Abstract
Purpose
Endocrine therapy reduces the risk of breast cancer recurrences and mortality in hormone receptor-positive (HR+) breast cancer survivors. However, non-adherence to treatment remains a significant problem. The aim of this study was to review current literature and ongoing trials to identify interventions employed to improve adherence to adjuvant endocrine therapy (AET) in breast cancer survivors.
Methods
We searched PubMed and the National Library of Medicine registry of clinical trials using the terms “breast cancer” and “adherence” or “compliance” and “intervention” and “medication” or “endocrine therapy” or “hormone therapy” to identify published studies as well as ongoing clinical trials.
Results
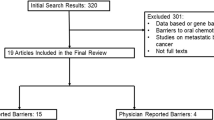
Three hundred and sixty-three studies were identified; five studies met the inclusion criteria. Most studies enrolled postmenopausal women diagnosed with early stage HR+ breast cancer. Providing educational materials was the most common intervention implemented to improve adherence to one or more aromatase inhibitors. None of the studies found a significant improvement in adherence with the intervention evaluated. Twelve clinical trials investigating various interventions, mostly based on technology, to improve AET adherence were also identified.
Conclusions
Improving adherence to AET in HR+ breast cancer survivors is an urgent medical need. While newer clinical trials are overcoming some of the limitations seen with published studies, tailored interventions led by clinicians need further investigation.
Implications for cancer survivors
Our study highlights the unmet clinical need to develop and test feasible interventions to improve AET adherence in HR+ breast cancer survivors to extend their long-term survival.


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017;67(1):7–30. https://doi.org/10.3322/caac.21387.
Giuliano M, Schifp R, Osborne CK, Trivedi MV. Biological mechanisms and clinical implications of endocrine resistance in breast cancer. Breast. 2011;20(Suppl 3):S42–9. https://doi.org/10.1016/S0960-9776(11)70293-4.
Davies C, Pan H, Godwin J, Gray R, Arriagada R, Raina V, et al. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet. 2013;381(9869):805–16. https://doi.org/10.1016/S0140-6736(12)61963-1.
Goss PE, Ingle JN, Pritchard KI, Robert NJ, Muss H, Gralow J, et al. Extending aromatase-inhibitor adjuvant therapy to 10 years. N Engl J Med. 2016;375(3):209–19. https://doi.org/10.1056/NEJMoa1604700.
Milata JL, Otte JL, Carpenter JS. Oral endocrine therapy nonadherence, adverse effects, decisional support, and decisional needs in women with breast cancer. Cancer Nurs. 2016:41(1):E9–E18. https://doi.org/10.1097/NCC.0000000000000430.
Murphy CC, Bartholomew LK, Carpentier MY, Bluethmann SM, Vernon SW. Adherence to adjuvant hormonal therapy among breast cancer survivors in clinical practice: a systematic review. Breast Cancer Res Treat. 2012;134(2):459–78. https://doi.org/10.1007/s10549-012-2114-5.
Cahir C, Dombrowski SU, Kelly CM, Kennedy MJ, Bennett K, Sharp L. Women’s experiences of hormonal therapy for breast cancer: exploring influences on medication-taking behaviour. Support Care Cancer. 2015;23(11):3115–30. https://doi.org/10.1007/s00520-015-2685-x.
Farias AJ, XL D. Association between out-of-pocket costs, race/ethnicity, and adjuvant endocrine therapy adherence among Medicare patients with breast cancer. J Clin Oncol. 2017;35(1):86–95. https://doi.org/10.1200/JCO.2016.68.2807.
Garreau JR, Delamelena T, Walts D, Karamlou K, Johnson N. Side effects of aromatase inhibitors versus tamoxifen: the patients’ perspective. Am J Surg. 2006;192(4):496–8. https://doi.org/10.1016/j.amjsurg.2006.06.018.
Hershman DL, Kushi LH, Shao T, Buono D, Kershenbaum A, Tsai WY, et al. Early discontinuation and nonadherence to adjuvant hormonal therapy in a cohort of 8,769 early-stage breast cancer patients. J Clin Oncol. 2010;28(27):4120–8. https://doi.org/10.1200/JCO.2009.25.9655.
Lin C, Clark R, Tu P, Bosworth HB, Zullig LL. Breast cancer oral anti-cancer medication adherence: a systematic review of psychosocial motivators and barriers. Breast Cancer Res Treat. 2017;165(2):247–60. https://doi.org/10.1007/s10549-017-4317-2.
Simon R, Latreille J, Matte C, Desjardins P, Bergeron E. Adherence to adjuvant endocrine therapy in estrogen receptor-positive breast cancer patients with regular follow-up. Can J Surg. 2014;57(1):26–32. https://doi.org/10.1503/cjs.006211.
Hadji P, Blettner M, Harbeck N, Jackisch C, Luck HJ, Windemuth-Kieselbach C, et al. The Patient’s anastrozole compliance to therapy (PACT) program: a randomized, in-practice study on the impact of a standardized information program on persistence and compliance to adjuvant endocrine therapy in postmenopausal women with early breast cancer. Ann Oncol. 2013;24(6):1505–12. https://doi.org/10.1093/annonc/mds653.
Markopoulos C, Neven P, Tanner M, Marty M, Kreienberg R, Atkins L, et al. Does patient education work in breast cancer? Final results from the global cariatide study. Future Oncol. 2015;11(2):205–17. https://doi.org/10.2217/fon.14.179.
KD Y, Zhou Y, Liu GY, Li B, He PQ, Zhang HW, et al. A prospective, multicenter, controlled, observational study to evaluate the efficacy of a patient support program in improving patients’ persistence to adjuvant aromatase inhibitor medication for postmenopausal, early stage breast cancer. Breast Cancer Res Treat. 2012;134(1):307–13. https://doi.org/10.1007/s10549-012-2059-8.
Ziller V, Kyvernitakis I, Knoll D, Storch A, Hars O, Hadji P. Influence of a patient information program on adherence and persistence with an aromatase inhibitor in breast cancer treatment—the COMPAS study. BMC Cancer. 2013;13(1):407. https://doi.org/10.1186/1471-2407-13-407.
Wagner VL, Jing W, Boscoe FP, Schymura MJ, Roohan PJ, Gesten FC. Improving adjuvant hormone therapy use in Medicaid managed care-insured women, New York State, 2012–2014. Prev Chronic Dis. 2016;13:E120. https://doi.org/10.5888/pcd13.160185.
Neven P, Markopoulos C, Tanner M, Marty M, Kreienberg R, Atkins L, et al. The impact of educational materials on compliance and persistence rates with adjuvant aromatase inhibitor treatment: first-year results from the compliance of aromatase inhibitors assessment in daily practice through educational approach (CARIATIDE) study. Breast. 2014;23(4):393–9. https://doi.org/10.1016/j.breast.2014.02.009.
Nogaret J-M, Coibion M, Neven P, Soepenberg O, Graas M-P, Deschamp V, (2011) Evaluating the impact of educational material on anastrozole treatment adherence—the final results of the artemis study. Cancer Res 71 (24 Suppl):Abstract nr P3–08-02. https://doi.org/10.1016/S0960-9776(11)70233-8
Hershman DL, Shao T, Kushi LH, Buono D, Tsai WY, Fehrenbacher L, et al. Early discontinuation and non-adherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer. Breast Cancer Res Treat. 2011;126(2):529–37. https://doi.org/10.1007/s10549-010-1132-4.
Huiart L, Ferdynus C, Giorgi R. A meta-regression analysis of the available data on adherence to adjuvant hormonal therapy in breast cancer: summarizing the data for clinicians. Breast Cancer Res Treat. 2013;138(1):325–8. https://doi.org/10.1007/s10549-013-2422-4.
Cuzick J. Statistical controversies in clinical research: long-term follow-up of clinical trials in cancer. Ann Oncol. 2015;26(12):2363–6. https://doi.org/10.1093/annonc/mdv392.
Kesmodel SB, Goloubeva OG, Rosenblatt PY, Heiss B, Bellavance EC, Chumsri S, et al. Patient-reported adherence to adjuvant aromatase inhibitor therapy using the morisky medication adherence scale: an evaluation of predictors. Am J Clin Oncol. 2016:1. https://doi.org/10.1097/COC.0000000000000314.
Liu Y, Malin JL, Diamant AL, Thind A, Maly RC. Adherence to adjuvant hormone therapy in low-income women with breast cancer: the role of provider-patient communication. Breast Cancer Res Treat. 2013;137(3):829–36. https://doi.org/10.1007/s10549-012-2387-8.
Chisholm-Burns MA, Kim Lee J, Spivey CA, Slack M, Herrier RN, Hall-Lipsy E, et al. US pharmacists’ effect as team members on patient care: systematic review and meta-analyses. Med Care. 2010;48(10):923–33. https://doi.org/10.1097/MLR.0b013e3181e57962.
Abughosh S, Wang X, Serna O, Esse T, Mann A, Masilamani S, et al. A motivational interviewing intervention by pharmacy students to improve medication adherence. J Manag Care Spec Pharm. 2017;23(5):549–60. https://doi.org/10.18553/jmcp.2017.23.5.549.
Albert US, Zemlin C, Hadji P, Ziller V, Kuhler B, Frank-Hahn B, et al. The impact of breast care nurses on Patients’ satisfaction, understanding of the disease, and adherence to adjuvant endocrine therapy. Breast Care (Basel). 2011;6(3):221–6. https://doi.org/10.1159/000329006.
Oberguggenberger AS, Sztankay M, Beer B, Schubert B, Meraner V, Oberacher H, et al. Adherence evaluation of endocrine treatment in breast cancer: methodological aspects. BMC Cancer. 2012;12(1):474. https://doi.org/10.1186/1471-2407-12-474.
Clarke Hillyer G, Neugut AI, Crew KD, Kalinsky K, Maurer MA, Rotsides DZ, et al. Use of a urine anastrozole assay to determine treatment discontinuation among women with hormone-sensitive breast cancer: a pilot study. J Oncol Pract. 2012;8(5):e100–4. https://doi.org/10.1200/JOP.2011.000487.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Ekinci, E., Nathoo, S., Korattyil, T. et al. Interventions to improve endocrine therapy adherence in breast cancer survivors: what is the evidence?. J Cancer Surviv 12, 348–356 (2018). https://doi.org/10.1007/s11764-017-0674-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-017-0674-4