Abstract
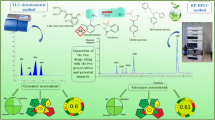
A simple green sample preparation procedure and RP-HPLC method were developed and validated for the determination and quantification of two anti-hemorrhoid drugs in semisolid dosage form as well as in rat rectal tissues. Sample cleanup was performed using solid-phase extraction on strata-X prefilled (100 mg, particle size 33 µm, 3 ml) cartridges; conditions were optimized to obtain maximum recoveries and minimal baseline noise. Separation was carried out on Xselect C18 column (250 × 4.6 mm id, 5 µm particle size), a mobile phase consisting of ethanol: 0.01% aqueous sodium carbonate solution (75:25 by volume) at a flow rate of 1 ml/min and UV detection at 220 nm. The method was validated as per the ICH guidelines and FDA guidance for bioanalytical method validation. Greenness of the method was assessed throughout the whole procedure of sample preparation, separation and quantification using Eco-scale and analytical method volume intensity metrics and was found to be environmentally friendly as it was also manifested by its greenness profile. The developed method was found to be suitable for the determination of the selected drugs in their marketed formulation as well as in biological samples. Being simple and eco-friendly technique, it can be used for routine analysis in quality control laboratories.




Similar content being viewed by others
References
Anastas PT (1999) Green Chemistry and the role of analytical methodology development. Crit Rev Anal Chem 29(3):167–175. https://doi.org/10.1080/10408349891199356
Armenta S, Garrigues S, de la Guardia M (2008) Green analytical chemistry. TrAC Trends Anal Chem 27(6):497–511. http://www.sciencedirect.com/science/article/pii/S016599360800109X
Ascaso M, Pérez-Lozano P, García M, García-Montoya E, Miñarro M, Ticó JR, Suñé-Negre JM (2015) Development and validation of a stability indicating RP-HPLC method for hydrocortisone acetate active ingredient, propyl parahydroxybenzoate and methyl parahydroxybenzoate preservatives, butylhydroxyanisole antioxidant, and their degradation products in a rectal gel formulation. J AOAC Int 98(1):27–34. https://doi.org/10.5740/jaoacint.13-411
British Pharmacopoeia (Her Majesty’s Stationary Office) (2014)
Capello C, Fischer U, Hungerbühler K (2007) What is a green solvent? A comprehensive framework for the environmental assessment of solvents. Green Chem 9(9):927. https://doi.org/10.1039/b617536h
Chen D, Tao Y, Liu Z, Zhang H, Liu Z, Wang Y, Yuan Z (2011) Development of a liquid chromatography-tandem mass spectrometry with pressurized liquid extraction for determination of glucocorticoid residues in edible tissues. J Chromatogr B Anal Technol Biomed Life Sci 879(2):174–180. https://doi.org/10.1016/j.jchromb.2010.11.039
El-Bagary RI, Elkady EF, Hosny M, Abo Elmaaty A (2012) Simultaneous determination of miconazole and hydrocortisone or mometasone using reversed phase liquid chromatography. Eur J Chem 3(4):421–425. https://doi.org/10.5155/eurjchem.3.4.421
El-Gindy A, Korany MA, Bedair MF (1998) First derivative spectrophotometric and high-performance liquid chromatographic determination of cinchocaine hydrochloride in presence of its acid degradation product. J Pharm Biomed Anal 17(8):1357–1370. https://doi.org/10.1016/S0731-7085(98)00041-7
Elsayed MMA (2007) Rapid determination of cinchocaine in skin by high-performance liquid chromatography. Biomed Chromatogr 1(2):491–496. https://doi.org/10.1002/bmc
FDA (1994) Center of Drug Evaluation and Research (FDA) reviewer guidance on validation of chromatographic methods [Online]. http://www.fda.gov/downloads/Drugs/Guidances/UCM134409.pdf. Accessed 5 Apr 2017
Flórez J, Armijo JA, Mediavilla A (1997) Farmacologia humana, 7th edn. S.A Barcelona, Masson
Gałuszka A, Migaszewski ZM, Konieczka P, Namieśnik J (2012) Analytical Eco-scale for assessing the greenness of analytical procedures. TrAC Trends Anal Chem 37(July):61–72. https://doi.org/10.1016/j.trac.2012.03.013
Guide for the Care and Use of Laboratory Animals (2011) Committee for the Update of the Guide for the Care and Use of Laboratory Animals, 1st edn. The National Academies Press, Washington, DC. https://doi.org/10.1163/1573-3912_islam_DUM_3825
Hartman R, Helmy R, Al-Sayah M, Welch CJ (2011) Analytical method volume intensity (AMVI): a green chemistry metric for HPLC methodology in the pharmaceutical industry. Green Chem 13(4):934. https://doi.org/10.1039/c0gc00524j
Ibrahim F, El-Deen AK, Shimizu K (2018) Comparative study of two different chromatographic approaches for quantitation of hydrocortisone acetate and pramoxine hydrochloride in presence of their impurities. J Food Drug Anal 26(3):1160–1170. https://doi.org/10.1016/j.jfda.2017.12.008
International Conference on Harmonization (ICH) (2005) Q2 (R1); validation of analytical procedures: text and methodology. Geneva. http://www.ich.org/LOB/media/MEDIA417.pdf. Accessed 14 Oct 2013
Keith LH, Gron LU, Young JL (2007) Green analytical methodologies. Chem Rev 107(6):2695–2708. https://doi.org/10.1021/cr068359e
Kitaichi Y, Miyamoto A, Uchikura K (2010) Determination of selected corticosteroids in sewage-treatment-plant samples by liquid chromatography–mass spectrometry. J Health Sci 56(5):547–556. https://doi.org/10.1248/jhs.56.547
Koel M, Kaljurand M (2006) Application of the principles of green chemistry in analytical chemistry. Pure Appl Chem 78(11):1993–2002. https://doi.org/10.1351/pac200678111993
Sweetman SC (2007) Martindale: The complete drug reference. Pharmaceutical Press, London UK
National Environmental Methods Index (2018) [Online]. www.nemi.gov. Accessed 20 Jan 2016
Pendela M, Kahsay G, Baekelandt I, Van Schepdael A, Adams E (2011) Simultaneous determination of lidocaine hydrochloride, hydrocortisone and nystatin in a pharmaceutical preparation by RP-LC. J Pharm Biomed Anal 56(3):641–644. https://doi.org/10.1016/j.jpba.2011.06.028
Peraman R, Kalva B, Shanka S, Reddy YP (2014) Analytical quality by design (AQbD) approach to liquid chromatographic method for quantification of acyclovir and hydrocortisone in dosage forms. Anal Chem Lett 4(5–6):329–342. https://doi.org/10.1080/22297928.2014.999121
Peraman R, Kalva B, Reddy YP, Sharma H (2016) Analytical quality by design approach in selection of method variables for simultaneous analysis of ciprofloxacin and hydrocortisone by LC method using Taguchi method. Anal Chem Lett 6(1):1–12. https://doi.org/10.1080/22297928.2016.1153431
Rms M, Ps E, Csab R, Sheila G (2017) Validation of a new RP-HPLC method for determination of hydrocortisone acetate complexed with HPβCD in a oral liquid pharmaceutical preparation. J Anal Pharm 6:3–7. https://doi.org/10.15406/japlr.2017.06.00180
Rosa AM, Sversut RA, Silva DB, Cardoso TFM, Amaral MS, Carollo ARH, Kassab NM (2017) Simultaneous determination of enrofloxacin, silver sulfadiazine, hydrocortisone acetate, hydrocortisone sodium succinate, and preservative excipients in pharmaceutical preparations using HPLC–DAD method. Chromatographia 80(11):1641–1649. https://doi.org/10.1007/s10337-017-3391-2
Šatínský D, Chocholouš P, Válová O, Hanusová L, Solich P (2013) Two-column sequential injection chromatography for fast isocratic separation of two analytes of greatly differing chemical properties. Talanta 114:311–317. https://doi.org/10.1016/j.talanta.2013.05.055
Tobiszewski M, Marć M, Gałuszka A, Namies̈nik J (2015) Green chemistry metrics with special reference to green analytical chemistry. Molecules 20(6):10928–10946. https://doi.org/10.3390/molecules200610928
Validation BM (2018) Guidance for industry bioanalytical method validation guidance for industry bioanalytical method validation. Fda, pp 1–22. http://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/default.htm. Accessed 10 June 2018
Wu S, Li W, Mujamdar T, Smith T, Bryant M, Tse FLS (2010) Supported liquid extraction in combination with LC–MS/MS for high-throughput quantitative analysis of hydrocortisone in mouse serum. Biomed Chromatogr 24(6):632–638. https://doi.org/10.1002/bmc.1339
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saad, A.S., El-Ghobashy, M.R., Ayish, N.S. et al. Greenness assessment as per Eco-scale and AMVI metrics for the chromatographic assay of selected drugs in a semisolid dosage form and in tissues. Chem. Pap. 73, 683–691 (2019). https://doi.org/10.1007/s11696-018-0619-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-018-0619-z