Abstract
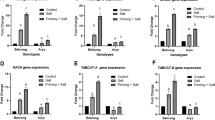
Though seed priming has been emerged as an effective and pragmatic approach, efforts are being made to discover and optimize the new priming agents which are cheaper and easily accessible to the farmers. Here, we established two independent experiments, to ascertain the role of aspirin priming in salinity tolerance of wheat. In the first experiment, various concentrations of aspirin (125, 250, 375, and 500 ppm) were examined for emergence and seedling growth of wheat. A non-primed control, hydropriming, and hydrogen peroxide priming treatments were also maintained for comparison. Among the different treatments, seeds primed with 125 and 250 ppm aspirin depicted better emergence, vigorous seedling growth, and higher starch metabolism. Therefore, these treatments were further used in the second experiment under salinity stress (10 dS m−1). Salinity stress caused delayed and erratic emergence hampered the shoot and root growth, chlorophyll contents, and enhanced the lipid peroxidation and phenolics content in wheat seedlings. However, wheat seed priming particularly with aspirin effectively alleviated the negative effects of salinity on most of the observed parameters. Aspirin priming also significantly enhanced the activities of antioxidant enzymes (catalase, peroxidase, and superoxide dismutase), and reduced oxidative stress in wheat seedlings. Vigorous growth and greater salinity tolerance of wheat seedlings derived from aspirin primed seeds were related with better starch metabolism, strong antioxidative defense system, and lower lipid peroxidation.







Similar content being viewed by others
References
Abdul-Baki BAA, Anderson JP (1973) Relationship between decarboxylation of glutamic acid and vigor in soybean seed. Crop Sci 13:227–234
Afzal I, Basra SMA, Farooq M, Nawaz A (2006) Alleviation of salinity stress in spring wheat by hormonal priming with ABA, salicylic acid and ascorbic acid. Int J Agric Biol 8:23–28
Ahanger MA, Tyagi SR, Wani MR, Ahmad P (2014) Drought tolerance: role of organic osmolytes, growth regulators, and mineral nutrients. Physiological mechanisms and adaptation strategies in plants under changing environment. Springer, New York, pp 25–55
Anjum SA, Tanveer M, Hussain S, Bao M, Wang L, Khan I, Ehsanullah Tung SA, Samad RA, Shahzad B (2015a) Cadmium toxicity in maize (Zea mays L.): consequences on antioxidative systems, reactive oxygen species and cadmium accumulation. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-015-4882-z
Anjum SA, Tanveer M, Hussain S, Ullah E, Wang LC, Khan I, Samad RA, Tung SA, Anam M, Shahzad B (2015b) Morpho-physiological responses of two contrasting maize cultivars to cadmium toxicity. Clean Soil Air Water. https://doi.org/10.1002/clen.201400905
Anjum SA, Ashraf U, Tanveer M, Khan I, Hussain S, Zohaib A, Abbas F, Saleem MF, Wang L (2017) Drought induced changes in growth, osmolyte accumulation and antioxidant metabolism of three maize hybrids. Front Plant Sci 8:69. https://doi.org/10.3389/fpls.2017.00069
AOSA (1983) Seed vigor hand testing book. Contribution No. 32 to the handbook on seed testing. Association of Official Seed Analysts (AOSA), Springfield
AOSA (1990) Rules for testing seeds. Association of Official Seed Analysts (AOSA). J Seed Technol 12:1–112
Bailly C, Benamar A, Corbineau F, Dome D (1996) Changes in malondialdehyde contents and in superoxide dismutase, catalase, glutathione reductase activities in sunflower seeds related to accelerated seed aging. Physiol Plant 97:104–110
Bradford KJ (1986) Manipulation of seed water relations via osmotic priming to improve germination under stress conditions. Hortic Sci 21:1105–1112
Chen W, Guo C, Hussain S, Zhu B, Deng F, Xue Y, Geng M, Wu L (2016) Role of xylo-oligosaccharides in protection against salinity-induced adversities in Chinese cabbage. Environ Sci Pollut Res 23:1254–1264
Dhindsa RS, Plumb-Dhindsa P, Thorpe TA (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J Exp Bot 32:93–101
Dubios M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugar and related substances. Anal Chem 28:350–356
Egley GH, Paul RN, Vaughn KC, Duke SO (1983) Role of peroxidase in the development of water impermeable seed coats in Sida spinosa L. Planta 157:224–232
Ellis RA, Roberts EH (1981) The quantification of ageing and survival in orthodox seeds. Seed Sci Technol 9:373–409
Fahad S, Hussain S, Matloob A, Khan FA, Khaliq A, Saud S et al (2014) Phytohormones and plant responses to salinity stress: a review. Plant Growth Regul 75:391–404
Farooq M, Basra SMA, Hafeez K, Ahmad N (2005) Thermal hardening: a new seed vigor enhancing tool in rice. J Integr Plant Biol 47:187–193
Farooq M, Basra SMA, Hafeez K (2006) Seed invigoration by osmohardening in coarse and fine rice. Seed Sci Technol 34:181–187
Giannopolitis CN, Ries SK (1977) Superoxide dismutase: I. occurrence in higher plants. Plant Physiol 59:309–314
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Hayat Q, Hayat S, Irfan M, Ahmad A (2010) Effect of exogenous salicylic acid under changing environment: a review. Environ Exp Bot 68:14–25
Hussain S, Khaliq A, Matloob A, Wahid MA, Afzal I (2013) Germination and growth response of three wheat cultivars to NaCl salinity. Soil Environ 32:36–43
Hussain S, Khaliq A, Matloob A, Fahad S, Tanveer A (2015a) Interference and economic threshold level of little seed canary grass in wheat under different sowing times. Environ Sci Pollut Res 22:441–449
Hussain S, Zheng M, Khan F, Khaliq A, Fahad S, Peng S, Huang J, Cui K, Nie L (2015b) Benefits of rice seed priming are offset permanently by prolonged storage and the storage conditions. Sci Rep 5:8101. https://doi.org/10.1038/srep08101
Hussain S, Khan F, Cao W, Wu L, Geng M (2016) Seed priming alters the production and detoxification of reactive oxygen intermediates in rice seedlings grown under sub-optimal temperature and nutrient supply. Front Plant Sci 7:439. https://doi.org/10.3389/fpls.2016.00439
Iqbal M, Ashraf M (2007) Seed treatment with auxins modulates growth and ion partitioning in salt-stressed wheat plants. J Integr Plant Biol 49:1003–1015
Jisha KC, Vijayakumari K, Puthur JT (2013) Seed priming for abiotic stress tolerance: an overview. Acta Physiol Plant 35:1381–1396
Khaliq A, Zia-ul-Haq M, Ali F, Aslam F, Matloob A, Navab A, Hussain S (2015) Salinity tolerance in wheat cultivars is related to enhanced activities of enzymatic antioxidants and reduced lipid peroxidation. Clean Soil Air Water 43:1248–1258
Khan HA, Pervez MA, Ayub CM, Ziaf K, Balal RM, Shahid MA, Akhtar N (2009) Hormonal priming alleviates salt stress in hot pepper (Capsicum annuum L.). Soil Environ 28:130–135
Lai K, Berkman PJ, Lorenc MT, Duran C, Smits L, Manoli S et al (2012) Wheat genome.info: an integrated database and portal for wheat genome information. Plant Cell Physiol 53:e2
Lee SS, Kim JH (2000) Total sugars, α-amylase activity, and emergence after priming of normal and aged rice seeds. Korean Crop Sci 45:108–111
Lichtenthaler HK (1987) Chlorophyll and carotenoids: pigments of photosynthetic bio-membranes. In: Packer L, Douce R (eds) Methods in enzymology. Academic Press, San Diego, pp 350–382
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681
Munns R, James RA, Lauchli A (2006) Approaches to increasing the salt tolerance of wheat and other cereals. J Exp Bot 57:1025–1043
Mustafa Z, Pervez MA, Ayyub CM, Matloob A, Khaliq A, Hussain S, Ihsan MZ, Butt M (2014) Morpho-physiological characterization of chilli genotypes under NaCl salinity. Soil Environ 33:133–141
Paparella S, Araújo SS, Rossi G, Wijayasinghe M, Carbonera D, Balestrazzi A (2015) Seed priming: state of the art and new perspectives. Plant Cell Rep. https://doi.org/10.1007/s00299-015-1784-y
Randhir R, Shetty K (2005) Developmental stimulation of total phenolics and related antioxidant activity in light and dark germinated maize by natural elicitors. Proc Biochem 40:1721–1732
Ruan S, Xue Q, Tylkowska K (2002) The influence of priming on germination of rice (Oryza sativa L.) seeds and seedling emergence and performance in flooded soils. Seed Sci Technol 30:61–67
Szalai G, Janda T (2009) Effect of salt stress on the salicylic acid synthesis in young maize (Zea mays L.) plants. J Agron Crop Sci 195:165–171
Tanveer M, Shah AN (2017) An insight into salt stress tolerance mechanisms of Chenopodium album. Environ Sci Pollut Res 24:16531–16535
Zhang S, Hu J, Zhang Y, Xie XJ, Knapp A (2007) Seed priming with brassinolide improves lucerne (Medicago sativa L.) seed germination and seedling growth in relation to physiological changes under salinity stress. Aust J Agric Res 58:811–815
Zheng M, Tao Y, Hussain S, Jiang Q, Peng S, Huang J, Cui K, Nie L (2016) Seed priming in dry direct-seeded rice: consequences for emergence, seedling growth and associated metabolic events under drought stress. Plant Growth Regul 78:167–178
Zhu JK (2001) Plant salt tolerance. Trends Plant Sci 6:66–71
Zhu JK (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Physiol 53:247–273
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by S. Weidner.
Rights and permissions
About this article
Cite this article
Hussain, S., Khaliq, A., Tanveer, M. et al. Aspirin priming circumvents the salinity-induced effects on wheat emergence and seedling growth by regulating starch metabolism and antioxidant enzyme activities. Acta Physiol Plant 40, 68 (2018). https://doi.org/10.1007/s11738-018-2644-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-018-2644-5