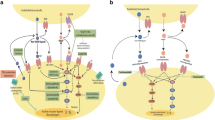
Opinion statement
With a growing understanding of the biologic drivers of different thyroid cancers, there is an ongoing revolution in the treatment of aggressive and advanced disease variants. This includes matching patients with specific point mutations or gene fusions to targeted therapies (e.g., selective RET inhibitors), delineating patients who are likely to respond to immune checkpoint inhibition (i.e., PD-L1-positive tumors) and even priming responses to traditional therapies such as radioactive iodine (via concomitant MAPK pathway inhibition). There is also a growing role for genomics in the prognostication of thyroid tumors to aid the adjudication of appropriate treatments. Taking stock of the current state of the field, recent successes should be celebrated, but there still remains a long road ahead to improve outcomes for patients, particularly for radioactive-iodine refractory differentiated thyroid cancer and anaplastic thyroid cancer. In this review, we summarize findings from recent clinical trials and highlight promising preclinical data supporting molecular-driven therapy in advanced thyroid cancer. Ultimately, enrollment in clinical trials remains paramount to the advancement of thyroid cancer care.


Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
TCGA. Integrated genomic characterization of papillary thyroid carcinoma. Cell. 2014;159(3):676–90. https://doi.org/10.1016/j.cell.2014.09.050.
Xing M. Molecular pathogenesis and mechanisms of thyroid cancer. Nat Rev Cancer. 2013;13(3):184–99. https://doi.org/10.1038/nrc3431.
Ganly I, Makarov V, Deraje S, Dong Y, Reznik E, Seshan V, et al. Integrated genomic analysis of Hürthle cell cancer reveals oncogenic drivers, recurrent mitochondrial mutations, and unique chromosomal landscapes. Cancer Cell. 2018;34(2):256–70.e5. https://doi.org/10.1016/j.ccell.2018.07.002.
Gopal RK, Kübler K, Calvo SE, Polak P, Livitz D, Rosebrock D, et al. Widespread chromosomal losses and mitochondrial DNA alterations as genetic drivers in Hürthle cell carcinoma. Cancer Cell. 2018;34(2):242–55.e5. https://doi.org/10.1016/j.ccell.2018.06.013.
Pozdeyev N, Gay LM, Sokol ES, Hartmaier R, Deaver KE, Davis S, et al. Genetic analysis of 779 advanced differentiated and anaplastic thyroid cancers. Clin Cancer Res. 2018;24(13):3059–68. https://doi.org/10.1158/1078-0432.Ccr-18-0373.
Landa I, Ibrahimpasic T, Boucai L, Sinha R, Knauf JA, Shah RH, et al. Genomic and transcriptomic hallmarks of poorly differentiated and anaplastic thyroid cancers. J Clin Invest. 2016;126(3):1052–66. https://doi.org/10.1172/jci85271.
Klubo-Gwiezdzinska J, Wartofsky L. The role of molecular diagnostics in the management of indeterminate thyroid nodules. J Clin Endocrinol Metab. 2018;103(9):3507–10. https://doi.org/10.1210/jc.2018-01081.
Xing M, Alzahrani AS, Carson KA, Shong YK, Kim TY, Viola D, et al. Association between BRAF V600E mutation and recurrence of papillary thyroid cancer. J Clin Oncol. 2015;33(1):42–50. https://doi.org/10.1200/jco.2014.56.8253.
Bournaud C, Descotes F, Decaussin-Petrucci M, Berthiller J, de la Fouchardière C, Giraudet AL, et al. TERT promoter mutations identify a high-risk group in metastasis-free advanced thyroid carcinoma. Eur J Cancer. 2019;108:41–9. https://doi.org/10.1016/j.ejca.2018.12.003.
Liu X, Qu S, Liu R, Sheng C, Shi X, Zhu G, et al. TERT promoter mutations and their association with BRAF V600E mutation and aggressive clinicopathological characteristics of thyroid cancer. J Clin Endocrinol Metab. 2014;99(6):E1130–6. https://doi.org/10.1210/jc.2013-4048.
Landa I, Ganly I, Chan TA, Mitsutake N, Matsuse M, Ibrahimpasic T, et al. Frequent somatic TERT promoter mutations in thyroid cancer: higher prevalence in advanced forms of the disease. J Clin Endocrinol Metab. 2013;98(9):E1562–6. https://doi.org/10.1210/jc.2013-2383.
Liu R, Bishop J, Zhu G, Zhang T, Ladenson PW, Xing M. Mortality risk stratification by combining BRAF V600E and TERT promoter mutations in papillary thyroid cancer: genetic duet of BRAF and TERT promoter mutations in thyroid cancer mortality. JAMA. Oncol. 2017;3(2):202–8. https://doi.org/10.1001/jamaoncol.2016.3288.
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26(1):1–133. https://doi.org/10.1089/thy.2015.0020.
Xu JY, Handy B, Michaelis CL, Waguespack SG, Hu MI, Busaidy N, et al. Detection and prognostic significance of circulating tumor cells in patients with metastatic thyroid cancer. J Clin Endocrinol Metab. 2016;101(11):4461–7. https://doi.org/10.1210/jc.2016-2567.
Qiu ZL, Wei WJ, Sun ZK, Shen CT, Song HJ, Zhang XY, et al. Circulating tumor cells correlate with clinicopathological features and outcomes in differentiated thyroid cancer. Cell Physiol Biochem. 2018;48(2):718–30. https://doi.org/10.1159/000491898.
Iyer PC, Cote GJ, Hai T, Gule-Monroe M, Bui-Griffith J, Williams MD, et al. Circulating BRAF V600E cell-free DNA as a biomarker in the management of anaplastic thyroid carcinoma. JCO Precis Oncol. 2018;2:1–11. https://doi.org/10.1200/po.18.00173.
Peng X, Zhang K, Ma L, Xu J, Chang W. The role of long non-coding RNAs in thyroid cancer. Front Oncol. 2020;10:941. https://doi.org/10.3389/fonc.2020.00941.
Lubitz CC, Zhan T, Gunda V, Amin S, Gigliotti BJ, Fingeret AL, et al. Circulating BRAF(V600E) levels correlate with treatment in patients with thyroid carcinoma. Thyroid. 2018;28(3):328–39. https://doi.org/10.1089/thy.2017.0322.
Durante C, Haddy N, Baudin E, Leboulleux S, Hartl D, Travagli JP, et al. Long-term outcome of 444 patients with distant metastases from papillary and follicular thyroid carcinoma: benefits and limits of radioiodine therapy. J Clin Endocrinol Metab. 2006;91(8):2892–9. https://doi.org/10.1210/jc.2005-2838.
Schlumberger M, Brose M, Elisei R, Leboulleux S, Luster M, Pitoia F, et al. Definition and management of radioactive iodine-refractory differentiated thyroid cancer. Lancet Diabetes Endocrinol. 2014;2(5):356–8. https://doi.org/10.1016/s2213-8587(13)70215-8.
Durante C, Puxeddu E, Ferretti E, Morisi R, Moretti S, Bruno R, et al. BRAF mutations in papillary thyroid carcinomas inhibit genes involved in iodine metabolism. J Clin Endocrinol Metab. 2007;92(7):2840–3. https://doi.org/10.1210/jc.2006-2707.
Chakravarty D, Santos E, Ryder M, Knauf JA, Liao XH, West BL, et al. Small-molecule MAPK inhibitors restore radioiodine incorporation in mouse thyroid cancers with conditional BRAF activation. J Clin Invest. 2011;121(12):4700–11. https://doi.org/10.1172/jci46382.
Liu D, Hu S, Hou P, Jiang D, Condouris S, Xing M. Suppression of BRAF/MEK/MAP kinase pathway restores expression of iodide-metabolizing genes in thyroid cells expressing the V600E BRAF mutant. Clin Cancer Res. 2007;13(4):1341–9. https://doi.org/10.1158/1078-0432.Ccr-06-1753.
Ho AL, Grewal RK, Leboeuf R, Sherman EJ, Pfister DG, Deandreis D, et al. Selumetinib-enhanced radioiodine uptake in advanced thyroid cancer. N Engl J Med. 2013;368(7):623–32. https://doi.org/10.1056/NEJMoa1209288.
Rothenberg SM, McFadden DG, Palmer EL, Daniels GH, Wirth LJ. Redifferentiation of iodine- refractory BRAF V600E-mutant metastatic papillary thyroid cancer with dabrafenib. Clin Cancer Res. 2015;21(5):1028–35. https://doi.org/10.1158/1078-0432.Ccr-14-2915.
Jaber T, Waguespack SG, Cabanillas ME, Elbanan M, Vu T, Dadu R, et al. Targeted therapy in advanced thyroid cancer to resensitize tumors to radioactive iodine. J Clin Endocrinol Metab. 2018;103(10):3698–705. https://doi.org/10.1210/jc.2018-00612.
Dunn LA, Sherman EJ, Baxi SS, Tchekmedyian V, Grewal RK, Larson SM, et al. Vemurafenib redifferentiation of BRAF mutant, RAI-refractory thyroid cancers. J Clin Endocrinol Metab. 2019;104(5):1417–28. https://doi.org/10.1210/jc.2018-01478.
Nagarajah J, Le M, Knauf JA, Ferrandino G, Montero-Conde C, Pillarsetty N, et al. Sustained ERK inhibition maximizes responses of BrafV600E thyroid cancers to radioiodine. J Clin Invest. 2016;126(11):4119–24. https://doi.org/10.1172/jci89067.
Ruegemer JJ, Hay ID, Bergstralh EJ, Ryan JJ, Offord KP, Gorman CA. Distant metastases in differentiated thyroid carcinoma: a multivariate analysis of prognostic variables. J Clin Endocrinol Metab. 1988;67(3):501–8. https://doi.org/10.1210/jcem-67-3-501.
Brose MS, Nutting CM, Jarzab B, Elisei R, Siena S, Bastholt L, et al. Sorafenib in radioactive iodine-refractory, locally advanced or metastatic differentiated thyroid cancer: a randomised, double-blind, phase 3 trial. Lancet. 2014;384(9940):319–28. https://doi.org/10.1016/s0140-6736(14)60421-9.
Schlumberger M, Tahara M, Wirth LJ, Robinson B, Brose MS, Elisei R, et al. Lenvatinib versus placebo in radioiodine-refractory thyroid cancer. N Engl J Med. 2015;372(7):621–30. https://doi.org/10.1056/NEJMoa1406470.
Cabanillas ME, de Souza JA, Geyer S, Wirth LJ, Menefee ME, Liu SV, et al. Cabozantinib as salvage therapy for patients with tyrosine kinase inhibitor-refractory differentiated thyroid cancer: results of a multicenter phase II international thyroid oncology group trial. J Clin Oncol. 2017;35(29):3315–21. https://doi.org/10.1200/jco.2017.73.0226.
Bible KC, Menefee ME, Lin CJ, Millward MJ, Maples WJ, Goh BC, et al. An international phase 2 study of pazopanib in progressive and metastatic thyroglobulin antibody negative radioactive iodine refractory differentiated thyroid cancer. Thyroid. 2020;30(9):1254–62. https://doi.org/10.1089/thy.2019.0269.
Gianoukakis AG, Dutcus CE, Batty N, Guo M, Baig M. Prolonged duration of response in lenvatinib responders with thyroid cancer. Endocr Relat Cancer. 2018;25(6):699–704. https://doi.org/10.1530/erc-18-0049.
Wirth LJ, Sherman E, Robinson B, Solomon B, Kang H, Lorch J, et al. Efficacy of selpercatinib in RET-altered thyroid cancers. N Engl J Med. 2020;383(9):825–35. https://doi.org/10.1056/NEJMoa2005651. In a phase I/II trial, patients with RET-mutant medullary thyroid cancer and RET fusion–positive differentiated thyroid cancer had marked, durable responses and limited adverse events after treatment with with the selective RET inhibitor selpercatinib.
MI-NH VS, Gainor JF, Mansfield AS, Alonso G, Taylor MH, Zhu VW, et al. Turner, Giuseppe Curigliano. Clinical activity of the RET inhibitor pralsetinib (BLU-667) in patients with RET fusion+ solid tumors. J Clin Oncol. 2020;38(suppl):abstr 109.
Drilon A, Laetsch TW, Kummar S, DuBois SG, Lassen UN, Demetri GD, et al. Efficacy of larotrectinib in TRK fusion-positive cancers in adults and children. N Engl J Med. 2018;378(8):731–9. https://doi.org/10.1056/NEJMoa1714448.
Doebele RC, Drilon A, Paz-Ares L, Siena S, Shaw AT, Farago AF, et al. Entrectinib in patients with advanced or metastatic NTRK fusion-positive solid tumours: integrated analysis of three phase 1–2 trials. Lancet Oncol. 2020;21(2):271–82. https://doi.org/10.1016/s1470-2045(19)30691-6.
Cabanillas ME, Drilon A, Farago AF, Brose MS, McDermott R, Sohal D, Oh DY, Almubarak M, Bauman J, Chu E, Kummar S. 1916P Larotrectinib treatment of advanced TRK fusion thyroid cancer. Ann Oncol 2020;31:S1086.
Chintakuntlawar AV, Foote RL, Kasperbauer JL, Bible KC. Diagnosis and management of anaplastic thyroid cancer. Endocrinol Metab Clin N Am. 2019;48(1):269–84. https://doi.org/10.1016/j.ecl.2018.10.010.
Xu B, Fuchs T, Dogan S, Landa I, Katabi N, Fagin JA, et al. Dissecting anaplastic thyroid carcinoma: a comprehensive clinical, histologic, Immunophenotypic, and molecular study of 360 cases. Thyroid. 2020;30:1505–17. https://doi.org/10.1089/thy.2020.0086.
Charles RP, Silva J, Iezza G, Phillips WA, McMahon M. Activating BRAF and PIK3CA mutations cooperate to promote anaplastic thyroid carcinogenesis. Mol Cancer Res. 2014;12(7):979–86. https://doi.org/10.1158/1541-7786.Mcr-14-0158-t.
McFadden DG, Vernon A, Santiago PM, Martinez-McFaline R, Bhutkar A, Crowley DM, et al. p53 constrains progression to anaplastic thyroid carcinoma in a Braf-mutant mouse model of papillary thyroid cancer. Proc Natl Acad Sci U S A. 2014;111(16):E1600–9. https://doi.org/10.1073/pnas.1404357111.
•• Subbiah V, Kreitman RJ, Wainberg ZA, Cho JY, Schellens JHM, Soria JC, et al. Dabrafenib and trametinib treatment in patients with locally advanced or metastatic BRAF V600-mutant anaplastic thyroid cancer. J Clin Oncol. 2018;36(1):7–13. https://doi.org/10.1200/jco.2017.73.6785. Phase II trial demonstrating remarkable response rate and overall survival (80% at 1-year) for BRAF plus MEK inhibition in patients with anaplastic thyroid cancer harboring BRAF V600E mutations.
Platini F, Ortolan E, Cavalieri S, et al. BRAF V600E-mutated anaplastic thyroid carcinoma (ATC) and treatment with BRAF-inhibitors: Real-world data from a single-institution, still far from the cure. J Clin Oncol. 2020;38(15_suppl): e18577–e18577.
Wang JR, Zafereo ME, Dadu R, Ferrarotto R, Busaidy NL, Lu C, et al. Complete surgical resection following neoadjuvant dabrafenib plus trametinib in BRAF(V600E)-mutated anaplastic thyroid carcinoma. Thyroid. 2019;29(8):1036–43. https://doi.org/10.1089/thy.2019.0133.
Dias-Santagata D, Lennerz JK, Sadow PM, Frazier RP, Govinda Raju S, Henry D, et al. Response to RET-specific therapy in RET fusion-positive anaplastic thyroid carcinoma. Thyroid. 2020;30(9):1384–9. https://doi.org/10.1089/thy.2019.0477.
Godbert Y, Henriques de Figueiredo B, Bonichon F, Chibon F, Hostein I, Pérot G, et al. Remarkable response to crizotinib in woman with anaplastic lymphoma kinase-rearranged anaplastic thyroid carcinoma. J Clin Oncol. 2015;33(20):e84–7. https://doi.org/10.1200/jco.2013.49.6596.
Leroy L, Bonhomme B, Le Moulec S, Soubeyran I, Italiano A, Godbert Y. Remarkable response to ceritinib and brigatinib in an anaplastic lymphoma kinase-rearranged anaplastic thyroid carcinoma previously treated with crizotinib. Thyroid. 2020;30(2):343–4. https://doi.org/10.1089/thy.2019.0202.
Takahashi S, Kiyota N, Yamazaki T, Chayahara N, Nakano K, Inagaki L, et al. A phase II study of the safety and efficacy of lenvatinib in patients with advanced thyroid cancer. Future Oncol. 2019;15(7):717–26. https://doi.org/10.2217/fon-2018-0557.
Wirth LJ, Brose MS, Sherman EJ, Misir S, Xie S, Almonte A, Ye W, Licitra L, Schlumberger M, Sherman SI, Cabanillas M. MON-521 an open-label, single-arm, multicenter, phase 2 trial of lenvatinib (LEN) for the treatment of anaplastic thyroid cancer (ATC). J Endocr Soc. 2020;4(Supplement_1):MON-521.
Bible KC, Suman VJ, Menefee ME, Smallridge RC, Molina JR, Maples WJ, et al. A multiinstitutional phase 2 trial of pazopanib monotherapy in advanced anaplastic thyroid cancer. J Clin Endocrinol Metab. 2012;97(9):3179–84. https://doi.org/10.1210/jc.2012-1520.
Ravaud A, de la Fouchardière C, Caron P, Doussau A, Do Cao C, Asselineau J, et al. A multicenter phase II study of sunitinib in patients with locally advanced or metastatic differentiated, anaplastic or medullary thyroid carcinomas: mature data from the THYSU study. Eur J Cancer. 2017;76:110–7. https://doi.org/10.1016/j.ejca.2017.01.029.
Hanna GJ, Busaidy NL, Chau NG, Wirth LJ, Barletta JA, Calles A, et al. Genomic correlates of response to everolimus in aggressive radioiodine-refractory thyroid cancer: a phase II study. Clin Cancer Res. 2018;24(7):1546–53. https://doi.org/10.1158/1078-0432.Ccr-17-2297.
Yoo SK, Song YS, Lee EK, Hwang J, Kim HH, Jung G, et al. Integrative analysis of genomic and transcriptomic characteristics associated with progression of aggressive thyroid cancer. Nat Commun. 2019;10(1):2764. https://doi.org/10.1038/s41467-019-10680-5.
Knauf JA, Luckett KA, Chen KY, Voza F, Socci ND, Ghossein R, et al. Hgf/Met activation mediates resistance to BRAF inhibition in murine anaplastic thyroid cancers. J Clin Invest. 2018;128(9):4086–97. https://doi.org/10.1172/jci120966.
Krishnamoorthy GP, Davidson NR, Leach SD, Zhao Z, Lowe SW, Lee G, et al. EIF1AX and RAS mutations cooperate to drive thyroid tumorigenesis through ATF4 and c-MYC. Cancer Discov. 2019;9(2):264–81. https://doi.org/10.1158/2159-8290.Cd-18-0606.
Delivanis DA, Gustafson MP, Bornschlegl S, Merten MM, Kottschade L, Withers S, et al. Pembrolizumab-induced thyroiditis: comprehensive clinical review and insights into underlying involved mechanisms. J Clin Endocrinol Metab. 2017;102(8):2770–80. https://doi.org/10.1210/jc.2017-00448.
Kotwal A, Kottschade L, Ryder M. PD-L1 inhibitor-induced thyroiditis is associated with better overall survival in cancer patients. Thyroid. 2020;30(2):177–84. https://doi.org/10.1089/thy.2019.0250.
Bastman JJ, Serracino HS, Zhu Y, Koenig MR, Mateescu V, Sams SB, et al. Tumor-infiltrating T cells and the PD-1 checkpoint pathway in advanced differentiated and anaplastic thyroid cancer. J Clin Endocrinol Metab. 2016;101(7):2863–73. https://doi.org/10.1210/jc.2015-4227.
Chintakuntlawar AV, Rumilla KM, Smith CY, Jenkins SM, Foote RL, Kasperbauer JL, et al. Expression of PD-1 and PD-L1 in anaplastic thyroid cancer patients treated with multimodal therapy: results from a retrospective study. J Clin Endocrinol Metab. 2017;102(6):1943–50. https://doi.org/10.1210/jc.2016-3756.
• Capdevila J, Wirth LJ, Ernst T, Ponce Aix S, Lin CC, Ramlau R, et al. PD-1 blockade in anaplastic thyroid carcinoma. J Clin Oncol. 2020;38(23):2620–7. https://doi.org/10.1200/jco.19.02727 Phase I/II trial demonstrating durable responses in patients with anaplastic thyroid cancer treated with immune-checkpoint (PD-1) inhibition.
Gunda V, Gigliotti B, Ndishabandi D, Ashry T, McCarthy M, Zhou Z, et al. Combinations of BRAF inhibitor and anti-PD-1/PD-L1 antibody improve survival and tumour immunity in an immunocompetent model of orthotopic murine anaplastic thyroid cancer. Br J Cancer. 2018;119(10):1223–32. https://doi.org/10.1038/s41416-018-0296-2.
Iyer PC, Dadu R, Gule-Monroe M, Busaidy NL, Ferrarotto R, Habra MA, et al. Salvage pembrolizumab added to kinase inhibitor therapy for the treatment of anaplastic thyroid carcinoma. J Immunother Cancer. 2018;6(1):68. https://doi.org/10.1186/s40425-018-0378-y.
Cabanillas ME, Dadu R, Ferrarotto R, Liu S, Fellman BM, Gross ND, et al. Atezolizumab combinations with targeted therapy for anaplastic thyroid carcinoma (ATC). J Clin Oncol. 2020;38(15_suppl):6514. https://doi.org/10.1200/JCO.2020.38.15_suppl.6514.
Chintakuntlawar AV, Yin J, Foote RL, Kasperbauer JL, Rivera M, Asmus E, et al. A phase 2 study of pembrolizumab combined with chemoradiotherapy as initial treatment for anaplastic thyroid cancer. Thyroid. 2019;29(11):1615–22. https://doi.org/10.1089/thy.2019.0086.
Taylor MH, Lee CH, Makker V, Rasco D, Dutcus CE, Wu J, et al. Phase IB/II trial of lenvatinib plus pembrolizumab in patients with advanced renal cell carcinoma, endometrial cancer, and other selected advanced solid tumors. J Clin Oncol. 2020;38(11):1154–63. https://doi.org/10.1200/jco.19.01598.
Kato Y, Tabata K, Kimura T, Yachie-Kinoshita A, Ozawa Y, Yamada K, et al. Lenvatinib plus anti-PD-1 antibody combination treatment activates CD8+ T cells through reduction of tumor- associated macrophage and activation of the interferon pathway. PLoS One. 2019;14(2):e0212513. https://doi.org/10.1371/journal.pone.0212513.
Gunda V, Gigliotti B, Ashry T, Ndishabandi D, McCarthy M, Zhou Z, et al. Anti-PD-1/PD-L1 therapy augments lenvatinib's efficacy by favorably altering the immune microenvironment of murine anaplastic thyroid cancer. Int J Cancer. 2019;144(9):2266–78. https://doi.org/10.1002/ijc.32041.
Chowdhury S, Veyhl J, Jessa F, Polyakova O, Alenzi A, MacMillan C, et al. Programmed death-ligand 1 overexpression is a prognostic marker for aggressive papillary thyroid cancer and its variants. Oncotarget. 2016;7(22):32318–28. https://doi.org/10.18632/oncotarget.8698.
Mehnert JM, Varga A, Brose MS, Aggarwal RR, Lin CC, Prawira A, et al. Safety and antitumor activity of the anti-PD-1 antibody pembrolizumab in patients with advanced, PD-L1-positive papillary or follicular thyroid cancer. BMC Cancer. 2019;19(1):196. https://doi.org/10.1186/s12885-019-5380-3.
Haugen B, French J, Worden FP, Konda B, Sherman EJ, Dadu R, et al. Lenvatinib plus pembrolizumab combination therapy in patients with radioiodine-refractory (RAIR), progressive differentiated thyroid cancer (DTC): results of a multicenter phase II international thyroid oncology group trial. J Clin Oncol. 2020;38(15_suppl):6512. https://doi.org/10.1200/JCO.2020.38.15_suppl.6512.
Haugen B, French J, Worden F, et al. Lenvatinib plus pembrolizumab combination therapy in patients with radioiodine-refractory (RAIR), progressive differentiated thyroid cancer (DTC): Results of a multicenter phase II international thyroid oncology group trial. J Clin Oncol 2020;38(15_suppl):6512.
Lorch JH, Barletta JA, Nehs M, Uppaluri R, Alexander EK, Haddad RI, et al. A phase II study of nivolumab (N) plus ipilimumab (I) in radioidine refractory differentiated thyroid cancer (RAIR DTC) with exploratory cohorts in anaplastic (ATC) and medullary thyroid cancer (MTC). J Clin Oncol. 2020;38(15_suppl):6513. https://doi.org/10.1200/JCO.2020.38.15_suppl.6513.
Ciampi R, Romei C, Ramone T, Prete A, Tacito A, Cappagli V, et al. Genetic landscape of somatic mutations in a large cohort of sporadic medullary thyroid carcinomas studied by next-generation targeted sequencing. iScience. 2019;20:324–36. https://doi.org/10.1016/j.isci.2019.09.030.
Moley JF. Medullary thyroid carcinoma: management of lymph node metastases. J Natl Compr Cancer Netw. 2010;8(5):549–56. https://doi.org/10.6004/jnccn.2010.0042.
Wells SA Jr, Robinson BG, Gagel RF, Dralle H, Fagin JA, Santoro M, et al. Vandetanib in patients with locally advanced or metastatic medullary thyroid cancer: a randomized, double-blind phase III trial. J Clin Oncol. 2012;30(2):134–41. https://doi.org/10.1200/jco.2011.35.5040.
Elisei R, Schlumberger MJ, Müller SP, Schöffski P, Brose MS, Shah MH, et al. Cabozantinib in progressive medullary thyroid cancer. J Clin Oncol. 2013;31(29):3639–46. https://doi.org/10.1200/jco.2012.48.4659.
Schlumberger M, Elisei R, Muller S, et al. Final overall survival analysis of EXAM, an international, double-blind, randomized, placebo-controlled phase III trial of cabozantinib (Cabo) in medullary thyroid carcinoma (MTC) patients with documented RECIST progression at baseline. Proc Am Soc Clin Oncol. 2015;33(suppl):6012.
Hu M, Subbiah V, Wirth LJ, Schuler M, Mansfield AS, Brose MS, Curigliano G, Leboulleux S, Zhu VW, Keam B, Matos I. 1913O Results from the registrational phase I/II ARROW trial of pralsetinib (BLU-667) in patients (pts) with advanced RET mutation-positive medullary thyroid cancer (RET+ MTC). Ann Oncol. 2020;31:S1084.
Bruce JY, Bible KC, Chintakuntlawar AV. Emergence of resistant clones in medullary thyroid cancer may not be rescued by subsequent salvage highly selective rearranged during transfection-inhibitor therapy. Thyroid. 2020. https://doi.org/10.1089/thy.2020.0449.
Maniakas A, Dadu R, Busaidy NL, Wang JR, Ferrarotto R, Lu C, et al. Evaluation of overall survival in patients with anaplastic thyroid carcinoma, 2000–2019. JAMA Oncol. 2020;6(9):1–8. https://doi.org/10.1001/jamaoncol.2020.3362.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Kevin C. Miller declares that he has no conflict of interest. Ashish V. Chintakuntlawar has received clinical trial funding (paid to his institution) from Merck, AstraZeneca/MedImmune, Kura Oncology, Inovio Pharmaceuticals, and Eisai, and has served on advisory boards for Kura Oncology and HRA Pharma (both institutional).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Head and Neck Cancer
Rights and permissions
About this article
Cite this article
Miller, K.C., Chintakuntlawar, A.V. Molecular-Driven Therapy in Advanced Thyroid Cancer. Curr. Treat. Options in Oncol. 22, 24 (2021). https://doi.org/10.1007/s11864-021-00822-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s11864-021-00822-7