Abstract
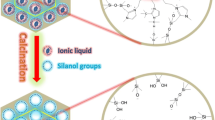
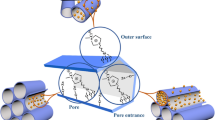
The long-chain ionic liquid 1-hexadecyl-3-methylimidazolium chloride (C16mimCl) was used as a template to prepare cerium-doped MCM-48 materials in basic medium by a hydrothermal synthesis procedure. The effect of the amount of Ce salt and C16mimCl/Si on the synthesis were discussed in detail. This mesoporous material exhibits a pore architecture which is cubic Ia3d gyroid and possesses a large surface area and a narrow pore distribution. Cerium in calcined porous framework exists in the form of well-dispersed tetrahedral coordination. The C16mimCl shows a high tendency toward self-aggregation that allows the formation of the cerium-doped gyroid mesostructure by using the appropriate amount of Ce salt.
Similar content being viewed by others
References
Laha S C, Mukherjee P, Sainkar S R, Kumar R. Cerium containing MCM-41-type mesoporous materials and their acidic and redox catalytic properties. J Catal, 2002, 207: 213–223
Kadgaonkar MD, Laha S C, Pandey RK, Kumar P, Mirajkar S P, Kumar R. Cerium-containing MCM-41 materials as selective acylation and alkylation catalysts. Catal Today, 2004, 97: 225–231
Yao W H, Chen Y J, Min L, Fang H, Yan Z Y, Wang H L, Wang J Q. Liquid oxidation of cyclohexane to cyclohexanol over cerium-doped MCM-41. J Mol Catal A: Chem, 2006, 246: 162–166
Shao Y F, Wang L Z, Zhang J L, Anpo M. Synthesis of hydrothermally stable and long-range ordered Ce-MCM-48 and Fe-MCM-48 materials. J Phys Chem B, 2005, 109(44): 20835–20841
Jin Z X, Tong HW, Yong G P, Sheng L Q, Liu Q L, Liu SM. Synthesis, characterization and application of Ce-MCM-48 as a mesoporous molecular Sieve. Chin J Chem Phys, 2005, 12(6): 1057–1061 (in Chinese)
Welton T. Room-temperature ionic liquids: solvents for synthesis and catalysis. Chem Rev, 1999, 99: 2071–2083
Seddon K R. Ionic liquids: a taste of the future. Nat Mater, 2003, 2: 363–365
Bleasdale T A, Tiddy G J T, Wyn-Jones E. Cubic phase formation in polar nonaqueous solvents. J Phys Chem, 1991, 95(14): 5385–5386
Neve F, Francescangeli O, Crispini A. Crystal architecture and mesophase structure of long-chain N-alkylpyridinium tetrachlorometallates. Inorg Chim Acta, 2002, 338: 51–58
Lee C K, Huang H W, Lin I J B. Simple amphiphilic liquid crystalline N-alkylimidazolium salts. A new solvent system providing a partially ordered environment. Chem Commun, 2000, 1911–1912
Antonietti M, Kuang D B, Smarsly B, Zhou Y. Ionic liquids for the convenient synthesis of functional nanoparticles and other inorganic nanostructures. Angew Chem Int Ed, 2004, 43: 4988–4992
Zhou Y, Antonietti M. Preparation of highly ordered monolithic super-microporous lamellar silica with a room-temperature ionic liquid as template via the nanocasting technique. Adv Mater, 2003, 15: 1452–1455
Zhou Y, Antonietti M. A series of highly ordered, supermicroporus, lamellar silicas prepared by nanocasting with ionic liquids. Chem Mater, 2004, 16: 544–550
Wang T W, Kaper H, Antonietti M, Smarsly B. Templating behavior of a long-chain ionic liquid in the hydrothermal synthesis of mesoporous silica. Langmuir, 2007, 23: 1489–1495
Seddon K R, Stark A, Torres MJ. Influence of chloride water and organic solvents on the physical properties of ionic liquids. Pure Appl Chem 2000, 72: 2275–2287
Kresge C T, Leonowicz ME, Roth WJ, Vartuli J C, Beck J S. Ordered mesoporous molecular sieves synthesized by a liquidcrystal template mechanism. Nature, 1992, 359: 710–712
Huo Q, Margolese D I, Stucky G D. Surfactant control of phases in the synthesis of mesoporous silca-based materials. Chem Mater, 1996, 8: 1147–1160
Alfredsson V, Anderson MW. Structure of MCM-48 revealed by transmission electron microscopy. Chem Mater, 1996, 8: 1141–1146
Morey MS, Brien S O, Schwarz S, Stucky G D. Hydrothermal and postsynthesis surface modification of Cubic, MCM-48, and ultralarge pore SBA-15 mesoporous silica with titanium. Chem Mater, 2000, 12: 898–911
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yue, H., Zhao, H., Liu, L. et al. Synthesis of ordered cerium-doped cubic mesoporous silica using long-chain ionic liquid as template. Front. Chem. Eng. China 2, 135–139 (2008). https://doi.org/10.1007/s11705-008-0031-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11705-008-0031-2