Abstract
Background
Chemotherapy-induced peripheral neuropathy (CIPN) is a significant and often lasting side effect of cancer treatment, with increasing CIPN severity associated with increasing deficits in balance, gait, and mobility. The 6-min walk test (6MWT) is a widely validated and utilized measure of general physical functioning and mobility, although its utility in a CIPN context is unclear. This study aimed to determine the utility of the 6MWT as an assessment of mobility deficits in a CIPN cohort and utilize the 6MWT to compare mobility data from CIPN patients to those of healthy and clinical populations.
Methods
Cancer survivors exposed to neurotoxic chemotherapies (N = 100; mean 17 ± 13 months post-treatment; mean age 59 ± 13 years) completed a single cross-sectional assessment of patient-reported and objective CIPN, mobility (6MWT), and disability.
Results
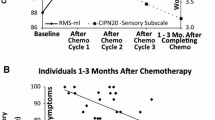
CIPN symptoms were reported in the majority of the cohort (87%). Increasing age, patient-reported and objective CIPN symptoms, and disability were associated with decreasing 6MWT distance (.48 ≤ R ≤ .63; p < .001) in bivariate models. Multiple regression models of 6MWT distance included age, sex, and patient-reported or objective CIPN severity as significant independent correlates (.62 ≤ R ≤ .64; p < .03). 6MWT distances in patients with CIPN symptom severity above the cohort mean were consistent with mean values reported in diabetic neuropathy and clinical populations.
Conclusions
Increased CIPN symptoms are associated with increased mobility deficits. The 6MWT demonstrates promising utility as a mobility assessment in a CIPN cohort.
Implications for Cancer Survivors
The impact of the progression of CIPN on mobility deficits in survivors emphasizes the need for effective interventions to treat and prevent CIPN.


Similar content being viewed by others
References
Seretny M, Currie GL, Sena ES, Ramnarine S, Grant R, MacLeod MR, et al. Incidence, prevalence, and predictors of chemotherapy-induced peripheral neuropathy: a systematic review and meta-analysis. PAIN. 2014;155(12):2461–70.
Park SB, Goldstein D, Krishnan AV, Lin CSY, Friedlander ML, Cassidy J, et al. Chemotherapy-induced peripheral neurotoxicity: a critical analysis. CA Cancer J Clin. 2013;63(6):419–37.
Wolf S, Barton D, Kottschade L, Grothey A, Loprinzi C. Chemotherapy-induced peripheral neuropathy: prevention and treatment strategies. Eur J Cancer. 2008;44(11):1507–15.
Winters-Stone KM, Horak F, Jacobs PG, Trubowitz P, Dieckmann NF, Stoyles S, et al. Falls, functioning, and disability among women with persistent symptoms of chemotherapy-induced peripheral neuropathy. J Clin Oncol. 2017;35:2604–12 JCO 2016.71. 3552.
Hile ES, Fitzgerald GK, Studenski SA. Persistent mobility disability after neurotoxic chemotherapy. Phys Ther. 2010;90(11):1649–57.
Gewandter J, Fan L, Magnuson A, Mustian K, Peppone L, Heckler C, et al. Falls and functional impairments in cancer survivors with chemotherapy-induced peripheral neuropathy (CIPN): a University of Rochester CCOP study. Support Care Cancer. 2013;21(7):2059–66.
Varedi M, Lu L, Howell CR, Partin RE, Hudson MM, Pui C-H, et al. Peripheral neuropathy, sensory processing, and balance in survivors of acute lymphoblastic leukemia. J Clin Oncol. 2018;36:2315–22 JCO2017.76. 7871.
Monfort SM, Pan X, Patrick R, Ramaswamy B, Wesolowski R, Naughton MJ, et al. Gait, balance, and patient-reported outcomes during taxane-based chemotherapy in early-stage breast cancer patients. Breast Cancer Res Treat. 2017;161(1):69–77.
Gewandter JS, Brell J, Cavaletti G, Dougherty PM, Evans S, Howie L, et al. Trial designs for chemotherapy-induced peripheral neuropathy prevention: ACTTION recommendations. Neurology. 2018;91(9):403–13.
Lord SR, Menz HB. Physiologic, psychologic, and health predictors of 6-minute walk performance in older people. Arch Phys Med Rehabil. 2002;83(7):907–11.
Harada ND, Chiu V, Stewart AL. Mobility-related function in older adults: assessment with a 6-minute walk test. Arch Phys Med Rehabil. 1999;80(7):837–41.
Kervio G, Carre F, Ville NS. Reliability and intensity of the six-minute walk test in healthy elderly subjects. Med Sci Sports Exerc. 2003;35(1):169–74.
Ries JD, Echternach JL, Nof L, Gagnon Blodgett M. Test-retest reliability and minimal detectable change scores for the timed “up & go” test, the six-minute walk test, and gait speed in people with Alzheimer disease. Phys Ther. 2009;89(6):569–79.
Hamilton DM, Haennel R. Validity and reliability of the 6-minute walk test in a cardiac rehabilitation population. J Cardiopulm Rehabil Prev. 2000;20(3):156–64.
Schmidt K, Vogt L, Thiel C, Jäger E, Banzer W. Validity of the six-minute walk test in cancer patients. Int J Sports Med. 2013;34(07):631–6.
Enright PL, Sherrill DL. Reference equations for the six-minute walk in healthy adults. Am J Respir Crit Care Med. 1998;158(5):1384–7.
Casanova C, Celli B, Barria P, Casas A, Cote C, De Torres J, et al. The 6-min walk distance in healthy subjects: reference standards from seven countries. Eur Respir J. 2011;37(1):150–6.
Troosters T, Gosselink R, Decramer M. Six minute walking distance in healthy elderly subjects. Eur Respir J. 1999;14(2):270–4.
Bittner V, Weiner DH, Yusuf S, Rogers WJ, Mcintyre KM, Bangdiwala SI, et al. Prediction of mortality and morbidity with a 6-minute walk test in patients with left ventricular dysfunction. Jama. 1993;270(14):1702–7.
Enright PL. The six-minute walk test. Respir Care. 2003;48(8):783–5.
Alberti P, Rossi E, Cornblath D, Merkies I, Postma T, Frigeni B, et al. Physician-assessed and patient-reported outcome measures in chemotherapy-induced sensory peripheral neurotoxicity: two sides of the same coin. Ann Oncol. 2013;25(1):257–64.
Cornblath D, Chaudhry V, Carter K, Lee D, Seysedadr M, Miernicki M, et al. Total neuropathy score validation and reliability study. Neurology. 1999;53(8):1660–4.
Cavaletti G, Frigeni B, Lanzani F, Piatti M, Rota S, Briani C, et al. The Total Neuropathy Score as an assessment tool for grading the course of chemotherapy-induced peripheral neurotoxicity: comparison with the National Cancer Institute-Common Toxicity Scale. J Peripher Nerv Syst. 2007;12(3):210–5.
Postma T, Aaronson N, Heimans J, Muller M, Hildebrand J, Delattre J-Y, et al. The development of an EORTC quality of life questionnaire to assess chemotherapy-induced peripheral neuropathy: the QLQ-CIPN20. Eur J Cancer. 2005;41(8):1135–9.
Binda D, Vanhoutte E, Cavaletti G, Cornblath D, Postma T, Frigeni B, et al. Rasch-built Overall Disability Scale for patients with chemotherapy-induced peripheral neuropathy (CIPN-R-ODS). Eur J Cancer. 2013;49(13):2910–8.
Australian Bureau of Statistics. Profiles of health, Australia, 2011-13. Canberra 2012.
Hinkle DE, Wiersma W, Jurs SG. Applied statistics for the behavioral sciences. Boston: Houghton Mifflin; 1988.
Mueller MJ, Tuttle LJ, LeMaster JW, Strube MJ, McGill JB, Hastings MK, et al. Weight-bearing versus nonweight-bearing exercise for persons with diabetes and peripheral neuropathy: a randomized controlled trial. Arch Phys Med Rehabil. 2013;94(5):829–38.
Wevers LE, Kwakkel G, van de Port IG. Is outdoor use of the six-minute walk test with a global positioning system in stroke patients' own neighbourhoods reproducible and valid? J Rehabil Med. 2011;43(11):1027–31.
Cote CG, Casanova C, Marin J, Lopez M, Pinto-Plata V, De Oca M, et al. Validation and comparison of reference equations for the 6-min walk distance test. Eur Respir J. 2008;31(3):571–8.
Peters E, Schulz LM, Reuss-Borst M. Quality of life after cancer—how the extent of impairment is influenced by patient characteristics. BMC Cancer. 2016;16(1):787.
Huang MH, Blackwood J, Godoshian M, Pfalzer L. Prevalence of self-reported falls, balance or walking problems in older cancer survivors from surveillance, epidemiology and end results—Medicare Health Outcomes Survey. J Geriatr Oncol. 2017;8(4):255–61.
Kaltsatou A, Mameletzi D, Douka S. Physical and psychological benefits of a 24-week traditional dance program in breast cancer survivors. J Bodyw Mov Ther. 2011;15(2):162–7.
LaStayo PC, Marcus RL, Dibble LE, Smith SB, Beck SL. Eccentric exercise versus usual-care with older cancer survivors: the impact on muscle and mobility-an exploratory pilot study. BMC Geriatr. 2011;11(1):5.
Basen-Engquist K, Taylor CLC, Rosenblum C, Smith MA, Shinn EH, Greisinger A, et al. Randomized pilot test of a lifestyle physical activity intervention for breast cancer survivors. Patient Educ Couns. 2006;64(1–3):225–34.
Yuen HK, Sword D. Home-based exercise to alleviate fatigue and improve functional capacity among breast cancer survivors. J Allied Health. 2007;36(4):257E–75E.
Bruce DG, Davis WA, Davis TM. Longitudinal predictors of reduced mobility and physical disability in patients with type 2 diabetes: the Fremantle Diabetes Study. Diabetes Care. 2005;28(10):2441–7.
van Sloten TT, Savelberg HH, Duimel-Peeters IG, Meijer K, Henry RM, Stehouwer CD, et al. Peripheral neuropathy, decreased muscle strength and obesity are strongly associated with walking in persons with type 2 diabetes without manifest mobility limitations. Diabetes Res Clin Pract. 2011;91(1):32–9.
Vita GL, Stancanelli C, Gentile L, Barcellona C, Russo M, Di Bella G, et al. 6MWT performance correlates with peripheral neuropathy but not with cardiac involvement in patients with hereditary transthyretin amyloidosis (hATTR). Neuromuscul Disord. 2018;29(3):213–20.
Spina E, Topa A, Iodice R, Tozza S, Ruggiero L, Dubbioso R, et al. Six-minute walk test is reliable and sensitive in detecting response to therapy in CIDP. J Neurol. 2019;266(4):860–65.
Hirvensalo M, Rantanen T, Heikkinen E. Mobility difficulties and physical activity as predictors of mortality and loss of independence in the community-living older population. J Am Geriatr Soc. 2000;48(5):493–8.
Passantino A, Lagioia R, Mastropasqua F, Scrutinio D. Short-term change in distance walked in 6 min is an indicator of outcome in patients with chronic heart failure in clinical practice. J Am Coll Cardiol. 2006;48(1):99–105.
Ottaiano A, Nappi A, Tafuto S, Nasti G, De Divitiis C, Romano C, et al. Diabetes and body mass index are associated with neuropathy and prognosis in colon cancer patients treated with capecitabine and oxaliplatin adjuvant chemotherapy. Oncology. 2016;90(1):36–42.
Bao T, Basal C, Seluzicki C, Li SQ, Seidman AD, Mao JJ. Long-term chemotherapy-induced peripheral neuropathy among breast cancer survivors: prevalence, risk factors, and fall risk. Breast Cancer Res Treat. 2016;159(2):327–33.
McCrary JM, Goldstein D, Sandler CX, Barry BK, Marthick M, Timmins HC, et al. Exercise-based rehabilitation for cancer survivors with chemotherapy-induced peripheral neuropathy. Support Care Cancer. 2019. https://doi.org/10.1007/s00520-019-04680-w.
Zimmer P, Trebing S, Timmers-Trebing U, Schenk A, Paust R, Bloch W, et al. Eight-week, multimodal exercise counteracts a progress of chemotherapy-induced peripheral neuropathy and improves balance and strength in metastasized colorectal cancer patients: a randomized controlled trial. Support Care Cancer. 2018;26(2):615–24.
Streckmann F, Lehmann H, Balke M, Schenk A, Oberste M, Heller A, et al. Sensorimotor training and whole-body vibration training have the potential to reduce motor and sensory symptoms of chemotherapy-induced peripheral neuropathy—a randomized controlled pilot trial. Support Care Cancer. 2019;27(7):2471–78.
Hershman DL, Lacchetti C, Dworkin RH, Lavoie Smith EM, Bleeker J, Cavaletti G, et al. Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2014;32(18):1941–67.
Acknowledgments
This study was supported by a Cancer Institute NSW Program Grant (14/TPG/1-05) and a National Health and Medical Research Council of Australia (NHMRC) Project Grant (#1080521). SBP is supported by a NHMRC Career Development Fellowship (#1148595).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
McCrary, J.M., Goldstein, D., Wyld, D. et al. Mobility in survivors with chemotherapy-induced peripheral neuropathy and utility of the 6-min walk test. J Cancer Surviv 13, 495–502 (2019). https://doi.org/10.1007/s11764-019-00769-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-019-00769-7