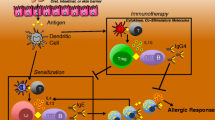
Abstract
There is a growing evidence that allergen immunotherapy (AIT) can provide significant and long-lasting clinical benefit for a number of allergic individuals. However, it is less clear if AIT results in clinical tolerance, which is characterized by a persistent state of clinical non-reactivity to allergens after therapy is finished. Addressing this knowledge gap is particularly relevant for patients undergoing AIT for food allergies, as anything less than complete tolerance could have potentially devastating consequences. An increasing number of studies, in particular those involving oral immunotherapy, are attempting to assess tolerance induction following AIT. Clinical tolerance does appear to be achievable in a subset of patients undergoing AIT, but whether this is equivalent to the type of tolerance observed in nonallergic individuals remains unknown. Developing established criteria for assessing tolerance induction, as well as the use of consistent terminology when describing clinical tolerance, will be important for determining the disease-modifying potential of AIT.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Cox L et al. Allergen immunotherapy: a practice parameter third update. J Allergy Clin Immunol. 2011;127(1 Suppl):S1–55.
Boyle RJ et al. Venom immunotherapy for preventing allergic reactions to insect stings. Cochrane Database Syst Rev. 2012;10:Cd008838.
Calderon MA, et al. Allergen injection immunotherapy for seasonal allergic rhinitis. Cochrane Database Syst Rev. 2007(1): p. Cd001936.
Abramson MJ, Puy RM, and Weiner JM. Injection allergen immunotherapy for asthma. Cochrane Database Syst Rev. 2010(8): p. Cd001186.
Radulovic S. et al. Sublingual immunotherapy for allergic rhinitis. Cochrane Database Syst Rev. 2010(12): p. Cd002893.
Moran TP, Vickery BP, Burks AW. Oral and sublingual immunotherapy for food allergy: current progress and future directions. Curr Opin Immunol. 2013;25(6):781–7.
Burks AW et al. Update on allergy immunotherapy: American Academy of Allergy, Asthma & Immunology/European Academy of Allergy and Clinical Immunology/PRACTALL consensus report. J Allergy Clin Immunol. 2013;131(5):1288–96.
Berin MC, Mayer L. Can we produce true tolerance in patients with food allergy? J Allergy Clin Immunol. 2013;131(1):14–22.
Novak N et al. Early suppression of basophil activation during allergen-specific immunotherapy by histamine receptor 2. J Allergy Clin Immunol. 2012;130(5):1153–1158.
Soyer OU et al. Mechanisms of peripheral tolerance to allergens. Allergy. 2013;68(2):161–70.
Platts-Mills TA, Woodfolk JA. Allergens and their role in the allergic immune response. Immunol Rev. 2011;242(1):51–68.
Meiler F et al. In vivo switch to IL-10-secreting T regulatory cells in high dose allergen exposure. J Exp Med. 2008;205(12):2887–98.
Akdis M et al. Immune responses in healthy and allergic individuals are characterized by a fine balance between allergen-specific T regulatory 1 and T helper 2 cells. J Exp Med. 2004;199(11):1567–75.
Platts-Mills T et al. Sensitisation, asthma, and a modified Th2 response in children exposed to cat allergen: a population-based cross-sectional study. Lancet. 2001;357(9258):752–6.
van de Veen W et al. IgG4 production is confined to human IL-10-producing regulatory B cells that suppress antigen-specific immune responses. J Allergy Clin Immunol. 2013;131(4):1204–12.
Kretschmer K et al. Inducing and expanding regulatory T cell populations by foreign antigen. Nat Immunol. 2005;6(12):1219–27.
Longo G et al. IgE-mediated food allergy in children. Lancet. 2013;382(9905):1656–64.
Karlsson MR, Rugtveit J, Brandtzaeg P. Allergen-responsive CD4+CD25+ regulatory T cells in children who have outgrown cow's milk allergy. J Exp Med. 2004;199(12):1679–88.
James JM, Sampson HA. Immunologic changes associated with the development of tolerance in children with cow milk allergy. J Pediatr. 1992;121(3):371–7.
Akdis M, Akdis CA. Mechanisms of allergen-specific immunotherapy: multiple suppressor factors at work in immune tolerance to allergens. J Allergy Clin Immunol. 2014;133(3):621–31.
Sicherer SH et al. The natural history of egg allergy in an observational cohort. J Allergy Clin Immunol. 2014;133(2):492–9.
Cox L, Cohn JR. Duration of allergen immunotherapy in respiratory allergy: when is enough, enough? Ann Allergy Asthma Immunol. 2007;98(5):416–26.
Burks AW et al. Oral immunotherapy for treatment of egg allergy in children. N Engl J Med. 2012;367(3):233–43. This is the first study to demonstrate sustained unresponsiveness in food allergic subjects who had completed OIT.
Buchanan AD et al. Egg oral immunotherapy in nonanaphylactic children with egg allergy. J Allergy Clin Immunol. 2007;119(1):199–205.
Vickery BP et al. Individualized IgE-based dosing of egg oral immunotherapy and the development of tolerance. Ann Allergy Asthma Immunol. 2010;105(6):444–50.
Keet CA et al. The safety and efficacy of sublingual and oral immunotherapy for milk allergy. J Allergy Clin Immunol. 2012;129(2):448–55.
Keet CA et al. Long-term follow-up of oral immunotherapy for cow's milk allergy. J Allergy Clin Immunol. 2013;132(3):737–739. This brief report describes clinical outcomes in patients up to 5 years following OIT for cow's milk allergy.
Staden U et al. Specific oral tolerance induction in food allergy in children: efficacy and clinical patterns of reaction. Allergy. 2007;62(11):1261–9.
Vickery BP et al. Sustained unresponsiveness to peanut in subjects who have completed peanut oral immunotherapy. J Allergy Clin Immunol. 2014;133(2):468–75. This is the first report of sustained unresponsiveness in peanut-allergic patients after OIT.
Jones SM et al. Clinical efficacy and immune regulation with peanut oral immunotherapy. J Allergy Clin Immunol. 2009;124(2):292–300.
Syed A et al. Peanut oral immunotherapy results in increased antigen-induced regulatory T-cell function and hypomethylation of forkhead box protein 3 (FOXP3). J Allergy Clin Immunol. 2014;133(2):500–10. This study evaluated clinical tolerance in peaut-allergic patients 6 months after completing OIT.
Chin SJ. et al. Sublingual versus oral immunotherapy for peanut-allergic children: A retrospective comparison. J Allergy Clin Immunol. 2013;132(2):476-8.
Naclerio RM et al. A double-blind study of the discontinuation of ragweed immunotherapy. J Allergy Clin Immunol. 1997;100(3):293–300.
Durham SR et al. Long-term clinical efficacy of grass-pollen immunotherapy. N Engl J Med. 1999;341(7):468–75.
Jacobsen L et al. Specific immunotherapy has long-term preventive effect of seasonal and perennial asthma: 10-year follow-up on the PAT study. Allergy. 2007;62(8):943–8.
Tabar AI et al. Three years of specific immunotherapy may be sufficient in house dust mite respiratory allergy. J Allergy Clin Immunol. 2011;127(1):57–63.
Durham SR et al. Long-term clinical efficacy in grass pollen-induced rhinoconjunctivitis after treatment with SQ-standardized grass allergy immunotherapy tablet. J Allergy Clin Immunol. 2010;125(1):131–8.
Durham SR et al. SQ-standardized sublingual grass immunotherapy: confirmation of disease modification 2 years after 3 years of treatment in a randomized trial. J Allergy Clin Immunol. 2012;129(3):717–725. This report describes that the clinical benefits of SLIT for pollen allergy can persist for at least 2 years after stopping therapy.
James LK et al. Long-term tolerance after allergen immunotherapy is accompanied by selective persistence of blocking antibodies. J Allergy Clin Immunol. 2011;127(2):509–516.
Didier A et al. Sustained 3-year efficacy of pre- and coseasonal 5-grass-pollen sublingual immunotherapy tablets in patients with grass pollen-induced rhinoconjunctivitis. J Allergy Clin Immunol. 2011;128(3):559–66.
Didier A et al. Post-treatment efficacy of discontinuous treatment with 300IR 5-grass pollen sublingual tablet in adults with grass pollen-induced allergic rhinoconjunctivitis. Clin Exp Allergy. 2013;43(5):568–77.
Bergmann KC et al. Efficacy and safety of sublingual tablets of house dust mite allergen extracts in adults with allergic rhinitis. J Allergy Clin Immunol. 2014;133(6):1608–14. This reference reports that SLIT for house dust mite allergy can result in clinical benefit that is sustained for at least 1 year following therapy.
Patel D et al. Fel d 1-derived peptide antigen desensitization shows a persistent treatment effect 1 year after the start of dosing: a randomized, placebo-controlled study. J Allergy Clin Immunol. 2013;131(1):103–9.
Senti G et al. Intralymphatic allergen administration renders specific immunotherapy faster and safer: a randomized controlled trial. Proc Natl Acad Sci U S A. 2008;105(46):17908–12.
Senti G et al. Intralymphatic immunotherapy for cat allergy induces tolerance after only 3 injections. J Allergy Clin Immunol. 2012;129(5):1290–6.
Witten M et al. Is intralymphatic immunotherapy ready for clinical use in patients with grass pollen allergy? J Allergy Clin Immunol. 2013;132(5):1248–1252.
Golden DB et al. Stinging insect hypersensitivity: a practice parameter update 2011. J Allergy Clin Immunol. 2011;127(4):852–4.
Golden DB et al. Discontinuing venom immunotherapy: outcome after five years. J Allergy Clin Immunol. 1996;97(2):579–87.
Golden DB, Kagey-Sobotka A, Lichtenstein LM. Survey of patients after discontinuing venom immunotherapy. J Allergy Clin Immunol. 2000;105(2 Pt 1):385–90.
Lerch E, Muller UR. Long-term protection after stopping venom immunotherapy: results of re-stings in 200 patients. J Allergy Clin Immunol. 1998;101(5):606–12.
Golden DB et al. Outcomes of allergy to insect stings in children, with and without venom immunotherapy. N Engl J Med. 2004;351(7):668–74.
Stritzke AI, Eng PA. Age-dependent sting recurrence and outcome in immunotherapy-treated children with anaphylaxis to Hymenoptera venom. Clin Exp Allergy. 2013;43(8):950–5.
Acknowledgments
We thank Arlene Mendoza-Moran for proof reading and grammatical review.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
A. Wesley Burks reports personal fees from ExploraMed Development, LLC, Food Allergy Initiative, Food Allergy Research & Education, GLG Research, Merck, Mylan Speciality, Novartis Pharma AG, Nutricia North America, Regeneron Pharmaceuticals, Inc., Unilever, ActoGeniX, SRA International, Genentech, Sanofi US Services;non-financial support from Mastcell Pharmaceuticals, Inc.; personal fees and grants from Hycor Biomedical and Allergen Research Corporation. Timothy P. Moran declares no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Immunotherapy and Immunomodulators
Rights and permissions
About this article
Cite this article
Moran, T.P., Burks, A.W. Is Clinical Tolerance Possible after Allergen Immunotherapy?. Curr Allergy Asthma Rep 15, 23 (2015). https://doi.org/10.1007/s11882-015-0523-3
Published:
DOI: https://doi.org/10.1007/s11882-015-0523-3