Abstract
Purpose
Advances in cancer detection and treatment have resulted in a growing population of long-term survivors, but even years after treatment has concluded, many survivors report physical symptoms that interfere with daily living. While there are studies of late effects following common cancers, less is known about these complications in rare cancers. This study focuses on the physical symptoms reported by long-term survivors enrolled in the NIH-sponsored Rare Cancer Genetics Registry.
Methods
The Rotterdam Symptom Checklist-Modified was administered to evaluate the severity of physical symptoms commonly reported by long-term cancer survivors. Logistic regression was used to assess association between symptoms and demographic and clinical factors.
Results
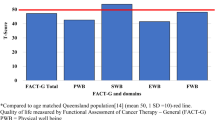
In 309 subjects with a median time of 7.6 years from a diagnosis of one or more rare cancers, the median number of symptoms present per participant was 7. The most prevalent symptom reported was tiredness/lack of energy, which was present/very bothersome in 70%/25% of registrants. Women, non-whites, current smokers, and upper GI cancer survivors are particularly affected. Overall, symptom prevalence was similar across rare cancer types, time since diagnosis, and type of treatment.
Conclusions
Rare cancer survivors continue to experience troublesome symptoms many years after diagnosis, regardless of cancer type or treatment modality.
Implications for Cancer Survivors
There is a need for continued emphasis on smoking cessation in cancer survivors as well as enhanced monitoring of long-term complications in female, non-white, and upper GI cancer survivors.

Similar content being viewed by others
References
Welch HG, Schwartz LM, Woloshin S. Are increasing 5-year survival rates evidence of success against cancer? JAMA. 2000;283(22):2975–8.
Bluethmann SM, Mariotto AB, Rowland JH. Anticipating the “silver tsunami”: prevalence trajectories and comorbidity burden among older cancer survivors in the United States. Cancer Epidemiol Biomark Prev. 2016;25(7):1029–36. https://doi.org/10.1158/1055-9965.EPI-16-0133.
Mayer DK, Nasso SF, Earp JA. Defining cancer survivors, their needs, and perspectives on survivorship health care in the USA. Lancet Oncol. 2017;18(1):e11–e8. https://doi.org/10.1016/S1470-2045(16)30573-3.
Pachman DR, Barton DL, Swetz KM, Loprinzi CL. Troublesome symptoms in cancer survivors: fatigue, insomnia, neuropathy, and pain. J Clin Oncol. 2012;30(30):3687–96. https://doi.org/10.1200/JCO.2012.41.7238.
Skerman HK, Yates PM, Battitutta D. Cancer-related symptom clusters for symptom management in outpatients after commencing adjuvant chemotherapy, at 6 months, and 12 months. Support Care Cancer. 2012;20(1):95–105. https://doi.org/10.1007/s00520-010-1070-z.
Tian Y, Schofield PE, Gough K, Mann GB. Profile and predictors of long-term morbidity in breast cancer survivors. Ann Surg Oncol. 2013;20(11):3453–60. https://doi.org/10.1245/s10434-013-3004-8.
Yang P, Cheville AL, Wampfler JA, Garces YI, Jatoi A, Clark MM, et al. Quality of life and symptom burden among long-term lung cancer survivors. J Thorac Oncol. 2012;7(1):64–70. https://doi.org/10.1097/JTO.0b013e3182397b3e.
Ganz PA. Late effects of cancer and its treatment. Semin Oncol Nurs. 2001;17(4):241–8.
de Haes JC, van Knippenberg FC, Neijt JP. Measuring psychological and physical distress in cancer patients: structure and application of the Rotterdam Symptom Checklist. Br J Cancer. 1990;62(6):1034–8.
Stein KD, Denniston M, Baker F, Dent M, Hann DM, Bushhouse S, et al. Validation of a modified Rotterdam symptom checklist for use with cancer patients in the United States. J Pain Symptom Manag. 2003;26(5):975–89.
Meyerowitz BE, Desmond KA, Rowland JH, Wyatt GE, Ganz PA. Sexuality following breast cancer. J Sex Marital Ther. 1999;25(3):237–50. https://doi.org/10.1080/00926239908403998.
Syrjala KL, Roth-Roemer SL, Abrams JR, Scanlan JM, Chapko MK, Visser S, et al. Prevalence and predictors of sexual dysfunction in long-term survivors of marrow transplantation. J Clin Oncol. 1998;16(9):3148–57. https://doi.org/10.1200/JCO.1998.16.9.3148.
Langendijk JA, Doornaert P, Verdonck-de Leeuw IM, Leemans CR, Aaronson NK, Slotman BJ. Impact of late treatment-related toxicity on quality of life among patients with head and neck cancer treated with radiotherapy. J Clin Oncol. 2008;26(22):3770–6. https://doi.org/10.1200/JCO.2007.14.6647.
Head MDA, Neck Cancer Symptom Working G, Kamal M, Rosenthal DI, Volpe S, Goepfert RP, et al. Patient reported dry mouth: instrument comparison and model performance for correlation with quality of life in head and neck cancer survivors. Radiother Oncol. 2018;126(1):75–80. https://doi.org/10.1016/j.radonc.2017.10.037.
Allum WH, Bonavina L, Cassivi SD, Cuesta MA, Dong ZM, Felix VN, et al. Surgical treatments for esophageal cancers. Ann N Y Acad Sci. 2014;1325:242–68. https://doi.org/10.1111/nyas.12533.
Bouras G, Markar SR, Burns EM, Huddy JR, Bottle A, Athanasiou T, et al. The psychological impact of symptoms related to esophagogastric cancer resection presenting in primary care: a national linked database study. Eur J Surg Oncol. 2017;43(2):454–60. https://doi.org/10.1016/j.ejso.2016.10.010.
Mustafa Ali M, Moeller M, Rybicki L, Moore HCF. Long-term peripheral neuropathy symptoms in breast cancer survivors. Breast Cancer Res Treat. 2017;166(2):519–26. https://doi.org/10.1007/s10549-017-4437-8.
Hwang IC, Yun YH, Kim YW, Ryu KW, Kim YA, Kim S, et al. Factors related to clinically relevant fatigue in disease-free stomach cancer survivors and expectation-outcome consistency. Support Care Cancer. 2014;22(6):1453–60. https://doi.org/10.1007/s00520-013-2110-2.
Thong MSY, Mols F, van de Poll-Franse LV, Sprangers MAG, van der Rijt CCD, Barsevick AM, et al. Identifying the subtypes of cancer-related fatigue: results from the population-based PROFILES registry. J Cancer Surviv. 2018;12(1):38–46. https://doi.org/10.1007/s11764-017-0641-0.
Kim IR, Kim SH, Ok ON, Kim SH, Lee S, Choi E, et al. Sexual problems in male vs. female non-Hodgkin lymphoma survivors: prevalence, correlates, and associations with health-related quality of life. Ann Hematol. 2017;96(5):739–47. https://doi.org/10.1007/s00277-017-2940-y.
Blinder VS, Griggs JJ. Health disparities and the cancer survivor. Semin Oncol. 2013;40(6):796–803. https://doi.org/10.1053/j.seminoncol.2013.09.003.
Blanchard CM, Courneya KS, Stein K, American Cancer Society’s SCS, II. Cancer survivors’ adherence to lifestyle behavior recommendations and associations with health-related quality of life: results from the American Cancer Society’s SCS-II. J Clin Oncol. 2008;26(13):2198–204. https://doi.org/10.1200/JCO.2007.14.6217.
Ditre JW, Gonzalez BD, Simmons VN, Faul LA, Brandon TH, Jacobsen PB. Associations between pain and current smoking status among cancer patients. Pain. 2011;152(1):60–5. https://doi.org/10.1016/j.pain.2010.09.001.
Kushi LH, Doyle C, McCullough M, Rock CL, Demark-Wahnefried W, Bandera EV, et al. American Cancer Society guidelines on nutrition and physical activity for cancer prevention: reducing the risk of cancer with healthy food choices and physical activity. CA Cancer J Clin. 2012;62(1):30–67. https://doi.org/10.3322/caac.20140.
Acknowledgements
The authors would like to gratefully acknowledge the Rare Cancer Genetics Registry participants and project managers, as well as the Principal Investigators of the Rare Cancer Genetics Registry: Susan Domchek (University of Pennsylvania), Claudine Isaacs (Georgetown University), Jan Lowery and Betsy Risendal (University of Colorado Denver), Patricia Moorman (Duke University), Kala Visvanathan (Johns Hopkins University).
Funding
This study was funded by the U.S. National Institutes of Health (grants RC1 CA144706 and R01 CA160233).
Author information
Authors and Affiliations
Contributions
DMF concept; NKH, DMF data analysis; NKH, AM, DMF manuscript writing; HLG, KLB recruitment and development of idea and tools; AM, HLG, KLB manuscript review.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Online Resource 1
(PDF 62 kb)
Online Resource 2
(PDF 36 kb)
Rights and permissions
About this article
Cite this article
Horick, N.K., Muzikansky, A., Gutierrez, H.L. et al. Physical symptoms in long-term survivors of rare cancer. J Cancer Surviv 12, 835–842 (2018). https://doi.org/10.1007/s11764-018-0721-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-018-0721-9