Abstract
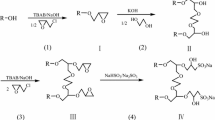
A series of alkyl sulphobetaine Gemini surfactants C n -GSBS (n = 8, 10, 12, 14, 16) was synthesized, using aliphatic amine, cyanuric chloride, ethylenediamine, N,N′-dimethyl-1,3-propyldiamine and sodium 2-chloroethane sulfonate as main raw materials. The chemical structures were confirmed by FT-IR, 1H NMR and elemental analysis. The Krafft points differ markedly with different carbon chain length, for C8-GSBS, C10-GSBS and C12-GSBS are considered to be below 0 °C and C14-GSBS, C16-GSBS are higher than 0 °C but lower than room temperature. Surface-active properties were studied by surface tension and electrical conductivity. Critical micelle concentrations were much lower than dodecyl sulphobetaine (BS-12) and decreased with increasing length of the carbon chain from 8 to 16, and can reach a minimum as low as 5 × 10−5 mol L−1 for C16-GSBS. Effects of carbon chain length and concentration of C n -GSBS on crude oil emulsion stability were also investigated and discussed.




Similar content being viewed by others
References
Rosen MJ, Kunjappu JT. Surfactants and interfacial phenomena. 4th ed. Hoboken: John; 2012.
Xu Q, Wang L, Xing F. Synthesis and properties of dissymmetric Gemini surfactants. J Surfactants Deterg. 2011;14(1):85–90.
Murguía MC, Cristaldi MD, Porto A, Di Conza J, Grau RJ. Synthesis, surface-active properties, and antimicrobial activities of new neutral and cationic trimeric surfactants. J Surfactants Deterg. 2008;11(1):41–8.
Zhou M, Zhao J, Hu X. Synthesis of Bis [N, N′-(alkylamideethyl) ethyl] triethylenediamine bromide surfactants and their oilfield application investigation. J Surfactants Deterg. 2012;15(3):309–15.
Dharaiya N, Patriati A, Kuperkar K, Putra EGR, Bahadur P. Spectral and scattering microstructural investigation in cationic gemini surfactants(12-s-12) induced by p-tolulidine. Colloids Surf A. 2012;396(7):1–7.
Zana R, Xia J. Gemini surfactants: synthesis, interfacial and solution-phase behavior, and applications. New York: Marcel Dekker; 2004.
Hujun X, Hui G, Peng K, Dandan C. Synthesis and properties of a novel liner alkylated diphenylmethane sulfonate Gemini surfactant. J Surfactants Deterg. 2013;16:57–61.
Chen H, Zhu B. A new anionic oxalamide lauryl succinate sodium sulfonate gemini surfactant: microwave-assisted synthesis and surface activities. J Surfactants Deterg. 2014;17:937–42.
Zhu YP, Masuyama A, Okahara M. Preparation and surface-active properties of amphipathic compounds with two sulfate groups and two lipophilic alkyl chains. J Am Oil Chem Soc. 1990;67:459–63.
Yoshimura T, Esumi K. Synthesis and surface properties of anionic gemini surfactants with amide groups. J Colloid Interface Sci. 2004;276:231–8.
Du X, Lu Y, Li L, Wang J, Yang Z. Synthesis and unusual properties of novel alkylbenzene sulfonate gemini surfactants. Colloids Surf A. 2007;290:132–7.
Li X, Hu ZY, Zhao HL, Zhao SF, Cao DL. Synthesis and properties of novel alkyl sulfonate gemini surfactants. J. Surfactants Deterg. 2010;13:353–9. doi:10.1007/s11743-010-1188-5.
Qiao W, Peng H, Zhu Y, Cai H. Synthesis and surface activity properties of symmetric double chains alkylbetaine surfactants derived from s-triazine. Colloids Surf A. 2012;405:45–50. doi:10.1016/j.colsurfa.2012.04.034.
Xie KL, Sun Y, Hou AQ. Diffusion properties of reactive dyes into net modified cotton cellulose with triazine derivative. J Appl Polym Sci. 2007;103:2166–71. doi:10.1002/app.25097.
Xue Z, Hu Z, Zhu H, Chen L. Synthesis and surface activity properties of cleavable s-triazine fatty sulfonate surfactants. China Surfactant Detergent Cosmet. 2008;3:151–5.
Petrova-Meladinova PM, Konstantinova TN. On the synthesis of some reactive triazine azo dyes containing tetramethylpiperidine fragment. Dyes Pigm. 2005;67:63–9.
Konstantionova TN, Petrova P. On the synthesis of some bifunctional reactive triazine dyes. Dyes Pigm. 2002;52:115–20.
Zana R, Lévy H, Danino D, Talmon Y, Kwetkat K. Mixed micellization of cetyltrimethyl ammonium bromide and an anionic dimeric (Gemini) surfactant in aqueous solution. Langmuir. 1997;13:402–8.
Van Biesen G, Bottaro CS. Linear solvation energy relationships of anionic dimeric surfactants in micellar electrokinetic chromatography I. Effect of the length of a hydrophobic spacer. J Chromatogr A. 2007;1157:437–45.
Van Biesen G, Bottaro CS. Liner solvation energy relationships of anionic dimeric surfactants in micellar electrokinetic chromatography II. Effect of the length of a hydrophilic spacer. J Chromatogr A. 2008;1180:171–8.
Tai S, Gao Z, Liu X, Zhang Q. Synthesis and properties of novel alkyl sulfate Gemini surfactants. Eur J Lipid Sci Technol. 2012;114:1062–9.
Wang Q, Zhang SX, Geng B, Zhang L, Zhao J, Shi J. Synthesis and surface activities of novel monofluoroalkyl phosphate surfactants. J Surfactants Deterg. 2012;15:83–8.
Chen L, Hu Z, Zhu H, Xue Z. Synthesis of cleavable aryl sulfonate anionic surfactants and a study of their surface activity. J Surfactants Deterg. 2008;11(2):97–102.
Yoshimura T, Sakato A, TsuchiyaK Ohkubo T, Sakai H, Abe M, Esumi K. Adsorption and aggregation properties of amino acid-based N-alkyl cysteine monomeric and N,N′-dialkyl cystine gemini surfactants. J Colloid Interface Sci. 2007;308(2):466–73.
Chai M, Zheng Z, Bao L, Qiao W. CO2/N2 triggered switchable surfactants with imidazole group. J Surfactants Deterg. 2014;17(3):383–90.
Huber K. Interactions in mixed interfaces of binary surfactant solutions. J Colloid Interface Sci. 1991;147(2):321–32.
Chauhan V, Singh S, Bhadani A. Synthesis, characterization and surface properties of long chain β-hydroxy-γ-alkyloxy-N-methylimidazolium surfactants. Colloids Surf A. 2012;395(395):1–9.
Li XW, Gao YA, Liu J, et al. Aggregation behavior of a chiral long-chain ionic liquid in aqueous solution. J Colloid Interface Sci. 2010;343(1):94–101.
MiZhou M, Zhong X, Zhao J, Wang X. Synthesis and surface active properties of 1,1,1,1-tetra-(2-oxypropyl sulfonate-3-alkylether -propoxy) neopentanes. J Surfactants Deterg. 2013;16(3):285–90.
Li P, Chen Q, Zhao J, Wang H, Li C, Hu Z, Cao D, et al. Synthesis and properties of X-type alkyl sulfonate gemini surfactants derived from cyanuric chloride. J Surfactants Deterg. 2011;15(4):449–56.
Cui ZG. Fundamentals of surfactants, colloids, and interface chemistry. 1st ed. Beijing: Chemical Industry Press; 2013.
Rosen MJ. Surfactants and interfacial phenomena. 2nd ed. New York: Wiley; 1989.
Fu Z, Liu M, Xu J, Wang Q, Fan Z. Stabilization of water-in-octane nano-emulsion. Part I. Stabilized by mixed surfactant systems. Fuel. 2010;89(10):2838–43.
Kang L, Xu B, Wang Y, Li Y, Shan X, An F, Liu J. Stability mechanism of W/O crude oil emulsion stabilized by polymer and surfactant. Colloids Surf A Physicochem Eng Asp. 2011;384(1–3):555–60.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Niu, R., Wang, C., Sun, Z. et al. A Novel Alkyl Sulphobetaine Gemini Surfactant Based on S-triazine: Synthesis and Properties. J Surfact Deterg 20, 1255–1262 (2017). https://doi.org/10.1007/s11743-017-1996-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11743-017-1996-y