Abstract
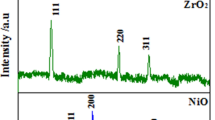
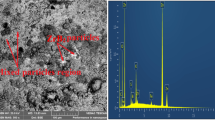
In this study, for marine application purposes, we evaluated the effect of process parameter and particle loading on the microstructure, mechanical reinforcement and corrosion resistance properties of a Zn-TiO2-WO3 nanocomposite produced via electrodeposition. We characterized the morphological properties of the composite coatings with a Scanning Electron Microscope (SEM) equipped with an Energy Dispersive Spectrometer (EDS). We carried out mechanical examination using a Dura Scan hardness tester and a CERT UMT-2 multi-functional tribological tester. We evaluated the corrosion properties by linear polarization in 3.5% NaCl. The results show that the coatings exhibited good stability and the quantitative particle loading greatly enhanced the structural and morphological properties, hardness behavior and corrosion resistance of the coatings. We observed the precipitation of this alloy on steel is greatly influenced by the composite characteristics.
Similar content being viewed by others
References
Afonja AA, 2009. Novel materials for energy applications. Nigerian Journal of Materials Science and Engineering, 1(1), 63–72.
Chin RJ, Nobe K, 1972. Electrodissolution kinetics of iron in chloride solutions. J. Electrochem. Soc., 119(11), 1457–1461.
Daniyan AA, Umoru LE, Fasasi AY, Borode JO, Oluwasegun KM, Olusunle SOO, 2014. Electrical properties of Nano-TiO2 thin film using spin coating method. Journal of Minerals and Materials Characterization and Engineering, 2, 15–20. DOI: 10.4236/jmmce.2014.21003
Fayomi OSI, Aigbodion VS, Popoola API, 2015. Properties of Tic/Tib modified Zn–Tic/Tib ceramic composite coating on mild steel. Journal of Failure Analysis and Prevention, 15(1), 54–64. DOI: 10.1007/s11668-014-9908-1
Fayomi OSI, Joseph OO, Mubiayi MP, Durodola BM, Gabriel O, 2016a. Microstructural evolution and characterization of super-induced MgO composite on zinc-rich coating. Egyptian J. of Basic and Appl. Sci., 3(1), 1–9. DOI: 10.1016/j.ejbas.2015.05.007
Fayomi OSI, Loto CA, Popoola API, Tau V, 2014a. Effect of process parameter on the in-situ intermetallic composite coating and microstructural evolution of Zn-Al2O3 in the presence of TEA/MEA on mild steel. Int. J. Electrochem. Sci.,9, 7359–7368.
Fayomi OSI, Popoola API, Adams FV, 2016b. Evolution and anticorrosion properties of ni-cu thin film coatings on low carbon steel. Asian Journal of Chemistry, 28(1), 129. DOI: 10.14233/ajchem.2016.19270
Fayomi OSI, Popoola API, Aigbodion VS, 2014b. Effect of thermal treatment on the interfacial reaction, microstructural and mechanical properties of Zn-Al-SnO2/TiO2 functional coating alloys. Journal of Alloys and Compounds, 617, 455–463. DOI: 10.1016/j.jallcom.2014.07.141
Ger MD, Grebe R, 2004. Electrochemical deposition of nickel/SiC composites in the presence of surfactants. Materials Chemistry and Physics, 87(1), 67–74. DOI: 10.1016/j.matchemphys.2004.04.022
Kwok CT, Cheng FT, Man HC, 2006. Cavitation erosion and corrosion behaviours of laser-aluminized mild steel. J Surf Coat Technol, 200(11), 3544–3552. DOI: 10.1016/j.surfcoat.2004.12.016
Noor EA, Al-Moubaraki AH, 2008. Corrosion behavior of mild steel in hydrochloric acid solutions. Int. J. Electrochem. Sci., 3, 806–818.
Ogundare O, Babatope B, Adetunji AR, Olusunle SOO, 2012. Atmospheric corrosion studies of ductile iron and austenitic stainless steel in an extreme marine environment. Journal of Minerals and Materials Characterization and Engineering, 11, 914–918.
Panek J, Bierska-Piech B, Karolus M, 2011. The corrosion resistance of zinc-nickel composite coatings. Journal of Achievements in Materials and Manufacturing Engineering, 45(2), 157–162.
Paunovic M, Mordechay S, 2006. Fundamentals of electrochemical deposition. 2nd ed, John Wiley and Son Inc., University of Windsor, Windsor,6, 388–391.
Popoola API, Fayomi OSI, 2011. ZnO as corrosion inhibitor for dissolution of zinc electrodeposited mild steel in varying HCl concentration. Int. J. of the Phy. Sci., 6, 2447–2454.
Popoola API, Fayomi OSI, Popoola OM, 2012. Comparative studies of microstructural, tribological and corrosion properties of plated Zn and Zn-alloy coatings. International Journal of Electrochemical Science, 7, 4860–4870.
Praveen BM, Venkatesha TV, 2011. Electrodeposition and corrosion resistance properties of Zn-Ni/TiO2 nano-composite coatings. International Journal of Electrochemistry, 1–4.
Rahman MJ, Sen SR, Moniruzzaman M, Shorowordi KM, 2009. Morphology and properties of electrodeposited Zn-Ni alloy coatings on mild steel. Journal of Mechanical Engineering, 40(1), 9–14.
Shibli SMA, Chacko F, Divya C, 2010. Al2O3–ZrO2 mixed oxide composite incorporated aluminium rich zinc coatings for high wear resistance. Journal Corrosion Science, 52(2), 518–525. DOI: 10.1016/j.corsci.2009.10.008
Shivakumara S, Manohar U, Arthoba Naik Y, Venkatesha TU, 2007. Influence of additives on electro-deposition of bright Zn-Ni alloy on mild steel from acid sulphates bath. Bull Mater Sci, 30, 455–462.
Su YL, Kao WH, 2011. Tribological behaviour and wear mechanism of MoS2–Cr coatings sliding against various counter body. Tribology International, 36(1), 11–23. DOI: 10.1016/S0301-679X(02)00095-6
Umoru LE, Fawehinmi IA, Fasasi AY, 2006. Investigation of the inhibitive influence of theobroma cacao and cola acuminata leaves extracts on the corrosion of mild steel in sea water. Journal of Applied Sciences Research, 2(4), 200–204.
Wang P, Cheng Y, Zhang Z, 2011. A study on the electrocodeposition processes and properties of Ni-SiC nanocomposite coating. Journal of Coatings Technology and Research, 8(3), 409–417. DOI: 10.1007/s11998-010-9310-1
Acknowledgement
This work is based upon the financial support of National Research Foundation and effort by the Surface Engineering Research Centre (SERC), Department of Chemical Metallurgical and Materials Engineering, the Tshwane University of Technology, Pretoria, South Africa. The privilege given by Department of Materials Science and Engineering, Obafemi Awolowo University to undertake this research work is also appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Popoola, A.P.I., Daniyan, A.A., Umoru, L.E. et al. Effect of WO3 nanoparticle loading on the microstructural, mechanical and corrosion resistance of Zn matrix/TiO2-WO3 nanocomposite coatings for marine application. J. Marine. Sci. Appl. 16, 102–109 (2017). https://doi.org/10.1007/s11804-017-1389-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11804-017-1389-7