Abstract
The opening of epoxy rings from (2′Z)-N-1-(oxiran-2-ylmethyl)indirubin (2) and (2′Z-3′E)-indirubin-3ʹ-[O-oxiran-2-ylmethyl)oxime] (6) with thiols gave 17 new derivatives of indirubin in good yields. Their structures were elucidated by 1D-, 2D-NMR and HRMS spectra. Screening for anticancer activity was performed with four human cancer cell lines: SW480, LU-1, HepG2 and HL-60 in comparison with indirubin, indirubin-3′-oxime and 6-mercaptopurine. The results showed that cytotoxic and anti-proliferative activities of five derivatives were found in the range of 1.35–19.24 µM. Among synthesized derivatives, 4f showed the strongest activity against all four tested cancer cell lines with IC50 values of 1.65, 2.21, 1.90 and 1.35 µM, respectively.
Similar content being viewed by others
Introduction
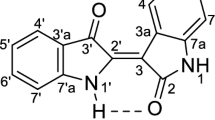
The well-known indole skeleton dyes including indigo, isoindigo and (2′Z)-indirubin can be early found in a number of plants (Cuong et al. 2010a, b, c; Bektas et al. 2016), and indigo, indirubin and their derivatives are the constituents of a preparation called indigo naturalis (Stasiak et al. 2014). In addition, indirubin was determined to be a main active ingredient of a traditional Chinese medicinal recipe including Indigofera tinctoria L. and Isatis tinctoria L, which were used for treatment of chronic myelocytic leukemia (CML) for decades (Xiao et al. 2002). Indirubin (1) could be easily synthesized from istatin and 3-acetoxyindole in alkaline medium (Riepl and Urmann 2012), and it was also reported to inhibit several kinases such as glycogen synthase kinase-3 (GSK-3) and cyclin-dependent kinases (CDKs) by interaction with the ATP-binding site of kinase through van der Waals interaction and hydrogen bonds, and induce apoptosis in human cancer cells (Meijer et al. 2003; Hoessel et al. 1999; Jung et al. 2016; Nam et al. 2005; Lectere et al. 2001). In 2011, Karapetyan reported the synthesis of some indirubin-N-glycosides and their activity against several human cancer cell lines such as A-427, Kyse-70 and breast MCF-7. Among these glycosides, the activity of indirubin-α-N-rhamnoside and indirubin-β-N-rhamnoside against the human breast cancer cell line was much higher (10- to 100 fold) than that of the non-glycosylated indirubins tested before (Karapetyan et al. 2011). Indirubin-3′-oxime, an indirubin derivative was considered as potential candidate to inhibit the growth of lymphocytes in leukemia patients. This compound also reduced CDK activity and gene expression (Girard et al. 2007), and indirubin-3′-oxime was found to causes arrest at G0/G1 phase accompanied by a significant decrease in the percentage of cell in S and G2/M phases in cell cycle (Liao and Leung 2013). Lo et al. also recognized that indirubin-3-oxime had anti-proliferative activity and apoptosis in both Ca27 and SHC-3 oral cancer cell lines though the activation of cytochrome C (Lo and Chang 2013). Additionally, indirubin-3′-oxime suppressed migration and invasion in Ca27 by inhibiting the expression of local adhesion kinase, urokinase-type plasminogen inhibitor and matrix metalloproteinase 9 (Lo and Chang 2013). In 2014, the activation of Wnt/b-catenin signaling and the inhibition of adipocyte differentiation and obesity were also reported (Choi et al. 2014). Recently, indirubin-3′-oxime derivatives have also attracted the interest of chemists due to their anticancer activity. Notably, a report of Cuong and colleagues demonstrated that (2′Z-3′E)-indirubin-3′-[O-(3-bromoprop-1-yl)-oxime] and (2′Z-3′E)-indirubin-3′-[O-(3-methoxycarrbonylmethyl)-oxime] revealed stimulative effects on MC3T3-E1 osteoblastic cell growth and differentiation (Cuong et al. 2010a, b, c). Another derivative of indirubin, 5-methoxyindirubin 3′-oxime also expressed the induction of cell death in pancreatic ductal adenocarcinoma both in vitro and in vivo (Sano et al. 2017).

In a search for new agents for cytotoxic effects, we suggest that exploring new indirubin conjugates containing nitrogen and sulfur heterocycles could achieve potential compounds for pharmacological purpose. Therefore, in present study, two series of conjugates from indirubin were designed, synthesized and evaluated for in vitro anticancer activity.
Experimental
All chemicals were purchased from Sigma-Aldrich (USA), Santa cruz (USA) except for 3a, 3b, 3c, 3d and 3f were prepared according to the references (Raju et al. 2015; Gilani et al. 2011; Vaughan 1957). Indirubin with (2′Z) conformation was isolated from Strobilanthes cusia in Vietnam as described by Cuong et al. (2007). Dry solvents were prepared according to procedures of Perrin and Armarego (1988). Melting points were measured in open capillary tubes on a Buchi 530 (Switzerland) melting point apparatus and were uncorrected. NMR spectra were recorded on a Bruker Advance 500 MHz operating at 500 MHz for 1H and 125 MHz for 13C. Chemical shifts are reported in parts per million (ppm) using tetramethylsilane (TMS) as internal standard. Mass spectra were recorded on an Agilent 6530 Accurate-Mas Q-TOG LC/MS. Progress of the reaction was monitored by thin-layer chromatography (TLC) using precoated TLC sheets (Merck 60F254), and spots were visualized by UV lamp at 254 nm. Multiplicities are shown as follows: s (singlet), d (doublet), t (triplet), m (multiplet). Column chromatography was carried out using silica gel 60 (0.04–0.06 mm) from Scharlau, Spain. Solvents were commercially available materials of reagent grade.
Synthesis of ( 2 ′Z)-indirubin derivatives 4a-h
Synthesis of (Z)-1ʹ-(oxiran-2-ylmethyl)-[2,3ʹ-biindolinylidene]-2ʹ,3-dione (2). A solution of compound 1 (786 mg, 3 mmol), epichlorohydrin (0.784 mL, 10 mmol), K2CO3 (1242 mg, 9 mmol), KI (50 mg, 0.3 mmol) and (1-butyl)triethylammonium bromide (71.5 mg, 0.3 mmol) in dried DMF (50 mL) was stirred at 45 °C for 4 days. The mixture was poured in ice water (200 ml) and the resulting precipitate was filtered and washed with water. Compound 2 was purified by chromatography on silica gel eluting with CH2Cl2/EtOAc 10:1
(Z)-1ʹ-(Oxiran-2-ylmethyl)-[2,3ʹ-biindolinylidene]-2ʹ,3-dione (2). Yield 40%, dark violet solid, m.p. 178–179 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.11 (s, 1H, H-1ʹ), 8.83 (d, J = 7.50 Hz, 1H, H-4), 7.68 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.61 (m, 1H, H-6ʹ), 7.43 (d, J = 8.0 Hz, 1H, H-7ʹ), 7.35 (m, 1H, H-6), 7.21 (d, J = 8.0 Hz, 1H, H-7), 7.10 (m, 1H, H-5), 7.06 (m, 1H, H-5ʹ), 4.20 (dd, J1 = 15.0 Hz, J2 = 3.75 Hz, 1H, –N1–CH2–), 3.87 (dd, J1 = 15.0 Hz, J2 = 5.25 Hz, 1H, –N1–CH2–), 3.24 (m, 1H, CHepoxy), 2.79 (t, J = 4.50 Hz, 1H, –CH2epoxy), 2.66 (dd, J1 = 5.0 Hz, J2 = 3.0 Hz, 1H, –CH2 epoxy). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 189.0 (C-3ʹ), 169.6 (C-2), 153.0 (C-7ʹa), 141.9 (C-7a), 139.3 (C-2ʹ), 137.7 (C-6ʹ), 129.6 (C-6), 125.0 (C-4), 124.9 (C-4ʹ), 122.4 (C-3a), 121.9 (C-5), 121.1 (C-5ʹ), 119.5 (C-3ʹa), 114.0 (C-7ʹ), 109.6 (C-7), 105.7 (C-3), 49.9 (–CH–epoxy), 45.2 (–CH2–epoxy), 41.6 (–N1–CH2–). HR–ESI–MS: calculated for C19H14N2O3 [M]+: 318.1004, found: 318.0998.
General procedure for the synthesis of ( 2 ′Z)-indirubin derivatives 4a-h
A solution of compound 2 (47.7 mg, 0.15 mmol), triethylamine (0.45 mmol, 63 µl) and corresponding thiols (3a-h) (0.2 mmol) in dried DMF (3 ml) was stirred at room temperature for 40 h. The mixture was then diluted with EtOAc (50 ml) and washed with NaCl 3% solution (3 × 100 ml). The combined EtOAc extract was dried on anhydrous sodium sulfate, and solvent was removed under reduced pressure to afford crudes 4a-h that was chromatographed using chloroform: EtOAc 2:1 as solvent to afford 4a-h in 63.1–77.0.3% yields.
(Z)-1ʹ-(2-Hydroxy-3-((5-phenyl-1,3,4-oxadiazol-2-yl)thio)propyl)-[2,3ʹ-biindolinylidene]-2ʹ,3-dione (4a). Yield 73.2%, dark violet solid, m.p. 222–223 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.1 (s, 1H, H-1ʹ), 8.82 (d, J = 8.0 Hz, 1H, H-4), 7.93 (d, J = 7.0 Hz, 2H, H-2″, H-6″), 7.67 (d, J = 7.0 Hz, 1H, H-4ʹ), 7.58 (m, 4H, H-3″, H-5″, H-6ʹ, H-4″), 7.41 (d, J = 8.0 Hz, 1H, H-7ʹ), 7.33 (t, J = 7.50 Hz, 1H, H-6), 7.2 (d, J = 7.50 Hz, 1H, H-7), 7.09 (t, J = 7.50 Hz, 1H, H-5), 7.04 (t, J = 7.25 Hz, 1H, H-5ʹ), 5.71 (d, J = 5.0 Hz, 1H, –CHOH–), 4.24 (m, 1H, –CHOH–), 3.98 (m, 2H, –N–CH2), 3.58 (dd, J1 = 13.50 Hz, J2 = 3.50 Hz, 1H, –CH2–S–), 3.4 (dd, J1 = 13.50 Hz, J2 = 7.50 Hz, 1H, –CH2–S). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 189.1 (C-3ʹ), 169.9 (C-2), 165.5 (phenyl–Coxadiazole), 164.5 (–S–Coxadiazole), 152.9 (C-7ʹa), 142.3 (C-7a), 139.1 (C-2ʹ), 137.7 (C-6ʹ), 132.4 (C-4″), 129.9 (C-3″, C-5″), 129.5 (C-6), 126.8 (C-2″, C-6″), 124.9 (C-4), 124.8 (C-4ʹ), 123.5 (C-1″), 122.2 (C-3a), 121.9 (C-5), 121.3 (C-5ʹ), 119.5 (C-3ʹa), 113.9 (C-7ʹ), 109.6 (C-7), 106.1 (C-3), 67.5(–CHOH–), 45.2 (–N–CH2–), 37.6(–CH2–S–). HR–ESI–MS: Caculated for C27H20N4O4S: [M]+: 496.1205, found: 496.1195.
(Z)-1ʹ-(2-Hydroxy-3-((5-(4-methoxyphenyl)-1,3,4-oxadiazol-2-yl)thio)propyl)-[2,3ʹ-biindolinylidene]-2ʹ,3-dione (4b). Yield 71.8%, dark violet solid, mp: 225–226 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.09 (s, 1H, H-1ʹ), 8.83 (d, J = 7.5 Hz, 1H, H-4), 7.85 (d, J = 8.5 Hz, 2H, H-2″, H-6″), 7.67 (d, J = 7.5 Hz, 1H, H-4ʹ), 7.59 (t, J = 7.5 Hz, 1H, H-6ʹ), 7.41 (d, J = 8.0 Hz, 1H, H-7ʹ), 7.33 (t, J = 7.75 Hz, 1H, H-6), 7.2 (d, J = 8.0 Hz, 1H, H-7), 7.10 (m, 3H, H-5, H-3″, H-5″), 7.04 (t, J = 7.5 Hz, 1H, H-5ʹ), 5.71 (d, J = 5.0 Hz, 1H, –CHOH–), 4.22 (m, 1H, –CHOH–), 3.97 (m, 2H, –N–CH2–), 3.83 (s, 3H, CH3O–), 3.56 (dd, J1 = 13.5 Hz, J2 = 4.0 Hz, 1H, –CH2–S–), 3.38 (m, 1H,–CH2–S–). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 189.1 (C-3ʹ), 169.9 (C-2), 165.4 (phenyl–Coxadiazole), 163.6 (–S–Coxadiazole), 162.4 (C-4″), 152.9 (C-7ʹa), 142.3 (C-7a), 139.1 (C-2ʹ), 137.7 (C-6ʹ), 129.5 (C-6), 128.6 (C-2″, C-6″), 124.9 (C-4), 124.8 (C-4ʹ), 122.2 (C-3a), 121.9 (C-5), 121.3 (C-5ʹ), 119.5 (C-3ʹa), 115.9 (C-1″), 115.3 (C-3″, C-5″), 113.9 (C-7ʹ), 109.6 (C-7), 106.1 (C-3), 67.6 (–CHOH–), 55.9 (CH3O–), 45.2 (–N–CH2–), 37.6 (–CH2–S–). HR–ESI–MS: Caculated for C28H22N4O5S: [M]+: 526.1311, found: 526.1303.
(Z)-1ʹ-(2-Hydroxy-3-((5-(4-nitrophenyl)-1,3,4-oxadiazol-2-yl)thio)propyl)-[2,3ʹ-biindolinylidene]-2ʹ,3-dione (4c). Yield 66.1%, dark violet solid, mp: 168–169 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.1 (s, 1H, H-1ʹ), 8.82 (d, J = 7.0 Hz, 1H, H-4), 8.36 (dd, J1 = 7.0 Hz, J2 = 2.0 Hz, 2H, H-3″, H-5″); 8.16 (dd, J1 = 7.0 Hz, J2 = 2.0 Hz, 2H, H-2″, H-6″), 7.65 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.58 (m, 1H, H-6ʹ), 7.39 (d, J = 8.0 Hz, 1H, H-7ʹ), 7.33 (m, 1H, H-6), 7.2 (d, J = 7.5 Hz, 1H, H-7), 7.09 (m, 1H, H-5), 7.03 (m, 1H, H-5ʹ), 5.74 (d, J = 5.50 Hz, 1H, –CHOH-), 4.26 (m, 1H, –CHOH–), 4.01 (dd, J1 = 14.50 Hz, J2 = 5.50 Hz, 1H, –N–CH2-), 3.95 (dd, J1 = 14.50 Hz, J2 = 6.50 Hz, 1H, –N–CH2-), 3.61 (dd, J1 = 13.50 Hz, J2 = 4.50 Hz, 1H, –CH2–S–), 3.43 (dd, J1 = 13.50 Hz, J2 = 7.50 Hz, 1H, –CH2–S–). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 189 (C-3ʹ), 169.9(C-2), 165.9 (phenyl–Coxadiazole), 164.1 (–S–Coxadiazole), 152.9 (C-7ʹa), 149.5 (C-4″), 142.3 (C-7a), 139.1 (C-2ʹ), 137.7 (C-6ʹ), 129.5 (C-6), 129 (C-1″), 128.1 (C-2″, C-6″), 125 (C-3″, C-5″), 124.9 (C-4), 124.8 (C-4ʹ), 122.3 (C-3a), 121.9 (C-5), 121.3 (C-5ʹ), 119.5 (C-3ʹa), 113.9 (C-7ʹ), 109.6 (C-7), 106 (C-3), 67.4(–CHOH–), 45.1(–N–CH2-), 37.6(–CH2–S–). HR-ESI–MS: Caculated for C27H19N5O6S: [M]+: 541.1056, found: 541.1047.
( Z )-1ʹ-(2-Hydroxy-3-((5-(pyridin-4-yl)-1,3,4-oxadiazol-2-yl)thio)propyl)-[2,3ʹ-biindolinylidene]-2ʹ,3-dione (4d).
Yield 65.0%, dark violet solid, mp: 183–184 °C. 1H NMR (DMSO-d6, 500 MHz),δδ(ppm): 11.1 (s, 1H, H-1ʹ), 8.82 (d, J = 7.0 Hz, 1H, H-4), 8.78 (dd, J1 = 4.50 Hz, J2 = 1.50 Hz, 2H, H-2″, H-6″), 7.84 (dd, J1 = 4.50 Hz, J2 = 1.50 Hz, 2H, H-3″, H-5″), 7.66 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.59 (m, 1H, H-6ʹ), 7.4 (d, J = 8.0 Hz, 1H, H-7ʹ), 7.32 (m, 1H, H-6), 7.19 (d, J = 8.0 Hz, 1H, H-7), 7.09 (m, 1H, H-5), 7.03 (m, 1H, H-5ʹ), 5.73 (d, J = 5.50 Hz, 1H, –CHOH), 4.25 (m, 1H, –CHOH–), 3.99 (m, 2H, –N–CH2-), 3.61 (dd, J1 = 13.50 Hz, J2 = 4.0 Hz, 1H, –CH2–S–), 3.42 (dd, J1 = 13.50 Hz, J2 = 7.50 Hz, 1H, –CH2–S–). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 189 (C-3ʹ), 169.9 (C-2), 166 (pyridine–Coxadiazole), 163.9 (–S–Coxadiazole), 152.9 (C-7ʹa), 151.4 (C-2″, C-6″), 142.3 (C-7a), 139.1 (C-2ʹ), 137.7 (C-6ʹ), 130.5 (C-4″), 129.5 (C-6), 124.9 (C-4), 124.8 (C-4ʹ), 122.2 (C-3a), 121.9 (C-5), 121.3 (C-5ʹ), 120.4 (C-3″, C-5″), 119.5 (C-3ʹa), 113.9 (C-7″), 109.6 (C-7), 106.1 (C-3), 67.5(–CHOH–), 45,1(–N–CH2-), 37,6(–CH2–S–). HR-ESI–MS: Caculated for C26H19N5O4S [M]+: 497.1158, found: 497.1157.
(Z)-1ʹ-(3((7H-Purin-6-yl)thio)-2-hydroxypropyl)-[2,3ʹ-biindolinylidene]-2ʹ,3-dione (4e). Yield 63.1%, dark violet solid, mp: 172–173 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 13.49 (s, 1H, NHpurine), 11.11 (s, 1H, H-1ʹ), 8.83 (d, J = 7.50 Hz, 1H, H-4), 8.49 (s, 1H, H-8″), 8.41 (s, 1H, H-2″), 7.67 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.59 (t, J = 7.75 Hz, 1H, H-6ʹ), 7.41 (d, J = 8.0 Hz, 1H, H-7ʹ), 7.31 (t, J = 7.75 Hz, 1H, H-6), 7.16 (d, J = 8.0 Hz, 1H, H-7), 7.08 (t, J = 7.75 Hz, 1H, H-5), 7.04 (t, J = 7.50 Hz, 1H, H-5ʹ), 5.56 (d, J = 5.0 Hz, 1H, –CHOH-), 4.16 (m, 1H, –CHOH–), 4.0 (dd, J1 = 14.0 Hz, J2 = 5.0 Hz, 1H, –N–CH2-), 3.92 (dd, J1 = 14.0 Hz, J2 = 7.0 Hz, 1H, –N–CH2-), 3.7 (m, 1H, –CH2–S–), 3.33 (m, 1H, –CH2–S–). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 189.1 (C-3ʹ), 169.9 (C-2), 159.1 (C-6″), 153 (C-7ʹa), 151.7 (C-4″, C-8″), 149.6 (C-5″), 143.4 (C-2″), 142.5 (C-7a), 138.9 (C-2ʹ), 137.7 (C-6ʹ), 129.5 (C-6), 124.9 (C-4), 124.8 (C-4ʹ), 122.1 (C-3a), 121.9 (C-5), 121.3 (C-5ʹ), 119.5 (C-3ʹa), 113.9 (C-7ʹ), 109.7 (C-7), 106.2 (C-3), 68.2 (–CHOH–), 45.5 (–N–CH2-), 33.2 (–CH2–S–). HR-ESI–MS: Caculated for C24H18N6O3S [M]+: 470.1161, found: 470.1159.
(Z)-1ʹ-(2-Hydroxy-3-((5-(phenylamino)-1,3,4-thiadiazol-2-yl)thio)propyl)-[2,3ʹ-biindolinylidene]-2ʹ,3-dione (4f). Yield 77.0%, dark violet solid, mp: 232–233 °C. IR (KBr, νmax(cm−1)): 3416 (O–H), 3316 (N–H), 2923 (C–H), 1710 (C=O), 1643 (C=N), 1607 and 1499 (C=C), 1319 (C–N), 1093 (N–N), 660 (C–S–C). 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.1 (s, 1H, H-1ʹ), 10.36 (s, 1H, NH-phenyl), 8.82 (d, J = 8.0 Hz, 1H, H-4), 7.67 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.6 (m, 1H, H-6ʹ), 7.55 (d, J = 7.50 Hz, 2H, H-2″, H-6″), 7.42 (d, J = 8.0 Hz, 1H, H-7ʹ), 7.33–7.31 (m, 3H, H-3″, H-5″, H-6), 7.18 (d, J = 8.0 Hz, 1H, H-7), 7.08 (t, J = 7.50 Hz, 1H, H-5), 7.04 (m, 1H, H-5ʹ), 7.0 (m, 1H, H-4″), 5.61 (d, J = 5.50 Hz, 1H, –CHOH-), 4.16 (m, 1H, –CHOH–), 3.94 (m, 2H, –N–CH2-), 3.43 (dd, J1 = 13.25 Hz, J2 = 4.50 Hz, 1H, –CH2–S–), 3.26 (dd, J1 = 13.25 Hz, J2 = 7.25 Hz, 1H, –CH2–S–). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 189.1 (C-3ʹ), 169.9 (C-2), 165.1 (–NH–Cthiadiazole-), 153.7 (–CH2–S–Cthiadiazole), 152.9 (C-7ʹa), 142.4 (C-7a), 140.9 (C-1″), 139 (C-2ʹ), 137.7 (C-6ʹ), 129.6 (C-3″, C-5″), 129.4 (C-6), 124.9 (C-4), 124.8 (C-4ʹ), 122.4 (C-4″), 122.2 (C-3a), 121.9 (C-5), 121.3 (C-5ʹ), 119.5(C-3ʹa), 117.8 (C-2″, C-6″), 113.9 (C-7ʹ), 109.7 (C-7), 106.2 (C-3), 67.9 (–CHOH–), 45.4 (–N–CH2-), 39.4 (–CH2–S–). HR-ESI–MS: Caculated for C27H21N5O3S2 [M]+: 527.1086, found: 527.1082.
(Z)-1ʹ-(3-((5-((2,3-Dimethylphenyl)amino)-1,3,4-thiadiazol-2-yl)thio)propyl)-[2,3ʹ-biindolinylidene]-2ʹ,3-dione (4 g). Yield 69.5%, dark violet solid, mp: 247–248 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.09 (s, 1H, H-1ʹ), 9.54 (s, 1H, NH-phenyl), 8.81 (d, J = 8.0 Hz, 1H, H-4), 7.67 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.59 (m, 1H, H-6ʹ), 7.44 (d, J = 8.0 Hz, 1H, H-4″), 7.41 (d, J = 8.0 Hz, 1H, H-7ʹ), 7.31 (m, 1H, H-6), 7.16 (d, J = 8.0 Hz, 1H, H-7), 7.08 (m, 2H, H-5, H-5″), 7.04 (t, J = 7.50 Hz, 1H, H-5ʹ), 6.99 (d, J = 7.50 Hz, 1H, H-6″), 5.58 (d, J = 5.50 Hz, 1H, –CHOH-), 4.12 (m, 1H, –CHOH–), 3.92 (m, 2H, –N–CH2-), 3.37 (m, 1H, –CH2–S–), 3.21 (dd, J1 = 13.50 Hz, J2 = 7.0 Hz, 1H, –CH2–S–), 2.26 (s, 3H, CH3–C-3″), 2.13 (s, 3H,CH3–C-2″). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 189 (C-3ʹ), 169.9 (C-2), 168.6 (–NH–Cthiadiazole-), 152.9 (C-7ʹa), 152.7 (–CH2–S–Cthiadiazole), 142.4(C-7a), 139.5 (C-1″), 139(C-2ʹ), 137.9 (C-3″), 137.7 (C-6ʹ), 129.7 (C-2″), 129.5 (C-6), 126.9 (C-4″), 126.4 (C-5″), 124.9 (C-4), 124.8 (C-4ʹ), 122.2 (C-3a), 121.9 (C-5), 121.3 (C-5ʹ), 121 (C-6″), 119.5 (C-3ʹa), 113.9 (C-7ʹ), 109.6 (C-7), 106.1 (C-3), 67.9 (–CHOH), 45.3(–N–CH2-), 40(–CH2–S–), 20.7 (CH3–C-3″), 14.3 (CH3–C-2″). HR-ESI–MS: Caculated for C29H25N5O3S2 [M]+: 555.1399, found: 555.1396.
(Z)-1ʹ-(3-((5-((2,4-Dimethylphenyl)amino)-1,3,4-thiadiazol-2-yl)thio)propyl)-[2,3ʹ-biindolinylidene]-2ʹ,3-dione (4 h). Yield 67.4%, dark violet solid, mp: 241–242 °C (decomposition). 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.09 (s, 1H, H-1ʹ), 9.42 (s, 1H, NH-phenyl), 8.81 (d, J = 7.50 Hz, 1H, H-4), 7.67 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.60 (m, 2H, H-6ʹ, H-5″), 7.41 (d, J = 8.0 Hz, 1H, H-7ʹ), 7.31 (1H, m, H-6), 7.16 (d, J = 8.0 Hz, 1H, H-7), 7.09 (d, J = 7.50 Hz, 1H, H-5), 7.04 (m, 2H, H-5ʹ, H-3″), 6.98 (d, J = 8.0 Hz, 1H, H-6″), 5.58 (d, J = 5.50 Hz, 1H, –CHOH-), 4.13 (m, 1H, –CHOH–), 3.92 (m, 2H, –N–CH2-), 3.37 (m, 1H, –CH2–S–), 3.21 (dd, J1 = 13.50 Hz, J2 = 7.0 Hz, 1H, –CH2–S–), 2.24 (s, 3H, CH3–C-4″), 2.19 (s, 3H, CH3–C-2″). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 189 (C-3ʹ), 169.9 (C-2), 168.1 (–NH–Cthiadiazole-), 152.9 (C-7ʹa), 152.8 (–CH2–S–Cthiadiazole), 142.4 (C-7a), 139 (C-2ʹ), 137.7 (C-6ʹ), 137.1 (C-1″), 133.9 (C-4″), 131.8 (C-5″), 130.1 (C-2″), 129.5 (C-6), 127.6 (C-3″), 124.9 (C-4), 124.8 (C-4ʹ), 122.4 (C-6″), 122.2 (C-3a), 121.9 (C-5), 121.3 (C-5ʹ), 119.5 (C-3ʹa), 113.9 (C-7ʹ), 109.6 (C-7), 106.1 (C-3), 67.9 (–CHOH–), 45.3 (–N–CH2-), 40 (–CH2–S–), 20.9 (CH3–C-4″), 18.3 (CH3–C-2″). HR-ESI–MS: Caculated for C29H25N5O3S2 [M]+: 555.1399, found: 555.1393.
Synthesis of (2′Z-3′E)-indirubin-3′-oxime derivatives 7a-i
Synthesis of (2Z,3E)-3-((Oxiran-2-ylmethoxy)imino)-[2,3ʹ-biindolinylidene]-2ʹ-one (6)
Indirubin-3′-oxime 5 with (2′Z-3′E) conformation was first obtained by the reaction of indirubin (1) with hydroxylamine in pyridine according to the procedure as described by Cuong and et al. (2010a, b, c), indirubin-3′-oxime (5) was then O-alkylated with epibromohydrin using a procedure described by Ichimaru (Ichimaru et al. 2015) to give ((2′Z-3′E)-Indirubin-3′-(O-oxiran-2-ylmethyl)oxime) 6 in 70% yield.
General procedure for the synthesis of indirubin-3′-oxime derivatives 7a-i
A solution of compound 6 (50 mg, 0.15 mmol), triethylamine (0.45 mmol, 63 µl) and the corresponding thiols 3a-3i (0.2 mmol) in dried DMF (3 ml) was stirred at room temperature for 40 h. The mixture was then diluted with EtOAc (50 ml) and washed with NaCl 3% solution (3 × 100 ml). The combined EtOAc extract was dried on anhydrous sodium sulfate and solvent was removed under reduced pressure. Crude 7a-i was chromatographed using chloroform: EtOAc as solvent to afford 7a-i in 59.2–75.0% yields.
(2′Z-3′E)-Indirubin-3ʹ-[O-2-hydroxy-3-(5-phenyl-1,3,4-oxadiazole-2-thio)prop-1-yl]oxime (7a). Yield 67.1%, red solid, m.p. 196–197 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.68 (s, 1H, H-1ʹ), 10.75 (s, 1H, H-1), 8.58 (d, J = 7.50 Hz, 1H, H-4), 8.22 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.89 (m, 2H, H-2″, H-6″), 7.6 (m, 1H, H-4″), 7.54 (t, J = 7.50 Hz, 2H, H-3″, H-5″), 7.43 (m, 2H, H-6ʹ, H-7ʹ), 7.11 (m, 1H, H-6), 7.03 (m, 1H, H-5ʹ), 6.96 (t, J = 7.75 Hz, 1H, H-5), 6.87 (d, J = 7.50 Hz, 1H, H-7), 5.86 (d, J = 5.50 Hz, 1H, –CHOH-), 4.7 (dd, J1 = 11.0 Hz, J2= 5.0 Hz, 1H, –O–CH2-), 4.65 (dd, J1= 11.0 Hz, J2 = 6.0 Hz, 1H, –O–CH2-), 4.42 (m, 1H, –CHOH–), 3.69 (dd, J1 = 13.50 Hz, J2 = 4.5 Hz, 1H, –CH2–S–), 3.53 (dd, J1 = 13.5 Hz, J2 = 7.0 Hz, 1H, –CH2–S–). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 171.3 (C-2), 165.5(phenyl–Coxadiazole-), 164.4(–S–Coxadiazole), 152.2 (C-3ʹ), 146 (C-2ʹ), 144.3 (C-7ʹa), 139.2 (C-7a), 133.4 (C-6ʹ), 132.4 (C-4″), 129.8 (C-3″, C-5″), 129.2 (C-4ʹ), 126.9 (C-6), 126.7 (C-2″, C-6″), 123.8 (C-4), 123.5 (C-1″), 122.7 (C-3a), 121.9 (C-5ʹ), 121.1 (C-5), 116.7 (C-3ʹa), 112.2 (C-7ʹ), 109.4 (C-7), 100.8 (C-3), 79.5 (–O–CH2-), 67.9 (–CHOH–), 36.6(–CH2–S–). HR-ESI–MS: calculated for C27H21O4N5S [M]+: 511.1314, found: 511.1305.
(2′Z-3′E)-Indirubin-3ʹ-[O-2-hydroxy-3-(5-(4-methoxyphenyl)-1,3,4-oxadiazole-2-thio)prop-1-yl]oxime (7b). Yield 69.4%, red solid, m.p. 207–208 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.67 (s, 1H, H-1ʹ), 10.75 (s, 1H, H-1), 8.58 (d, J = 8.0 Hz, 1H, H-4), 8.21 (d, J = 8.0 Hz, 1H, H-4ʹ), 7.8 (dd, J1= 6.75 Hz, J2 = 2.25 Hz, 2H, H-2″, H-6″), 7.43 (m, 2H, H-6ʹ, H-7ʹ), 7.12 (m, 1H, H-6), 7.04 (m, 3H, H-3″, H-5″, H-5ʹ), 6.96 (m, 1H, H-5), 6.88 (d, J = 7.50 Hz, 1H, H-7), 5.86 (d, J = 5.50 Hz, 1H, –CHOH-), 4.67 (m, 2H, –O–CH2-), 4.42 (m, 1H, –CHOH–), 3.84 (s, 3H, CH3O-), 3.66 (dd, J1 = 13.25 Hz, J2 = 4.75 Hz, 1H, –CH2–S–), 3.5 (dd, J1 = 13.75 Hz, J2 = 7.25 Hz,1H, –CH2–S–). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 171.3(C-2), 165.5(phenyl–Coxadiazole-), 163.5 (–S–Coxadiazole), 162.4 (C-4″), 152.2 (C-3ʹ), 146 (C-2ʹ), 144.2 (C-7ʹa), 139.2 (C-7a), 133.4 (C-6ʹ), 129.1(C-4ʹ), 128.6 (C-2″, C-6″), 126.9 (C-6), 123.8 (C-4), 122.7 (C-3a), 121.9 (C-5ʹ), 121.1 (C-5), 116.7 (C-3ʹa), 115.8 (C-1″), 115.2 (C-3″, C-5″), 112.2 (C-7ʹ), 109.4 (C-7), 100.8 (C-3), 79.4 (–O–CH2-), 67.9 (–CHOH–), 55.9 (CH3O-), 36.7 (–CH2–S–). HR-ESI–MS: calculated for C28H23N5O5S [M]+: 541.1420, found: 541.1414.
(2′Z-3′E)-Indirubin-3ʹ-[O-2-hydroxy-3-(5-(4-nitrophenyl)-1,3,4-oxadiazole-2-thio)prop-1-yl]oxime (7c). Yield 75.0%, red solid, m.p. 213–214 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.64 (s, 1H, H-1ʹ), 10.71 (s, 1H, H-1), 8.53 (d, J = 8.0 Hz, 1H, H-4), 8.3 (m, 2H, H-3″, H-5″), 8.21 (d, J = 7.50 Hz, 1H, H-4ʹ), 8.06 (m, 2H, H-2″, H-6″), 7.45 (m, 1H, H-6ʹ), 7.4 (d, J = 7.50 Hz, 1H, H-7ʹ), 7.1 (m, 1H, H-6), 7.03 (m, 1H, H-5ʹ), 6.94 (m, 1H, H-5), 6.83 (d, J = 7.50 Hz, 1H, H-7), 5.89 (d, J = 5.50 Hz, 1H, –CHOH-), 4.7 (dd, J1 = 11.50 Hz, J2= 5.50 Hz, 1H, –O–CH2-), 4.65 (dd, J1 = 11.25 Hz, J2 = 5.75 Hz, 1H, –O–CH2-), 4.44 (m, 1H, –CHOH–), 3.72 (dd, J1 = 13.50 Hz, J2 = 5.0 Hz, 1H, –CH2–S–), 3.57 (dd, J1 = 13.50 Hz, J2 = 6.50 Hz, 1H, –CH2–S–). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 171.3 (C-2), 165.7 (phenyl–Coxadiazole-), 164.1 (–S–Coxadiazole), 152.2 (C-3ʹ), 149.4 (C-4″), 146 (C-2ʹ), 144.1 (C-7ʹa), 139.1 (C-7a), 133.4 (C-6ʹ), 129.2 (C-4ʹ), 128.9 (C-1″), 127.9 (C-2″, C-6″), 126.9 (C-6), 124.8 (C-3″, C-5″), 123.7 (C-4), 122.7 (C-3a), 121.9 (C-5ʹ), 121 (C-5), 116.6 (C-3ʹa), 112.2 (C-7ʹ), 109.4 (C-7), 100.8 (C-3), 79.1 (–O–CH2-), 67.8 (–CHOH), 36.7 (–CH2–S–). HR-ESI–MS: calculated for C27H20O6N6S [M]+: 556.1165, found: 556.1155.
(2′Z-3′E)-Indirubin-3ʹ-[O-2-hydroxy-3-(5-(pyridin-4-yl)-1,3,4-oxadiazole-2-thio)prop-1-yl]oxime (7d). Yield 61.7%, red solid, m.p. 216–217 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.67 (s, 1H, H-1ʹ), 10.74 (s, 1H, H-1), 8.75 (dd, J1 = 4.50 Hz, J2 = 1.50 Hz, 2H, H-2″, H-6″), 8.56 (d, J = 8.0 Hz, 1H, H-4), 8.22 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.78 (dd, J1 = 4.25 Hz, J2 = 1.75 Hz, 2H, H-3″, H-5″), 7.43 (m, 2H, H-6ʹ, H-7ʹ), 7.1 (t, J = 7.50 Hz, 1H, H-6), 7.03 (m, 1H, H-5ʹ), 6.96 (t, J = 7.75 Hz, 1H, H-5), 6.85 (d, J = 7.50 Hz, 1H, H-7), 5.88 (d, J = 5.50 Hz, 1H, –CHOH-), 4.7 (dd, J1 = 11.25 Hz, J2= 5.25 Hz, 1H, –O–CH2-), 4.65 (dd, J1 = 11.25 Hz, J2= 5.75 Hz, 1H, –O–CH2-), 4.43 (m, 1H, –CHOH–), 3.72 (dd, J1= 13.50 Hz, J2= 5.0 Hz, 1H, –CH2–S–), 3.56 (dd, J1 = 13.50 Hz, J2 = 7.0 Hz, 1H, –CH2–S–). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 171.3 (C-2), 165.8 (pyridine–Coxadiazole-), 164 (–S–Coxadiazole), 152.2 (C-3ʹ), 151.3 (C-2″, C-6″), 146 (C-2ʹ), 144.2 (C-7ʹa), 139.1 (C-7a), 133.4 (C-6ʹ), 130.5 (C-4″), 129.2 (C-4ʹ), 126.9 (C-6), 123.8 (C-4), 122,7 (C-3a), 121.9 (C-5ʹ), 121.1 (C-5), 120.3 (C-3″, C-5″), 116.7 (C-3ʹa), 112.2 (C-7ʹ), 109.4 (C-7), 100.8 (C-3), 79,3(–O–CH2-), 67.8 (–CHOH–), 36.7(–CH2–S–). HR-ESI–MS: calculated for C26H20O4N6S [M]+: 512.1267, found: 512.1259.
(2′Z-3′E)-Indirubin-3ʹ-[O-2-hydroxy-3-(purine-6-thio)prop-1-yl]oxime (7e). Yield 59.2%, red solid, m.p. 255–256 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 13.52 (s, 1H, NHpurine), 11.69 (s, 1H, H-1ʹ), 10.76 (s, 1H, H-1), 8.56 (s, 1H, H-8″), 8.55 (d, J = 8.0 Hz, 1H, H-4), 8.44 (s, 1H, H-2″), 8.24 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.44 (m, 2H, H-6ʹ, H-7ʹ), 7.08 (t, J = 7.50 Hz, 1H, H-6), 7.03 (m, 1H, H-5ʹ), 6.86 (m, 2H, H-5, H-7), 5.74 (d, J = 5.0 Hz, 1H, –CHOH-), 4.69 (dd, J1 = 11.0 Hz, J2 = 5.0 Hz, 1H, –O–CH2-), 4.64 (dd, J1 = 11.0 Hz, J2 = 6.0 Hz, 1H, –O–CH2-), 4.35 (m, 1H,–CHOH), 3.79 (dd, J1 = 13.50 Hz, J2 = 5.50 Hz, 1H, –CH2–S–), 3.56 (dd, J1 = 13.75 Hz, J2 = 6.75 Hz, 1H, –CH2–S–). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 171.3 (C-2), 159.1 (C-6″), 152 (C-3ʹ), 151.7 (C-4″, C-8″), 149.7 (C-5″), 145.9 (C-2ʹ), 144.3 (C-7ʹa), 143.5 (C-2″), 139.1 (C-7a), 133.4 (C-6ʹ), 129.1 (C-4ʹ), 126.8 (C-6), 123.8 (C-4), 122.7 (C-3a), 121.9 (C-5ʹ), 121 (C-5), 116.7 (C-3ʹa), 112.2 (C-7ʹ), 109.4 (C-7), 100.7 (C-3), 79.9 (–O–CH2-), 68.5 (–CHOH–), 31.9 (–CH2–S–). HR-ESI–MS: calculated for C24H19N7O3S [M]+: 485.1270, found: 485.1268.
(2′Z-3′E)-Indirubin-3ʹ-[O-2-hydroxy-3-(5-phenylamino)-1,3,4-thiadiazole-2-thio)prop-1-yl]oxime (7f). Yield 69.6%, red solid, m.p. 236–237 °C. IR (KBr, νmax(cm−1)): 3416 (O–H overlap N–H), 1735 (C =O), 1616 (C=N), 1600 and 1524 (C=C), 1338 (C–N), 1086 (N–N), 642 (C–S–C). 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.67 (s, 1H, H-1ʹ), 10.72 (s, 1H, H-1), 10.32 (s, 1H, NH-phenyl), 8.58 (d, J = 7.50 Hz, 1H, H-4), 8.19 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.53 (d, J = 8.0 Hz, 2H, H-2″, H-6″), 7.41 (m, 2H, H-6ʹ, H-7ʹ), 7.32 (t, J = 7.75 Hz, 2H, H-3″, H-5″), 7.1 (t, J = 7.50 Hz, 1H, H-6), 7.01 (m, 3H, H-5ʹ, H-4″, H-5), 6.89 (d, J = 7.50 Hz, 1H, H-7), 5.76 (d, J = 5.50 Hz, 1H, –CHOH-), 4.66 (dd, J1 = 11.0 Hz, J2 = 4.50 Hz, 1H, –O–CH2-), 4.59 (dd, J1 = 11.0 Hz, J2 = 6.0 Hz, 1H, –O–CH2-), 4.34 (m, 1H, –CHOH), 3.53 (dd, J1 = 13.50 Hz, J2 = 5.0 Hz, 1H, –CH2–S–), 3.37 (1H, –CH2–S–). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 171.4 (C-2), 165.2 (–NH–Cthiadiazole-), 153.5 (–CH2–S–Cthiadiazole), 152.1 (C-3ʹ), 145.9 (C-2ʹ), 144.3 (C-7ʹa), 140.9 (C-1″), 139.1 (C-7a), 133.3 (C-6ʹ), 129.6 (C-3″, C-5″), 129.2 (C-4ʹ), 126.9 (C-6), 123.9 (C-4), 122.8 (C-3a), 122.5 (C-4″), 121.9 (C-5ʹ), 121.2 (C-5), 117.9 (C-2″, C-6″), 116.7 (C-3ʹa), 112.1 (C-7ʹ), 109.4 (C-7), 100.8 (C-3), 79.6 (–O–CH2-), 68.3 (–CHOH–), 38.3 (–CH2–S–). HR-ESI–MS: calculated for C27H22O3N6S2 [M]+: 542.1195, found: 542.1190.
(2′Z-3′E)-Indirubin-3ʹ-[O-2-hydroxy-3-(5-((2,3-dimethylphenyl)amino)-1,3,4-thiadiazole-2-thio)prop-1-yl]oxime (7 g). Yield 72.3%, red solid, m.p. 231–232 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.7 (s, 1H, H-1ʹ), 10.77 (s, 1H, H-1), 9.55 (s, 1H, NH-phenyl), 8.58 (d, J = 8.0 Hz, 1H, H-4), 8.18 (d, J = 7.50 Hz, 1H, H-4ʹ), 7.43 (m, 3H, H-6ʹ, H-7ʹ, H-4″), 7.13 (m, 1H, H-6), 7.05 (m, 1H, H-5″), 7.00 (m, 3H, H-5ʹ, H-6″, H-5), 6.9 (d, J = 7.50 Hz, 1H, H-7), 5.75 (d, J = 5.50 Hz, 1H, –CHOH-), 4.66 (dd, J1 = 11.0 Hz, J2 = 4.50 Hz, 1H, –O–CH2-), 4.59 (dd, J1= 11.0 Hz, J2= 6.50 Hz, 1H, –O–CH2-), 4.31 (m, 1H, –CHOH–), 3.47 (dd, J1 = 13.50 Hz, J2 = 5.0 Hz, 1H, –CH2–S–), 3.34 (1H, –CH2–S–), 2.26 (s, 3H, CH3 –C-3″), 2.11 (s, 3H, CH3–C-2″). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 171.3 (C-2), 168.8 (–NH–Cthiadiazole-), 152.6 (–CH2–S–Cthiadiazole), 152.1 (C-3ʹ), 146 (C-2ʹ), 144.3 (C-7ʹa), 139.5 (C-7a), 139.1 (C-1″), 138 (C-3″), 133.3 (C-6ʹ), 129.7 (C-2″), 129.1 (C-4ʹ), 127 (C-4″), 126,9 (C-6), 126.4 (C-5″), 123.9 (C-4), 122.8 (C-3a), 121.9 (C-5ʹ), 121.2 (C-5), 121.1 (C-6″), 116.6 (C-3ʹa), 112.2 (C-7ʹ), 109.4 (C-7), 100.7 (C-3), 79.6 (–O–CH2), 68.3 (–CHOH–), 38.5 (–CH2–S–), 20.7 (CH3–C-3″), 14.2 (CH3–C-2″). HR-ESI–MS: calculated for C29H26O3N6S2 [M]+: 570.1508, found: 570.1501.
(2′Z-3′E)-Indirubin-3ʹ-[O-2-hydroxy-3-(5-((2,4-dimethylphenyl)amino)-1,3,4-thiadiazole-2-thio)prop-1-yl]oxime (7 h). Yield 66.8%, red solid, m.p. 233–234 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.78 (s, 1H, H-1ʹ), 10.87 (s, 1H, H-1), 9.53 (s, 1H, NH-phenyl), 8.69 (d, J = 7.50 Hz, 1H, H-4), 8.28 (d, J = 8.0 Hz, 1H, H-4ʹ), 7.65 (d, J = 8.0 Hz, 1H, H-5″), 7.53 (m, 2H, H-6ʹ, H-7ʹ), 7.23 (m, 1H, H-6), 7.10 (m, 4H, H-5ʹ, H-3″, H-6″, H-5), 7.01 (d, J = 8.0 Hz, 1H, H-7), 5.85 (d, J = 5.50 Hz, 1H, –CHOH-), 4.76 (dd, J1 = 11.0 Hz, J2= 4.50 Hz, 1H, –O–CH2-), 4.69 (dd, J1 = 11.0 Hz, J2 = 6.0 Hz, 1H, –O–CH2-), 4.42 (m, 1H, –CHOH–), 3.58 (dd, J1 = 13.50 Hz, J2 = 5.50 Hz, 1H, –CH2–S–), 3.44 (1H, –CH2–S–), 2.35 (s, 3H, CH3–C-4″), 2.12 (s, 3H, CH3–C-2″). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 171.3 (C-2), 168.2 (–NH–Cthiadiazole-), 152.6 (–CH2–S–Cthiadiazole), 152.1 (C-3ʹ), 146 (C-2ʹ), 144.3 (C-7ʹa), 139.1 (C-7a), 137 (C-1″), 133.9 (C-4″), 133.3 (C-6ʹ), 131.8 (C-5″), 130.1 (C-2″), 129.1 (C-4ʹ), 127.6 (C-3″), 126.9 (C-6), 123.7 (C-4), 122.8 (C-3a), 122.4 (C-6″), 121.9 (C-5ʹ), 121.2 (C-5), 116.7 (C-3ʹa), 112.2 (C-7ʹ), 109.4 (C-7), 100.8 (C-3), 79.6 (–O–CH2-), 68.3 (–CHOH–), 38.4 (–CH2–S–), 20.9 (CH3–C-4″), 18.2 (CH3–C-2″). HR-ESI–MS: calculated for C29H26O3N6S2 [M]+: 570.1508, found: 570.1502.
(2′Z-3′E)-Indirubin-3ʹ-[O-2-hydroxy-3-(5-((4-ethylphenyl)amino)-1,3,4-thiadiazole-2-thio)prop-1-yl]oxime (7i). Yield 65.6%, red solid, m.p. 221–222 °C. 1H NMR (DMSO-d6, 500 MHz),δ(ppm): 11.68 (s, 1H, H-1ʹ), 10.74 (s, 1H, H-1), 10.24 (s, 1H, NH-phenyl), 8.58 (d, J = 7.50 Hz, 1H, H-4), 8.19 (d, J = 8.0 Hz, 1H, H-4ʹ), 7.43 (d, J = 8.50 Hz, 2H, H-2″, H-6″), 7.40 (m, 2H, H-6ʹ, H-7ʹ), 7.15 (d, J = 8.50 Hz, 2H, H-3″, H-5″), 7.1 (m, 1H, H-6), 7.03 (m, 1H, H-5ʹ), 6.98 (m, 1H, H-5), 6.88 (d, J = 8.0 Hz, 1H, H-7), 5.75 (d, J = 5.50 Hz, 1H, –CHOH-), 4.66 (dd, J1 = 11.0 Hz, J2 = 4.50 Hz, 1H, –O–CH2-), 4.59 (dd, J1 = 11.0 Hz, J2 = 6.50 Hz, 1H, –O–CH2-), 4.34 (m, 1H, –CHOH–), 3.52 (dd, J1 = 13.50 Hz, J2 = 5.0 Hz, 1H, –CH2–S–), 3.34 (1H, –CH2–S–), 2.55 (q, J = 7.50 Hz, 2H, CH3–CH2-), 1.16 (t, J = 7.50 Hz, 3H, CH3–CH2-). 13C NMR (DMSO-d6, 125 MHz),δ(ppm): 170.9 (C-2), 165.0 (–NH–Cthiadiazole-), 152.5(–CH2–S–Cthiadiazole), 151.6 (C-3ʹ), 145.5 (C-2ʹ), 143.8 (C-7ʹa), 138.7 (C-7a), 138.2 (C-1″), 137.5 (C-4″), 132.9 (C-6ʹ), 128.7 (C-4ʹ), 128.3 (C-3″, C-5″), 126.4 (C-6), 123.4 (C-4), 122.3 (C-3a), 121.5 (C-5ʹ), 120.8 (C-5), 117.6 (C-2″, C-6″), 116.2 (C-3ʹa), 111.7 (C-7ʹ), 108.9 (C-7), 100.3 (C-3), 79.2 (–O–CH2-), 67.8 (–CHOH–), 37.9 (–CH2–S–), 27.5 (CH3–CH2-), 15.7 (CH3–CH2-). HR-ESI–MS: calculated for C29H26O3N6S2 [M]+: 570.1508, found: 570.1502.
Results and discussion
(2′Z)-Indirubin derivatives 4a-h (Table 1) were synthesized via a two-step procedure (Scheme 1). Indirubin was first converted into the key intermediate 2 by the nucleophilic substitution at N1 position with epichlorohydrin using K2CO3 as a catalyst in the presence of a transfer calalyst (1-butyl)triethylammonium bromide) to give 2 in 40% yield. The N1-alkylation of indirubin was confirmed by the interactions of protons in –N1–CH2- at 4.20 ppm (dd, J1 = 15.0 Hz, J2 = 3.75 Hz) and 3.88 ppm (dd, J1 = 15.0 Hz, J2 = 5.25 Hz) with C-2 (169.6 ppm) and C-7a (141.9 ppm) of indirubin in HMBC spectrum. Epoxy 2 was then opened with thiols 3a-h to obtain target compounds 4a-h in 63.1–77.0% yields (Scheme 1).
The structure of 4a-h was determined by 1D, 2D and HRMS spectra. The spectral signal distinction among derivatives 4a-h was found due to the resonance of protons and carbons in thiol moieties. Compound 4f was selected to assign structure supported extensively by HSQC and HMBC correlations. The data given from 1H- and 13C-NMR spectra of 4f as well as the support from HSQC and HMBC spectra agreed well with the structure 4f. The structure of the remaining was in good agreement with NMR and HRMS data.
Series of new (2′Z-3′E)-indirubin-3′-oxime derivatives (7a-i) were produced by opening epoxy ring at oxime position of the key intermediate 6 that was synthesized from indirubin (1) in two steps. Compound 5 with (2E,3Z) conformation was first obtained in 93% yield by a condensation reaction of indirubin with hydroxylamine hydrochloride in pyridine (Cuong et al. 2010a, b, c). Compound 5 was then O-alkylated using epibromohydrin catalyzed by triethylamine according to a known procedure (Ichimaru et al. 2015) to afford 6 in 70% yield (Scheme 2). In the last step, 6 was opened with the corresponding thiols 3a-i in DMF in the presence of triethylamine as a catalyst to give 7a-i in 59.2–75.0% yields (Table 1).
The structure of 7a-i was confirmed by 1D, 2D and HRMS spectra. The similarity of the multiplicity and signal position of indirubin moiety was easily observed in 1H- and 13C-NMR spectra of compounds 7a-i and the distinction among those was also found due to the different structure of thiol components. Compound 7f was selected to assign structure supported extensively by HSQC and HMBC correlations. In 1H-NMR spectra, protons H-1, H-1′ of indirubin and NH of thiol moiety appeared as singlet signals at 10.72, 11.67 and 10.32 ppm respectively. Two doublets at 8.58 ppm (J = 7.50 Hz) and 8.19 ppm (J = 7.50 Hz) attributed to H-4 and H-4′ protons, and the overlapped doublet signal (δ: 7.53 ppm, J = 8.0 Hz) was assigned to protons H-2′’ and H-6′’. Protons H-3′’ and H-5′’ were recognized by a triplet at 7.32 ppm (J = 7.75 Hz) and other triplet at 7.10 ppm (J = 7.50 Hz) arose from H-6. Besides a multiplet at 7.41 ppm arising from H-6′ and H-7′ of indirubin moiety, signal of the remaining thiol proton H-4′’ overlapped by protons H-5 and H-5′ of indirubin component was found due to a multiplet at (7.01 ppm). The final proton H-7 in indirubin resonates as a doublet (J1 = 7.50 Hz) at 6.89 ppm. In 2-ol-1,3-propylen linker, a doublet (J = 5.50 Hz) at 5.76 ppm was assigned to proton –OH and methine proton attached to this group appeared as a multiplet at 4.34 ppm. In addition, doublet–doublet signals at 4.66 ppm (J1 = 11.0 Hz, J2 = 4.50 Hz) and 4.59 ppm (J1 = 11.0 Hz, J2 = 6.0 Hz) were determined to be resonance signals of protons O–CH2-, and the signals at 3.53 ppm and 3.37 ppm were characteristic of two protons, which were neighbor to sulfur (S–CH2). Based on HSQC spectrum, the chemical shifts of the corresponding carbons were elucidated as: C-4: 123.9, C-4′: 129.2, (C-2′’, C-6′’): 117.9, (C-3′’, C-5′’): 129.6; CCHOH: 68.3, C–OCH2-: 79.6, CS–CH2–CHOH–CH2-: 38.3 ppm. In addition, the key cross-picks of Cthiadiazole (153.5 ppm) with HS–CH2–CHOH– in HMBC approved the correct structure of 7f (Scheme 2).
Bioactivity
In the present study, indirubin (1), indirubin-3′-oxime (5) and 6-mercaptopurine (6-MP), a medicine for treatment of blood cancer were also evaluated for cytotoxicity against SW480, LU-1, HepG2 and HL-60 cell lines along with indirubin and indirubin-3′-oxime derivatives (4a-h and 7a-i), and ellipticine was used as positive control for SAR evaluation (Table 1). Cytotoxic activity against SW480, LU-1, HepG2 cell lines and anti-proliferative activity of HL-60 cell line were undertaken using the method as described by Monks (Monks et al. 1991). The derivatives 4a-h, 7a-i were judged to have no activity when their IC50 values > 20 µM.
In screening for cytotoxic and anti-proliferative activities against four cancer cell lines: SW480, LU-1, HepG2 and HL 60, indirubin did not show direct activity. This was also observed in some its derivative 4a-h excluding 4f and 4 h (Table 2). Obviously, the derivatives derived from N1 position did not give the clear advantages to parent compound, indirubin, even derivative 4e formed by the opening epoxy with 6-mercaptopurine (6-MP). However, in some cases, the structure of thiol component has a significant effect on cytotoxic and anti-proliferative activities. Compounds 4f, 4 h derived from indirubin and 5-(phenylamino)-1,3,4-thiadiazole-2-thiol or 5-((2,4-dimethylphenyl)amino)-1,3,4-thiadiazole-2-thiol showed strong activity against SW480, LU-1, HepG2 and HL-60 cell lines in which compound 4f exhibited the strongest activity with IC50 values of 1.65, 2.21, 1.90 and 1.35 µM, respectively, much stronger than those of 6-mercaptopurin, indirubin-3′-oxime and nearly equivalent to those of ellipticine.
In series 7a-i, data on cytotoxic and anti-proliferative activities indicated that 3′-oxime group could be an important anticancer activity center of indirubin-3′-oxime. The chemical intervention in this group resulted in the decreased activity, and the synthesized derivatives showed no activity against SW480, LU-1, HepG2, and HL-60 cell lines except derivatives 7a, 7f, 7 g and 7 h containing phenylaminothiadiazole, phenylthiazole and 6-mecarptopurin exhibited weak activity with IC50 values in range of 15.34–19.24 µM and not so prominent. An expected result was also found with derivative 7e, a conjugate of indirubin-3′-oxime and a medicine for treatment of blood cancer, 6-mecarptopurine was negative with tested cell lines. Evidently, the evaluation of cytotoxic and anti-proliferative activities of indirubin and indirubin-3′-oxime derivatives resulted in conclusion that the particular structure of parent compound, (2′Z)-indirubin and 3′-oxime group were main activity centers of (2′Z-3′E)-indirubin-3′-oxime. We suggest this be an important orientation for the design, synthesis of indirubin-3-oxime derivatives in a search for new cytotoxic agents in our future research.
Conclusions
The design, synthesis and screening for cytotoxic and anti-proliferative activities of indirubin derivatives containing thiols were successfully performed. The cytotoxic and anti-proliferative activities of five in 17 new derivatives were also carried out. Data on cytotoxic and anti-proliferative activities of two series 4a-h and 7a-i could lead to conclusion that the combination of specific configuration of indirubin and 3′-oxime group in its derivative was necessary for anticancer activity. Therefore, chemical transformation at oxime group did not improve anticancer activity while chemical intervention at N1 position could produce new derivatives that showed strong cytotoxic and anti-proliferative activities in some cases. This may be an important orientation in finding candidates from indirubin that can be used in cancer treatment in the future.
References
Bektas I, Karaman S, Diraz E, Celik M (2016) The role of natural indigo dye in alleviation of genotoxicity of sodium dithionite as reducing agent. Cytotechnology 63:2245–2255. https://doi.org/10.1007/s10616-016-0018-7
Choi OM, Cho YH, Choi S, Lee SH, Seo SH, Kim HY, Han G, Min DS, Park T, Choi KY (2014) The small molecule indirubin-3′-oxime activates Wnt/b-catenin signaling and inhibits adipocyte differentiation and obesity. Int J Obes 38:1044–1052. https://doi.org/10.1038/ijo.2013.209
Cuong NM, Thuy DTT, Ha NV, Tai BH (2007) Isolation of indirubin from the leaves of Strobilanthes cusia. J Sci Technol 45(3A):195–199
Cuong NM, Tai BH, Hoan DH, Long PQ, Choi EM, Kim YH (2010a) Synthesis and anti-osteoporosis potential of two new indirubin-3-oxime derivatives. J Korean Soc Appl Biol Chem 53:22–26. https://doi.org/10.3839/jksabc.2010.004
Cuong NM, Tai BH, Hoan DH, Huong TT, Kim YH, Hyun JH, Kang HK (2010b) Inhibitory effects of indirubin derivatives on the growth of HL-60 leukemia cell. Nat Prod Commun 5:103–106
Cuong NM, Tai BH, Hoan DH (2010c) Studies on acetylation and NMR reassignment of indirubin derivatives. Nat Prod Res 24:99–105. https://doi.org/10.1080/14786410802300469
Gilani SJ, Khan SA, Alam O, Siddiqui N (2011) Synthesis and in vitro antimicrobial evaluation of condensed heterocyclic 6-substituted 1,2,4-triazolo-[3.4-b]-1,3,4-thiadiazole and 1,3,4-oxadiazole derivatives of isoniazid. Acta Pol Pharm 68:205–211
Girard SK, Mialou V, Chebel A, Chien WW, Tigaud I, Mokdad F, Badiou C, Ffrench M (2007) Inhibition of normal lymphocyte proliferation by Indirubin-30-monoxime: a multifactorial process. Leukemia & Lymphoma 48:605–615. https://doi.org/10.1080/10428190601059696
Hoessel R, Leclerc S, Endicott JA, Nobel ME, Lawrie A, Tunnah P, Leost M, Damiens E, Marie D, Marko D, Niederberger E, Tang W, Eisenbrand G, Meijer L (1999) Indirubin, the active constituent of a Chinese antileukaemia medicine, inhibits cyclin-dependent kinases. Nat Cell Biol 1:60–67. https://doi.org/10.1038/9035
Ichimaru Y, Saito H, Uchiyama T, Metori K, Tabata K, Suzuki T, Miyairi S (2015) Indirubin 3´-(O-oxiran-2-ylmethyl)oxime: a novel anticancer agent. Bioorg Med Chem Lett 25:1403–1406. https://doi.org/10.1016/j.bmcl.2015.02.053
Jung ME, Byun BJ, Kim HM, Lee JY, Park JH, Lee N, Son YH, Choi SU, Yang KM, Kim SJ, Lee KH, Kim YC, Choi G (2016) Discovery of indirubin derivatives as new class of DRAK2 inhibitors from high throughput screening. Bioorganic Med Chem Lett 26:2719–2723. https://doi.org/10.1016/j.bmcl.2016.03.111
Karapetyan G, Chakrabarty K, Hein M, Langer P (2011) Synthesis and Bioactivity of carbohydrate derivatives of indigo, its isomers and heteroanalogues. Med Chem Med 6:25–37. https://doi.org/10.1002/cmdc.201000374
Lectere S, Garnier M, Hoessel R, Marko D, Bibb JA, Snyder GL, Greengard P, Biernard J, Wu YZ, Mandelkow EM, Eisenbrand G, Meijer L (2001) Indirubins inhibit glycogen synthase kinase-3β and CDK5/P25, two protein kinases involved abnormal tau phosphorylation in alzheimer’s disease. J Biol Chem 276:251–260. https://doi.org/10.1074/jbc.M002466200
Liao XM, Leung KN (2013) Indirubin-3-oxime induces mytochondrial dysfunction and triggers growth inhibition in cell cycle arrest in human neuroblastoma cells. Oncol Rep 29:371–379. https://doi.org/10.3892/or.2012.2094
Lo WY, Chang NW (2013) An indirubin derivative, indirubin-3′-monoxime suppresses oral cancer tumorigenesis through the down regulation of survivin. PLoS ONE. https://doi.org/10.1371/journal.pone.0070198
Meijer L, Skaltsounis AL, Magiatis P, Polychronopoulos P, Leost M, Ryan XP, Vonica CA, Brivanlou A, Dajani R, Crovace C, Tarricone C, Musacchio A, Roe SM, Pearl L, Greengard P (2003) GSK-3-Selective inhibitors derived from tyrian purple indirubins. Chem Biol 10:1255–1266. https://doi.org/10.1016/j.chembiol.2003.11.010
Monks A, Scudiero D, Skehan P, Shoemaker R, Paull K, Vistica D, Hose C, Langley J, Cronise P, Vaigro-Wolff A, Gray-Goodrich M (1991) Feasibility of a high-flux anticancer drug screen using a diverse panel of cultured human tumor cell lines. J Natl Cancer Inst 83:757–766. https://doi.org/10.1093/jnci/83.11.757
Nam SK, Buettner R, Turkson J, Kim DH, Cheng JQ, Muehlbeyer S, Hippe F, Vatter S, Merz KH, Eisenbrand G, Jove R (2005) Indirubin derivatives inhibit stat3 signaling and induce apoptosis in human cancer cells. PNAS 102:5998–6003. https://doi.org/10.1073/pnas.0409467102
Perrin DD, Armarego WLF (1988) Purification of Laboratory Chemical, 3rd edn. Pergamon Press, Oxford
Raju GN, Prathyusha TG, Sowmya PL, Mounika SJ, Nadendla RR (2015) Synthesis, characterization and biological activity of some 1,3,4-oxadiazole derivatives with benzothiazole moiety. Der Pharmacia Sinica 6:1–8
Riepl HM, Urmann C (2012) Improved synthesis of indirubin derivatives by sequential Buid-up of indoxyl unit: first preparation of fluorescent indirubins. Helv Chim Acta 95:1461–1477. https://doi.org/10.1002/hlca.201200042
Sano M, Ichimaru Y, Kurita M, Hayashi E, Homma T, Saito H, Masuda S, Nemoto N, Hemmi A, Suzuki T, Miyairi S, Hao H (2017) Induction of cell death in pancreatic ductal adenocarcinoma by indirubin 3′-oxime and 5-methoxyindirubin 3′-oxime in vitro and in vivo. Can Lett 397:72–82. https://doi.org/10.1016/j.canlet.2017.03.031
Stasiak N, Koch WK, Glowniak K (2014) Modern industrial and pharmacological applications of indigo dye and its derivatives—a review. Acta Poloniae Pharmaceutica Drug Research 71:215–221
Vaughan JR (1957) Heterocyclic sulphonamides. US patent 2783240
Xiao Hao Y, Liu B, Qian L (2002) Indirubin and Mesoindigo in the Treatment of Chronic Myelogenous Leukemia in China. Leukemia and lymphoma 43:1763–1768
Acknowledgements
The authors gratefully acknowledge Vietnam-Russia Tropical Center and Vietnam Academy of Science and Technology (VAST) for financial support via project VAST.HTQT.BELARUS.04/15-16.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Nguyen, D.T., Truong, G.N., Van Vuong, T. et al. Synthesis of new indirubin derivatives and their in vitro anticancer activity. Chem. Pap. 73, 1083–1092 (2019). https://doi.org/10.1007/s11696-018-0659-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-018-0659-4