary tract infection. J Urol. 2013;189(5):1721–4. https://doi.org/10.1016/j.juro.2012.11.167.
Lachiewicz MP, Moulton LJ, Jaiyeoba O. Pelvic surgical site infections in gynecologic surgery. Infect Dis Obstet Gynecol. 2015;2015:614950–8. https://doi.org/10.1155/2015/614950.
Steiner HL, Strand EA. Surgical-site infection in gynecologic surgery: pathophysiology and prevention. Am J Obstet Gynecol. 2017;217(2):121–8. https://doi.org/10.1016/j.ajog.2017.02.014.
Goede WJ, Lovely JK, Thompson RL, Cima RR. Assessment of prophylactic antibiotic use in patients with surgical site infections. Hosp Pharm. 2013;48(7):560–7. https://doi.org/10.1310/hpj4807-560.
•z Shapiro R, Laignel R, Kowcheck C, White V, Hashmi M. Modifying pre-operative antibiotic overuse in gynecologic surgery. Int J Health Care Qual Assur. 2018;31(5):400–5. https://doi.org/10.1108/IJHCQA-04-2017-0066This reference classified non-adherence to antibiotic prophylaxis recommendations.
Wright JD, Hassan K, Ananth CV, Herzog TJ, Lewin SN, Burke WM, et al. Use of guideline-based antibiotic prophylaxis in women undergoing gynecologic surgery. Obstet Gynecol. 2013;122(6):1145–53. https://doi.org/10.1097/AOG.0b013e3182a8a36a.
Joyce J, Langsjoen J, Sharadin C, Kuehl TJ, Larsen WI. Inappropriate use of antibiotics in patients undergoing gynecologic surgery. Proc (Bayl Univ Med Cent). 2017;30(1):30–2. https://doi.org/10.1080/08998280.2017.11929518.
Turner LC, Kantartzis K, Lowder JL, Shepherd JP. The effect of age on complications in women undergoing minimally invasive sacral colpopexy. Int Urogynecol J. 2014;25(9):1251–6. https://doi.org/10.1007/s00192-014-2391-0.
Kantartzis K, Sutkin G, Winger D, Wang L, Shepherd J. Introduction of laparoscopic sacral colpopexy to a fellowship training program. Int Urogynecol J. 2013;24(11):1877–81. https://doi.org/10.1007/s00192-013-2085-z.
Deffieux X, Letouzey V, Savary D, Sentilhes L, Agostini A, Mares P, et al. Prevention of complications related to the use of prosthetic meshes in prolapse surgery: guidelines for clinical practice. Eur J Obstet Gynecol Reprod Biol. 2012;165(2):170–80. https://doi.org/10.1016/j.ejogrb.2012.09.001.
Mangir N, Roman S, Chapple CR, MacNeil S. Complications related to use of mesh implants in surgical treatment of stress urinary incontinence and pelvic organ prolapse: infection or inflammation? World J Urol. 2020;38(1):73–80. https://doi.org/10.1007/s00345-019-02679-w.
•• ACOG Practice Bulletin No. 195: Prevention of infection after gynecologic procedures. Obstet Gynecol. 2018;131(6):e172–e89. https://doi.org/10.1097/AOG.0000000000002670This reference provided antibiotic recommendations and evidence for a number of procedures.
Theofanides MC, Sui W, Sebesta EM, Onyeji I, Matulay JT, Chung DE. Vesicovaginal fistulas in the developed world: an analysis of disease characteristics, treatments, and complications of surgical repair using the ACS-NSQIP database. Neurourol Urodyn. 2017;36(6):1622–8. https://doi.org/10.1002/nau.23167.
Morhason-Bello IO, Ojengbede OA, Adedokun BO, Okunlola MA, Oladokun A. Uncomplicated midvaginal vesico-vaginal fistula repair in Ibadan: a comparison of the abdominal and vaginal routes. Ann Ib Postgrad Med. 2008;6(2):39–43. https://doi.org/10.4314/aipm.v6i2.64051.
Abreu AL, Chopra S, Dharmaraja A, Djaladat H, Aron M, Ukimura O, et al. Robot-assisted bladder diverticulectomy. J Endourol. 2014;28(10):1159–64. https://doi.org/10.1089/end.2014.0149.
Du K, Mulroy EE, Wallis MC, Zhang C, Presson AP, Cartwright PC. Enterocystoplasty 30-day outcomes from National Surgical Quality Improvement Program Pediatric 2012. J Pediatr Surg. 2015;50(9):1535–9. https://doi.org/10.1016/j.jpedsurg.2015.04.009.
Wu SY, Kuo HC. A real-world experience with augmentation enterocystoplasty-high patient satisfaction with high complication rates. Neurourol Urodyn. 2018;37(2):744–50. https://doi.org/10.1002/nau.23339.
Osborn DJ, Dmochowski RR, Kaufman MR, Milam DF, Mock S, Reynolds WS. Cystectomy with urinary diversion for benign disease: indications and outcomes. Urology. 2014;83(6):1433–7. https://doi.org/10.1016/j.urology.2014.02.030.
Cohn JA, Large MC, Richards KA, Steinberg GD, Bales GT. Cystectomy and urinary diversion as management of treatment-refractory benign disease: the impact of preoperative urological conditions on perioperative outcomes. Int J Urol. 2014;21(4):382–6. https://doi.org/10.1111/iju.12284.
Pariser JJ, Anderson BB, Pearce SM, Han Z, Rodriguez JA, 3rd, Landon E et al. The effect of broader, directed antimicrobial prophylaxis including fungal coverage on perioperative infectious complications after radical cystectomy. Urol Oncol. 2016;34(3):121 e9–14. doi:https://doi.org/10.1016/j.urolonc.2015.10.007.
Togan T, Azap OK, Durukan E, Arslan H. The prevalence, etiologic agents and risk factors for urinary tract infection among spinal cord injury patients. Jundishapur J Microbiol. 2014;7(1):e8905. https://doi.org/10.5812/jjm.8905.
Yoon SB, Lee BS, Lee KD, Hwang SI, Lee HJ, Han ZA. Comparison of bacterial strains and antibiotic susceptibilities in urinary isolates of spinal cord injury patients from the community and hospital. Spinal Cord. 2014;52(4):298–301. https://doi.org/10.1038/sc.2014.10.
Nguyen JN, Yang ST. Perioperative outcomes after robotic versus vaginal surgery for pelvic organ prolapse. J Robot Surg. 2020;14(3):415–21. https://doi.org/10.1007/s11701-019-01006-0.
Altman D, Falconer C. Perioperative morbidity using transvaginal mesh in pelvic organ prolapse repair. Obstet Gynecol. 2007;109(2 Pt 1):303–8. https://doi.org/10.1097/01.AOG.0000250970.23128.63.
Hill AJ, Walters MD, Unger CA. Perioperative adverse events associated with colpocleisis for uterovaginal and posthysterectomy vaginal vault prolapse. Am J Obstet Gynecol. 2016;214(4):501 e1- e6. doi:https://doi.org/10.1016/j.ajog.2015.10.921.
Raup VT, Hess DS, Hanske J, Schmid M, Varda B, Das A, et al. Patient characteristics and perioperative outcomes of female urethral diverticulectomy: analysis of a multi-institutional prospective database. Urology. 2015;86(4):712–5. https://doi.org/10.1016/j.urology.2015.07.007.
Unger CA, Walters MD, Ridgeway B, Jelovsek JE, Barber MD, Paraiso MF. Incidence of adverse events after uterosacral colpopexy for uterovaginal and posthysterectomy vault prolapse. Am J Obstet Gynecol. 2015;212(5):603 e1–7. doi:https://doi.org/10.1016/j.ajog.2014.11.034.
Sori DA, Azale AW, Gemeda DH. Characteristics and repair outcome of patients with Vesicovaginal fistula managed in Jimma University teaching Hospital, Ethiopia. BMC Urol 2016;16(1):41. doi:https://doi.org/10.1186/s12894-016-0152-8.
Eilber KS, Kavaler E, Rodriguez LV, Rosenblum N, Raz S. Ten-year experience with transvaginal vesicovaginal fistula repair using tissue interposition. J Urol. 2003;169(3):1033–6. https://doi.org/10.1097/01.ju.0000049723.57485.e7.
Stanford EJ, Paraiso MF. A comprehensive review of suburethral sling procedure complications. J Minim Invasive Gynecol. 2008;15(2):132–45. https://doi.org/10.1016/j.jmig.2007.11.004.
Swartz M, Ching C, Gill B, Li J, Rackley R, Vasavada S, et al. Risk of infection after midurethral synthetic sling surgery: are postoperative antibiotics necessary? Urology. 2010;75(6):1305–8. https://doi.org/10.1016/j.urology.2009.11.081.
Doganay M, Cavkaytar S, Kokanali MK, Ozer I, Aksakal OS, Erkaya S. Risk factors for postoperative urinary tract infection following midurethral sling procedures. Eur J Obstet Gynecol Reprod Biol. 2017;211:74–7. https://doi.org/10.1016/j.ejogrb.2017.02.006.
Ingber MS, Vasavada SP, Firoozi F, Goldman HB. Incidence of perioperative urinary tract infection after single-dose antibiotic therapy for midurethal slings. Urology. 2010;76(4):830–4. https://doi.org/10.1016/j.urology.2010.05.038.
Richter HE, Albo ME, Zyczynski HM, Kenton K, Norton PA, Sirls LT, Kraus SR, Chai TC, Lemack GE, Dandreo KJ, Varner RE, Menefee S, Ghetti C, Brubaker L, Nygaard I, Khandwala S, Rozanski TA, Johnson H, Schaffer J, Stoddard AM, Holley RL, Nager CW, Moalli P, Mueller E, Arisco AM, Corton M, Tennstedt S, Chang TD, Gormley EA, Litman HJ, Urinary Incontinence Treatment Network. Retropubic versus transobturator midurethral slings for stress incontinence. N Engl J Med 2010;362(22):2066–2076. doi:https://doi.org/10.1056/NEJMoa0912658.
Harmanli O, Hong EK, Rubin R, Jones KA, Boyer RL, Metz S. Is antibiotic prophylaxis necessary for midurethral sling procedures? A series of 174 cases without preoperative antibiotics. Int Urogynecol J. 2012;23(5):621–3. https://doi.org/10.1007/s00192-011-1624-8.
Brubaker L, Norton PA, Albo ME, Chai TC, Dandreo KJ, Lloyd KL et al. Adverse events over two years after retropubic or transobturator midurethral sling surgery: findings from the Trial of Midurethral Slings (TOMUS) study. Am J Obstet Gynecol. 2011;205(5):498 e1–6. doi:https://doi.org/10.1016/j.ajog.2011.07.011.
•• Clennon EK, Martinez Acevedo A, Sajadi KP. Safety and effectiveness of zero antimicrobial prophylaxis protocol for outpatient cystourethroscopy. BJU Int. 2019;123(5A):E29–33. https://doi.org/10.1111/bju.14662This reference emphasized the need for antibiotic stewardship and the efficacy of doing so.
Garcia-Perdomo HA, Lopez H, Carbonell J, Castillo D, Catano JG, Seron P. Efficacy of antibiotic prophylaxis in patients undergoing cystoscopy: a randomized clinical trial. World J Urol. 2013;31(6):1433–9. https://doi.org/10.1007/s00345-013-1034-2.
Herr HW. Should antibiotics be given prior to outpatient cystoscopy? A plea to urologists to practice antibiotic stewardship. Eur Urol. 2014;65(4):839–42. https://doi.org/10.1016/j.eururo.2013.08.054.
•• Gregg JR, Bhalla RG, Cook JP, Kang C, Dmochowski R, Talbot TR, et al. An evidence-based protocol for antibiotic use prior to cystoscopy decreases antibiotic use without impacting post-procedural symptomatic urinary tract infection rates. J Urol. 2018;199(4):1004–10. https://doi.org/10.1016/j.juro.2017.10.038This reference emphasized the need for antibiotic stewardship and the efficacy of doing so.
•• Cameron AP, Campeau L, Brucker BM, Clemens JQ, Bales GT, Albo ME, et al. Best practice policy statement on urodynamic antibiotic prophylaxis in the non-index patient. Neurourol Urodyn. 2017;36(4):915–26. https://doi.org/10.1002/nau.23253This reference provided antibiotic recommendations and evidence for select procedures.
Yokoyama M, Fujii Y, Yoshida S, Saito K, Koga F, Masuda H, et al. Discarding antimicrobial prophylaxis for transurethral resection of bladder tumor: a feasibility study. Int J Urol. 2009;16(1):61–3. https://doi.org/10.1111/j.1442-2042.2008.02188.x.
Ibrahim AI, Rashid M. Comparison of local povidone-iodine antisepsis with parenteral antibacterial prophylaxis for prevention of infective complications of TURP: a prospective randomized controlled study. Eur Urol. 2002;41(3):250–6. https://doi.org/10.1016/s0302-2838(02)00013-1.
May A, Broggi E, Lorphelin H, Tabchouri N, Giretti G, Pereira H, et al. Comparison of the risk of postoperative infection between transurethral vaporesection and transurethral resection of the prostate. Lasers Surg Med. 2014;46(5):405–11. https://doi.org/10.1002/lsm.22240.
Mouttalib S, Khan S, Castel-Lacanal E, Guillotreau J, De Boissezon X, Malavaud B, et al. Risk of urinary tract infection after detrusor botulinum toxin A injections for refractory neurogenic detrusor overactivity in patients with no antibiotic treatment. BJU Int. 2010;106(11):1677–80. https://doi.org/10.1111/j.1464-410X.2010.09435.x.
Khan MH, Baldo O, Koenig P, Shaikh N. Use of prophylactic antibiotics for intra-vesicle Botox(R) injection. Neurourol Urodyn. 2017;36(3):828. https://doi.org/10.1002/nau.23034.
Nitti VW, Dmochowski R, Herschorn S, Sand P, Thompson C, Nardo C, et al. OnabotulinumtoxinA for the treatment of patients with overactive bladder and urinary incontinence: results of a phase 3, randomized, placebo controlled trial. J Urol. 2013;189(6):2186–93. https://doi.org/10.1016/j.juro.2012.12.022.
Houman J, Moradzadeh A, Patel DN, Asanad K, Anger JT, Eilber KS. What is the ideal antibiotic prophylaxis for intravesically administered Botox injection? A comparison of two different regimens. Int Urogynecol J. 2019;30(5):701–4. https://doi.org/10.1007/s00192-018-3721-4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights
All reported studies/experiments with human or animal subjects performed by the authors have been previously published and complied with all applicable ethical standards (including the Helsinki declaration and its amendments, institutional/national research committee standards, and international/national/institutional guidelines).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Reconstructed Bladder Function & Dysfunction
Rights and permissions
About this article
Cite this article
Kunkel, G.M., Cohn, J.A. Antibiotic Prophylaxis in Pelvic Floor Surgery. Curr Bladder Dysfunct Rep 15, 267–274 (2020). https://doi.org/10.1007/s11884-020-00601-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11884-020-00601-4