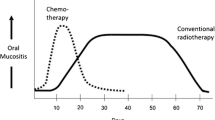
Opinion statement
Despite its history as one of the most impactful toxicities associated with cytotoxic cancer therapy, oral mucositis (OM) remains an unmet clinical need which affects hundreds of thousands of patients. Descriptions of its complex pathogenesis have provided mechanistic targets which are being exploited to develop an effective therapeutic intervention. Favorable results of recently completed clinical trials in which agents focused on interrupting the early stages of the mucositis biological cascade were assessed provide reason for optimism, not only for oral mucositis but also for halo indications which share its pathobiogenesis.
Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance
Lalla RV, et al. Oral mucositis due to high-dose chemotherapy and/or head and neck radiation therapy. J Natl Cancer Inst Monogr. 2019;2019(53):lgz011.
Panghal M, Kaushal V, Kadayan S, Yadav JP. Incidence and risk factors for infection in oral cancer patients undergoing different treatments protocols. BMC Oral Health. 2012. Jul 20;12:22. https://doi.org/10.1186/1472-6831-12-22.
Berger K, Schopohl D, Bollig A, Strobach D, Rieger C, Rublee D, et al. Burden of oral mucositis: a systematic review and implications for future research. Oncol Res Treat. 2018;41(6):399–405.
Sonis ST. Mucositis: the impact, biology and therapeutic opportunities of oral mucositis. Oral Oncol. 2009;45:1015–20.
• Bachour P, Sonis ST. Predicting the risk of mucositis associated with cytotoxic cancer treatment regimens: rationale, complexity and challenges. Curr Opinion Support Palliat Care. 2018;12:1998–210 Mucositis risk is not uniform. This paper discusses risk prediction, especially in the context of genomics.
Saunders DP, et al. Systematic review of antimicrobials, mucosal coating agents, anesthetics, and analgesics for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer. 2020. May;28(5):2473–84.
Chan A, Ignoffo RJ. Survey of topical oral solutions for the treatment of chemo-induced oral mucositis. J Oncol Pharm Pract. 2005;11:139–43.
Dodd MJ, Dibble SL, Miaskowski C, MacPhail L, Greenspan D, Paul SM, et al. Randomized clinical trial of the effectiveness of 3 commonly used mouthwashes to treat chemotherapy-induced mucositis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;90:39–47.
Treister N, Nieder M, Baggott C, Olson E, Chen L, Dang H, et al. Caphosol for prevention of oral mucositis in pediatric myeloablative haematopoietic cell transplantation. Br J Cancer. 2017. Jan 3;116(1):21–7.
Rao NG, Trotti A, Kim J, Schell MJ, Zhao X, Amdur RJ, et al. Phase II multicenter trial of Caphosol for the reduction of mucositis in patients receiving radiation therapy for head and neck cancer. Oral Oncol. 2014;50:765–9.
Park SH, Lee HS. Meta-analysis of oral cryotherapy in preventing oral mucositis associated with cancer therapy. Int J Nurs Pract. 2019. Oct;25(5):e12759.
Zecha JA, et al. Low-level laser therapy/photobiomodulation in the management of side effects of chemoradiation therapy in head and neck cancer: part 1: mechanisms of action, dosimetric, and safety considerations. Support Care Cancer. 2016;24:2781–92.
Pearlman R, Zaki M, Laszewski P, et al. Does low level laser therapy improve clinical outcomes related to oral mucositis in patients treated for head and neck cancer. Int J Radiat Oncol Biol Phys 2020; 108, S3: E812.
Sonis ST, Hashemi S, Epstein JB, Nair RG, Raber-Durlacher JE. Could the biological robustness of low-level laser therapy (photobiomodulation) impact its use in the management of mucositis in head and neck cancer patients? Oral Oncol. 2016;54:7–14.
Vasconcelos RM, Sanfilippo N, Paster BJ, Kerr AR, Li Y, Ramalho L, et al. Host-microbiome crosstalk in oral mucositis. J Dent Res. 2016;95:725–33.
Giles FJ, Miller CB, Hurd DD, Wingard JR, Fleming TR, Sonis ST, et al. A phase III, randomized, double-blind, placebo-controlled, multinational trial of Iseganan for the prevention of oral mucositis in patients receiving stomatotoxic chemotherapy (PROMPT-CT trial). Leuk Lymphoma. 2003;44:1165–72.
Trotti A, et al. A multinational, randomized phase III trial of Iseganan HCl oral solution for reducing the severity of oral mucositis in patients receiving radiotherapy for head and neck malignancy. Int J Radiat Oncol Phys. 2004;3:674–81.
Foote RL, Loprinzi CL, Frank AR, O’Fallon JR, Gulavita S, Tewfik HH, et al. Randomized trial of a chlorhexidine mouthwash for alleviation of radiation-induced mucositis. JCO. 1994;12:2630–3.
El-Sayed S, et al. Prophylaxis of radiation-associated mucositis in conventionally treated patients with head and neck cancer: a double-blind, phase III, randomized, controlled trial evaluating the clinical efficacy of an antimicrobial lozenge using a validated mucositis scoring system. J Clin Oncol. 2002;20:3956–63.
Stokman MA, Spijkervet FKL, Burlage FR, Dijkstra PU, Manson WL, de Vries EGE, et al. Oral mucositis and selective elimination of oral flora in head and neck cancer patients receiving radiotherapy: a double-blind randomized trial. Brit J Cancer. 2003;88:1012–6.
Spielberger R, Stiff P, Bensinger W, Gentile T, Weisdorf D, Kewalramani T, et al. Palifermin for oral mucositis after intensive therapy for hematologic cancers. N Engl J Med. 2004;351:2590–8.
Henke M, Alfonsi M, Foa P, Giralt J, Bardet E, Cerezo L, et al. Palifermin decreases severe oral mucositis of patients undergoing postoperative radiochemotherapy for head and neck cancer: a randomized, placebo-controlled trial. J Clin Oncol. 2011;29:2815–20.
Le QT, et al. Palifermin reduces severe mucositis in definitive chemoradiotherapy of locally advanced head and neck cancer: a randomized, placebo-controlled study. J Clin Oncol. 2011;29:2808–14.
Borges L, Rex KL, Chen JN, Wei P, Kaufman S, Scully S, et al. A protective role for keratinocyte growth factor in a murine model of chemotherapy and radiotherapy-induced mucositis. Int J Radiat Oncol Biol Phys. 2006;66:254–62.
Russi EG, Raber-Durlacher JE, Sonis ST. Local and systemic pathogenesis and consequences of regimen-induced inflammatory responses in patients with head and neck cancer receiving chemoradiation. Mediators Inflamm 2014; 518261. https://doi.org/10.1155/2014/518261.
Blijlevens N, Sonis S. Palifermin (recombinant keratinocyte growth factor-1): a pleiotropic growth factor with multiple biological activities in preventing chemotherapy- and radiotherapy-induced mucositis. Ann Oncol. 2007;18:817–26.
• Bowen J, et al. The pathogenesis of mucositis: updated perspectives and emerging targets. Support Care Cancer. 2019;27:4023–33 This recent review provides an update in the pathogenesis of mucositis and provides a perspective for the mechanistic basis for a number of agents under development.
Sonis ST. The pathobiology of mucositis. Nat Rev Cancer. 2004;4:277–84.
Oral mucositis market size, growth and trends by cause (oral mucositis caused by chemotherapy, oral mucositis caused by radiotherapy, oral mucositis caused by hematopoietic stem cell transplantation, and others), end-user (hospitals, dental clinics, oncology hospitals, and research institutes), and region (Americas, Europe, Asia-Pacific, and Middle East & Africa)—global forecast till 2025; ID: MRFR/Pharma/1002-HCR | September 2020.
Sonis ST. A comparison and assessment of scoring scales for mucositis. In Sonis, ST, Oral Mucositis. Springer Handbook Series 2012; pps 39–46.
Stokman MA, Sonis ST, Dijkstra PU, Burgerhof JGM, Spijkervet FKL. Assessment of oral mucositis in clinical trials: impact of training on evaluators in a multi-centre trial. Eur J Cancer. 2005;41:1735–8.
Quinn B, Stone R, Uhlenhopp M, et al. Ensuring accurate oral mucositis assessment in the European Group for Blood and Marrow Transplantation Prospective Oral Mucositis Audit. Eur J Oncol Nurs. 2007;11 Suppl 1:S10–8.
Colevas AD, Yom SS, Pfister DG, Spencer S, Adelstein D, Adkins D, et al. NCCN guidelines insights: head and neck cancers, version 1.2018. J Natl Compr Cancer Netw. 2018;16:479–90.
Vawda N, Banerjee RN, Debenham BJ. Impact of smoking on outcomes of HPV-related oropharyngeal cancer treated with primary radiation or surgery. Int J Radiat Oncol Biol Phys. 2019;103:1125–31.
Noronha V, Joshi A, Patil VM, Agarwal J, Ghosh-Laskar S, Budrukkar A, et al. Once-a-week versus once-every-3-weeks cisplatin chemoradiation for locally advanced head and neck cancer: a phase III randomized noninferiority trial. J Clin Oncol. 2018;36:1064–72.
Szturz P, Wouters K, Kiyota N, Tahara M, Prabhash K, Noronha V, et al. Weekly low-dose versus three-weekly high-dose cisplatin for concurrent chemoradiation in locoregionally advanced non-nasopharyngeal head and neck cancer: a systematic review and meta-analysis of aggregate data. Oncologist. 2017;22:1056–66.
• Anderson CM, et al. Phase 2b, randomized, double-blind trial of GC4419 vs placebo to reduce severe oral mucositis in head and neck cancer patients receiving concurrent radiotherapy and cisplatin. J Clin Oncol. 2019;37:3256–65 Results of a phase 2 trial testing the efficacy of a superoxide dismutase mimetic. The clinical trial design is representative of most of studies being done for an OM indication. The efficacy data support the importance of superoxide formation as a driver of OM and validate superoxide dismutases’ potential in mitigating its pathogenesis.
Wang Y, Branicky R, Noë A, Hekimi S. Superoxide dismutases: dual roles in controlling ROS damage and regulating ROS signaling. J Cell Biol. 2018;217:1915–28.
Escribano A, García-Grande A, Montañés P, Miralles L, García A. Aerosol orgotein (Ontosein) for the prevention of radiotherapy-induced adverse effects in head and neck cancer patients: a feasibility study. Neoplasma. 2002;49:201–8.
Murphy CK, Fey EG, Watkins BA, Wong V, Rothstein D, Sonis ST. Efficacy of superoxide dismutase mimetic M40403 in attenuating radiation-induced oral mucositis in hamsters. Clin Cancer Res. 2008;14:4292–7.
Nguyen T, Nioi P, Pickett CB. The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. JBC. 2009;284:13291–5.
Sher DJ, Bonomi M, Blakaj DM, et al. Results of a randomized, open label, multicenter trial assessing the safety, dose and schedule of RRx-001 (R001) in reducing severe oral mucositis (SOM) in patients receiving chemoradiation (CRT) for oral cavity/oropharynx squamous cell carcinoma (OSCC). Int J Radiat Oncol Biol Phys 2020; 108, S3:S107–108.
Osei-Fofie D, et al. Phase1b, international, dose-escalation study to evaluate the safety, pharmacokinetics (PK) and efficacy of ST-617, a dithiolethione, to attenuate oral mucositis (OM) in patients receiving chemoradiation for head and neck cancers. ESMO 2020; Meeting Abstract 3692.
• Kudrimoti M, et al. Dusquetide: reduction in oral mucositis associated with enduring ancillary benefits in tumor resolution and decreased mortality in head and neck cancer patients. Biotechnol Rep (Amst). 2017;15:24–6 The pre-clinical and clinical results described support the importance of the innate immune response in the initiation of mucositis. The difference in response based on the concomitant chemoradiation regimen is noteworthy.
Choi S, Shin SH, Lee HR, Sohn KY, Yoon SY, Kim JW. 1-Palmitoyl-2-linoleoyl-3-acetyl-rac-glycerol ameliorates chemoradiation-induced oral mucositis. Oral Dis. 2020;26:111–21.
Innovation Pharmaceuticals Press Release. Innovation Pharmaceuticals Phase 2 Oral Mucositis Trial Additional Data Show Brilacidin-OM Demonstrated A Significant Reduction in the Incidence of Severe Oral Mucositis in Patients with Head and Neck Cancer (HNC) Receiving Aggressive Chemotherapy Regimen. April 9, 2018.
Scott, R.W., Sonis, S., Korczak, B., Brilacidin, Host Defense Peptide Mimetic, One of a New Class of Immunomodulatory Agents That Can Target Multiple Disease Indications et al. 25th European Congress of Clinical Microbiology and Infectious Diseases (ECCMID 2015).
• Giralt J, et al. Randomized phase 2 trial of a novel clonidine mucoadhesive buccal tablet for the amelioration of oral mucositis in patients treated with concomitant chemoradiation therapy for head and neck cancer. Int J Radiat Oncol Biol Phys. 2020;106:320–6 This paper describes the results of a clinical trial in which a topically delivered formulation impacted the course and severity of mucositis development.
Grbic J, Wexler I, Celenti R, Altman J, Saffer A. A phase II trial of transmucosal herbal patch for the treatment of gingivitis. J Am Dent Assoc. 2011;142:1168–75.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Stephen T. Sonis is an employee of Biomodels LLC and Primary Endpoint Solutions, LLC; both companies assist industry (including companies described in this paper), government, and academics to study and enable drugs, biologicals, and devices to treat patients for a wide range of indications including oral mucositis. Dr. Sonis does not have equity in any of the companies with which he works, nor does he receive direct compensation from them. Dr. Sonis is also a founder of Inform Genomics, Inc.; and is listed as an inventor on the following issued patents: 6458777, 6663850, 6713463, 6841578B2, 7297123, and 10,475539.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Palliative and Supportive Care
Rights and permissions
About this article
Cite this article
Sonis, S.T. Treatment for Oral Mucositis—Current Options and an Update of Small Molecules Under Development. Curr. Treat. Options in Oncol. 22, 25 (2021). https://doi.org/10.1007/s11864-021-00823-6
Accepted:
Published:
DOI: https://doi.org/10.1007/s11864-021-00823-6