Abstract
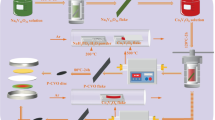
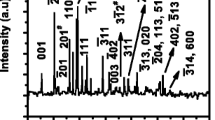
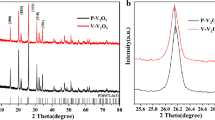
To meet the ever-increasing demand for higher energy density and power density, developing cathode materials with high specific capacity has become an urgent problem. V2O5 has been considered as one of the most promising cathodes for its high theoretical specific capacity (443 mAh g−1), abundant source, and low cost. In this work, cobalt-doped vanadium pentoxide (Co-V2O5) microflowers were synthesized through a simple solvothermal method. The doped Co not only improves the electronic conductivity but also increases the diffusion coefficient of the lithium ions. Benefiting from the appropriate amount of Co doping, the charge transfer impedance decreases from 51 to 28 Ω and the diffusion coefficient of the lithium ions increased from 2.81×10−17 cm2 s−1 to 3.31×10−16 cm2 s−1. The optimal Co-V2O5 microflowers deliver a high reversible capacity of 166 mAh g−1 after 200 cycles.






Similar content being viewed by others
References
A. Tomaszewska, Z. Chu, X. Feng, S. O’Kane, X. Liu, J. Chen, C. Ji, E. Endler, R. Li, L. Liu, Y. Li, S. Zheng, S. Vetterlein, M. Gao, J. Du, M. Parkes, M. Ouyang, M. Marinescu, G. Offer, and B. Wu, eTransportation 1, 100011. (2019).
W. Chen, J. Liang, Z. Yang, and G. Li, Energy Procedia 158, 4363. (2019).
C. Liu, J. Lin, H. Cao, Y. Zhang, and Z. Sun, J. Clean. Prod. 228, 801. (2019).
J. Song, S. Guo, L. Kou, H. Liu, K. Kajiyoshi, J. Su, and P. Zheng, J. Mater. Sci. Mater. Electron. 31, 16037. (2020).
R. Gu, R. Qian, Y. Lyu, and B. Guo, ACS Sustain. Chem. Eng. 8, 25. (2020).
T. Tian, T.-W. Zhang, Y.-C. Yin, Y.-H. Tan, Y.-H. Song, L.-L. Lu, and H.-B. Yao, Nano Lett. 20, 1. (2020).
Z. Cai, Y. Ma, X. Huang, X. Yan, Z. Yu, S. Zhang, G. Song, Y. Xu, C. Wen, and W. Yang, J. Energy Storage 27, 101036. (2020).
H. Lee, S. Kim, N.S. Parmar, J. Chung, K. Kim, and J. Choi, J. Power Sources 434, 226713 (2019).
Y. Shao, B. Huang, Z. Lu, Y. Liu, X. Meng, L. Du, H. Song, and S. Liao, Energy Technol. 7, 4. (2019).
X. Zeng, M. Li, D. Abd El-Hady, W. Alshitari, A.S. Al-Bogami, J. Lu, and K. Amine, Adv. Energy Mater. 9, 27. (2019).
J. Song, S. Guo, D. Ren, H. Liu, L. Kou, J. Su, and P. Zheng, Ceram. Int. 46(Part B), 8. (2020).
P. He, Z. Ding, X. Zhao, J. Liu, S. Yang, P. Gao, and L.-Z. Fan, Inorg. Chem. 58, 19. (2019).
M. Jing, M. Zhou, G. Li, Z. Chen, W. Xu, X. Chen, Z. Hou, and A.C.S. Appl, Mater. Interfaces 9, 11. (2017).
X. Wang, L. Sun, X. Sun, X. Li, and D. He, X. Wang, L. Sun, X. Sun, X. Li, and D. He, Mater. Res. Bull. 96, 533–537. (2017).
H. Fu, Z. Xu, T. Wang, K. Li, X. Shen, J. Li, and J. Huang, J. Electrochem. Soc. 165, 3. (2018).
H. Li, Q. Su, J. Kang, M. Huang, M. Feng, H. Feng, P. Huang, and G. Du, Mater. Lett. 217, 276. (2018).
X. Zhang, Y. Zhu, A.M. Bruck, L.M. Housel, L. Wang, C.D. Quilty, K.J. Takeuchi, E.S. Takeuchi, A.C. Marschilok, and G. Yu, Energy Storage Mater. 19, 439. (2019).
J. Zeng, J. Huang, J. Liu, T. Xie, C. Peng, Y. Lu, P. Lu, R. Zhang, and J. Min, Carbon 154, 24. (2019).
S. Natarajan, S.-J. Kim, and V. Aravindan, J. Mater. Chem. A 8, 19. (2020).
Y. Yue, and H. Liang, Adv. Energy Mater. 7, 17. (2017).
N. Aliahmad, Y. Liu, J. Xie, M. Agarwal, and A.C.S. Appl, Mater. Interfaces 10, 19. (2018).
M. Panagopoulou, D. Vernardou, E. Koudoumas, N. Katsarakis, D. Tsoukalas, and Y.S. Raptis, The. J. Phys. Chem. C 121, 1. (2017).
Y. Ji, D. Fang, C. Wang, Z. Zhou, Z. Luo, J. Huang, and J. Yi, J. Alloys Compd. 742, 567. (2018).
Z. Li, C. Zhang, C. Liu, H. Fu, X. Nan, K. Wang, X. Li, W. Ma, X. Lu, and G. Cao, Electrochim. Acta 222, 1831. (2016).
K.K. Purushothaman, B. Saravanakumar, G. Muralidharan, and M. Dhanashankar, Mater. Technol. 32, 9. (2017).
P. Bhattacharya, T. Joo, M. Kota, and H.S. Park, Ceram. Int. 44, 1. (2018).
K Karthik, K Pradeeswari, R Mohan Kumar, and R Murugesan, Mater. Res. Innov. (2019)
M. Farahmandjou, M. Farahmandjou, J. Nanomed. Res. 5, 00103. (2017).
G. Wang, Y. Meng, L. Wang, J. Xia, F. Zhu, and Y. Zhang, Int. J. Electrochem. Sci. 12, 2618 (2017).
M. Aghazadeh, and M. Aghazadeh, Int. J. Electrochem. Sci. 11, 11002. (2016).
Y. Zhang, Y. Wang, Z. Xiong, Y. Hu, W. Song, Q.-A. Huang, X. Cheng, L.-Q. Chen, C. Sun, and H. Gu, ACS Omega 2, 3. (2017).
J. Wu, I. Byrd, C. Jin, J. Li, H. Chen, T. Camp, R. Bujol, A. Sharma, and H. Zhang, ChemElectroChem 4, 5. (2017).
N. Wu, W. Du, G. Liu, Z. Zhou, H.-R. Fu, Q. Tang, X. Liu, Y.-B. He, and A.C.S. Appl, Mater. Interfaces 9, 50. (2017).
Y. Zhu, M. Yang, Q. Huang, D. Wang, R. Yu, J. Wang, Z. Zheng, and D. Wang, Adv. Mater. 32, 7. (2020).
Acknowledgements
Authors thank to Shaanxi Key Laboratory of Green Preparation and Functionalization for Inorganic Materials for their experimental platform and testing conditions. This work was supported by the National Natural Science Foundation of China (No. 52073166), the Scientific Research Startup Program for Introduced Talents of Shaanxi University of Technology (No. SLGRCQD2025), the Xi’an Key Laboratory of Green Manufacture of Ceramic Materials Foundation (No. 2019220214SYS017CG039), the Key Program for International S&T Cooperation Projects of Shaanxi Province (2020KW-038,2020GHJD-04), Science and Technology Program of Xi'an, China (2020KJRC0009) and Scientific Research Program Funded by Shaanxi Provincial Education Department (No. 20JY001), Science and Technology Resource Sharing Platform of Shaanxi Province (2020PT-022).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The corresponding authors declare no conflict of interest. There are no interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kou, L., Cao, L., Song, J. et al. Cobalt-doped Vanadium Pentoxide Microflowers as Superior Cathode for Lithium-Ion Battery. JOM 73, 808–814 (2021). https://doi.org/10.1007/s11837-020-04552-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-020-04552-3