Abstract
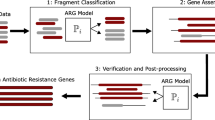
Analysis of environmental samples for bacterial antibiotic resistance genes may have different objectives and analysis strategies. In some cases, the purpose was to study diversity and evolution of genes that could be grouped within a mechanism of antibiotic resistance. Different protocols have been designed for detection and confirmation that a functional gene was found. In this study, we present a sequence-based screening of candidate genes encoding beta-lactamases in 14 metagenomes of Antarctic microbial mats. The samples were obtained from different sites, representing diverse biogeographic regions of maritime and continental Antarctica. A protocol was designed based on generation of Hidden Markov Models from the four beta-lactamase classes by Ambler classification, using sequences from the Comprehensive Antibiotic Resistance Database (CARD). The models were used as queries for metagenome analysis and recovered contigs were subsequently annotated using RAST. According to our analysis, 14 metagenomes analyzed contain A, B and C beta-lactamase genes. Class D genes, however, were identified in 11 metagenomes. The most abundant was class C (46.8%), followed by classes B (35.5%), A (14.2%) and D (3.5%). A considerable number of sequences formed clusters which included, in some cases, contigs from different metagenomes. These assemblies are clearly separated from reference clusters, previously identified using CARD beta-lactamase sequences. While bacterial antibiotic resistance is a major challenge of public health worldwide, our results suggest that environmental diversity of beta-lactamase genes is higher than that currently reported, although this should be complemented with gene function analysis.

Similar content being viewed by others
References
Al Bayssari C, Olaitan A O, Dabboussi F, Hamze M, Rolain J M (2015). Emergence of OXA-48-producing Escherichia coli clone ST38 in fowl. Antimicrobial Agents and Chemotherapy, 59(1): 745–746
Allen H K, Donato J, Wang H H, Cloud-Hansen K A, Davies J, Handelsman J (2010). Call of the wild: Antibiotic resistance genes in natural environments. Nature Reviews. Microbiology, 8(4): 251–259
Allen H K, Moe L A, Rodbumrer J, Gaarder A, Handelsman J (2009). Functional metagenomics reveals diverse ß-lactamases in a remote Alaskan soil. The ISME Journal, 3(2): 243–251
Aziz R K, Bartels D, Best A A, DeJongh M, Disz T, Edwards R A, Formsma K, Gerdes S, Glass E M, Kubal M, Meyer F, Olsen G J, Olson R, Osterman A L, Overbeek R A, McNeil L K, Paarmann D, Paczian T, Parrello B, Pusch G D, Reich C, Stevens R, Vassieva O, Vonstein V, Wilke A, Zagnitko O (2008). The RAST Server: rapid annotations using subsystems technology. BMC Genomics, 9(1): 75–90
Babic M, Hujer A M, Bonomo R A (2006). What’s new in antibiotic resistance? Focus on beta-lactamases. Drug Resistance Updates: Reviews and Commentaries in Antimicrobial and Anticancer Chemotherapy, 9(3): 142–156
Bennett J W, Chung K T (2001). Alexander Fleming and the discovery of penicillin. Advances in Applied Microbiology, 49: 163–184
Berglund F, Österlund T, Boulund F, Marathe N P, Larsson D G J, Kristiansson E (2019). Identification and reconstruction of novel antibiotic resistance genes from metagenomes. Microbiome, 7(1): 52–66
Bonnet R (2004). Growing group of extended-spectrum ß-lactamases: The CTX-M enzymes. Antimicrobial Agents and Chemotherapy, 48 (1): 1–14
Boolchandani M, D’Souza A W, Dantas G (2019). Sequencing-based methods and resources to study antimicrobial resistance. Nature Reviews. Genetics
Bush K, Courvalin P, Dantas G, Davies J, Eisenstein B, Huovinen P, Jacoby G A, Kishony R, Kreiswirth B N, Kutter E, Lerner S A, Levy S, Lewis K, Lomovskaya O, Miller J H, Mobashery S, Piddock L J, Projan S, Thomas C M, Tomasz A, Tulkens P M, Walsh T R, Watson J D, Witkowski J, Witte W, Wright G, Yeh P, Zgurskaya H I (2011). Tackling antibiotic resistance. Nature Reviews. Microbiology, 9(12): 894–896
Chen Y P, Lee S H, Chou C H, Tsai H J (2012). Detection of florfenicol resistance genes in Riemerella anatipestifer isolated from ducks and geese. Veterinary Microbiology, 154(3–4): 325–331
Coudron P E, Moland E S, Thomson K S (2000). Occurrence and detection of AmpC beta-lactamases among Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis isolates at a veterans medical center. Journal of Clinical Microbiology, 38(5): 1791–1796
Davies J, Davies D (2010). Origins and evolution of antibiotic resistance. Microbiology and Molecular Biology Reviews: MMBR, 74(3): 417–433
de Been M, Lanza V F, de Toro M, Scharringa J, Dohmen W, Du Y, Hu J, Lei Y, Li N, Tooming-Klunderud A, Heederik D J, Fluit A C, Bonten M J, Willems R J, de la Cruz F, van Schaik W (2014). Dissemination of cephalosporin resistance genes between Escherichia coli strains from farm animals and humans by specific plasmid lineages. PLOS Genetics, 10(12): e1004776–e1004793
Evans B A, Amyes S G (2014). OXA ß-lactamases. Clinical Microbiology Reviews, 27(2): 241–263
Fu L, Niu B, Zhu Z, Wu S, Li W (2012). CD-HIT: accelerated for clustering the next-generation sequencing data. Bioinformatics (Oxford, England), 28(23): 3150–3152
Garau G, García-Sáez I, Bebrone C, Anne C, Mercuri P, Galleni M, Frère J M, Dideberg O (2004). Update of the standard numbering scheme for class B ß-lactamases. Antimicrobial Agents and Chemotherapy, 48(7): 2347–2349
Gibson M K, Forsberg K J, Dantas G (2015). Improved annotation of antibiotic resistance determinants reveals microbial resistomes cluster by ecology. The ISME Journal, 9(1): 207–216
Hall B G, Barlow M (2004). Evolution of the serine ß-lactamases: Past, present and future. Drug Resistance Updates: Reviews and Commentaries in Antimicrobial and Anticancer Chemotherapy, 7(2): 111–123
Hall B G, Barlow M (2005). Revised Ambler classification of ß- lactamases. The Journal of Antimicrobial Chemotherapy, 55(6): 1050–1051
Högberg L D, Heddini A, Cars O (2010). The global need for effective antibiotics: Challenges and recent advances. Trends in Pharmacological Sciences, 31(11): 509–515
Hughes K A, Thompson A (2004). Distribution of sewage pollution around a maritime Antarctic research station indicated by faecal coliforms, Clostridium perfringens and faecal sterol markers. Environmental Pollution (Barking, Essex: 1987), 127(3): 315–321
Jacoby G A (2009). AmpC ß-lactamases. Clinical Microbiology Reviews, 22(1): 161–182
Jeong S H, Bae I K, Lee J H, Sohn S G, Kang G H, Jeon G J, Kim Y H, Jeong B C, Lee S H (2004). Molecular characterization of extendedspectrum beta-lactamases produced by clinical isolates of Klebsiella pneumoniae and Escherichia coli from a Korean nationwide survey. Journal of Clinical Microbiology, 42(7): 2902–2906
June C M, Vallier B C, Bonomo R A, Leonard D A, Powers R A (2014). Structural origins of oxacillinase specificity in class D ß-lactamases. Antimicrobial Agents and Chemotherapy, 58(1): 333–341
Lisa M N, Palacios A R, Aitha M, González M M, Moreno D M, Crowder M W, Bonomo R A, Spencer J, Tierney D L, Llarrull L I, Vila A J (2017). A general reaction mechanism for carbapenem hydrolysis by mononuclear and binuclear metallo-ß-lactamases. Nature Communications, 8(1): 538–549
Martínez J L (2008). Antibiotics and antibiotic resistance genes in natural environments. Science, 321(5887): 365–367
Naas T, Poirel L, Nordmann P (2008). Minor extended-spectrum ß- lactamases. Clinical Microbiology and Infection, 14(Suppl 1): 42–52
Nesme J, Cécillon S, Delmont T O, Monier J M, Vogel T M, Simonet P (2014). Large-scale metagenomic-based study of antibiotic resistance in the environment. Current Biology: CB, 24(10): 1096–1100
Peng Y, Leung H C, Yiu S M, Chin F Y (2012). IDBA-UD: a de novo assembler for single-cell and metagenomic sequencing data with highly uneven depth. Bioinformatics (Oxford, England), 28(11): 1420–1428
Pérez-Pérez F J, Hanson N D (2002). Detection of plasmid-mediated AmpC ß-lactamase genes in clinical isolates by using multiplex PCR. Journal of Clinical Microbiology, 40(6): 2153–2162
Quince C, Walker A W, Simpson J T, Loman N J, Segata N (2017). Shotgun metagenomics, from sampling to analysis. Nature Biotechnology, 35(9): 833–844
Ruppé E, Ghozlane A, Tap J, Pons N, Alvarez A S, Maziers N, Cuesta T, Hernando-Amado S, Clares I, Martínez J L, Coque T M, Baquero F, Lanza V F, Máiz L, Goulenok T, de Lastours V, Amor N, Fantin B, Wieder I, Andremont A, van Schaik W, Rogers M, Zhang X, Willems R J L, de Brevern A G, Batto J M, Blottière H M, Léonard P, Léjard V, Letur A, Levenez F, Weiszer K, Haimet F, Doré J, Kennedy S P, Ehrlich S D (2019). Prediction of the intestinal resistome by a threedimensional structure-based method. Nature Microbiology, 4(1): 112–123
Segawa T, Takeuchi N, Rivera A, Yamada A, Yoshimura Y, Barcaza G, Shinbori K, Motoyama H, Kohshima S, Ushida K (2013). Distribution of antibiotic resistance genes in glacier environments. Environmental Microbiology Reports, 5(1): 127–134
Shaikh S, Fatima J, Shakil S, Rizvi S M D, Kamal M A (2015). Antibiotic resistance and extended spectrum beta-lactamases: Types, epidemiology and treatment. Saudi Journal of Biological Sciences, 22(1): 90–101
Van Goethem MW, Pierneef R, Bezuidt O K I, Van De Peer Y, Cowan D A, Makhalanyane T P (2018). A reservoir of ‘historical’ antibiotic resistance genes in remote pristine Antarctic soils. Microbiome, 6(1): 40–52
Wallace J C, Port J A, Smith M N, Faustman E M (2017). FARME DB: A functional antibiotic resistance element database. Database (Oxford), 2017: baw165–7
Wright G D (2010). Antibiotic resistance in the environment: A link to the clinic? Current Opinion in Microbiology, 13(5): 589–594
Author information
Authors and Affiliations
Corresponding author
Additional information
Highlights
• Beta-lactamase genes were found in all samples from distant places in Antarctica.
• Class C beta-lactamase coding genes were the most frequently found.
• Diversity of sequences exceeds that of the beta-lactamases from clinical environment.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Azziz, G., Giménez, M., Romero, H. et al. Detection of presumed genes encoding beta-lactamases by sequence based screening of metagenomes derived from Antarctic microbial mats. Front. Environ. Sci. Eng. 13, 44 (2019). https://doi.org/10.1007/s11783-019-1128-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11783-019-1128-1