Abstract
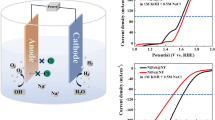
In the case of hydrogen production involving seawater electrolysis, one of the main targets is to develop more active cathodic materials, to optimize the efficiency of hydrogen evolution reaction (HER) and, by doing so, enhance the overall energy efficiency of electrolysis. Thus, to develop suitable HER electrocatalysts either an increase of the electrode active surface area or a design of a material having high intrinsic catalytic activity should be taken into consideration, both of them decreasing the HER overpotential. In the present work, various Ni–Mo alloy nanostructures (10–40 wt% Mo) have been prepared involving electrochemical deposition from aqueous and deep eutectic solvent (DES)-based electrolytes as potential cathodic materials suitable for hydrogen evolution reaction during water electrolysis. The electrocatalytic activity of the obtained layers has been investigated using real seawater electrolyte. The determined Tafel slopes suggested that the electrodeposited Ni–Mo alloy coatings follow an HER mechanism controlled by the Volmer reaction step. The EIS results indicated that the use of choline chloride-based ionic liquids as electrolytes facilitated Ni–Mo alloy coatings showing a significant increase in surface roughness. Studies of the intrinsic activity showed that the main contribution towards the apparent activity comes from the increase of the real surface area, although a slight increase of the intrinsic electrocatalytic activity in the case of Ni–Mo alloy coatings electrodeposited on Ni foam was also noticed. These results showed that Ni–Mo alloy coatings electrodeposited from the novel electrolytes based on choline chloride–urea–citric acid ternary mixtures associated with a porous substrate may represent a promising technological approach to build cathodic materials suitable for seawater electrolysis.












Similar content being viewed by others
References
Abbott AP, McKenzie KJ (2006) Application of ionic liquids to the electrodeposition of metals. Phys Chem Chem Phys 8:4265–4279
Abbott AP, Boothby D, Capper G, Davies DL, Rasheed RK (2004) Deep eutectic solvents formed between choline chloride and carboxylic acids: versatile alternatives to ionic liquids. J Am Chem Soc 126:9142–9147
Abdel-Aal HK, Zohdy KM, Abdel Kareem M (2013) Hydrogen Production using sea water electrolysis. Open Fuel Cells J 3:1–7. https://doi.org/10.2174/1875932701003010001
Angelo ACD, Lasia A (1995) Surface effects in the hydrogen evolution reaction on Ni-Zn alloy electrodes alkaline solutions. J Electrochem Soc 142:3313–3319. https://doi.org/10.1149/1.2049980
Anicai L, Pertache A, Visan T (2007) Thin black layers on aluminum substrate – electrochemical synthesis and characterization. Surf Interface Anal 40:818–821. https://doi.org/10.1002/sia.2782
Anicai L, Petica A, Costovici S, Prioteasa P, Visan T (2013) Electrodeposition of Sn and NiSn alloys coatings using choline chloride based ionic liquids—Evaluation of corrosion behavior. Electrochim Acta 114:868–877
Armstrong RD, Henderson MH (1972) Impedance plane display of a reaction with an adsorbed intermediate. J Electroanal Chem 39:81–90. https://doi.org/10.1016/S0022-0728(72)80477-7
Arul Raj I (1993) Nickel-based, binary composite electrocatalysts for the cathodes in the energy-eficient industrial production of hydrogen from alkaline-water electrolytic cells. J Mater Sci 28:4375–4382. https://doi.org/10.1007/BF01154945
Arul Raj I, Vasu KI (1990) Transition metal-based hydrogen electrodes in alkaline solution—Electrocatalysis on nickel based binary alloy coatings. J Appl Electrochem 20:32–38. https://doi.org/10.1007/BF01012468
Badea GE, Caraban A, Cret P, Corbu I (2007) Water Electrolysis. University of Oradea, Fascicle of Management and Technological Engineering VI, XVI
Baniasadi E, Dincer I, Naterer GF (2013) Electrochemical analysis of seawater electrolysis with molybdenum-oxo catalysts. Int J Hydrogen Energy 38:2589–2595. https://doi.org/10.1016/j.ijhydene.2012.11.106
Bennett JE (1980) Electrodes for generation of hydrogen and oxygen from seawater. Int J Hydrogen Energy 5:401–408. https://doi.org/10.1016/0360-3199(80)90021-X
Brenner A (1963) Electrodeposition of alloys, vol 1 and 2. Academic Press, New York
Corbu I (2004) Ecological sea electro power station, Romanian Patent Application No. A00840/05.10.2004, copyright ©AMPI 205072
Costovici S, Manea AC, Visan T, Anicai L (2016) Investigation of Ni-Mo and Co-Mo alloys electrodeposition involving choline chloride based ionic liquids. Electrochim Acta 207:97–111. https://doi.org/10.1016/j.electacta.2016.04.17
Damian A, Omanovic S (2006) Ni and NiMo hydrogen evolution electrocatalysts electrodeposited in a polyaniline matrix. J Power Sour 158(1):464–476
El-Manharawy S, Hafez A (2000) Technical management of RO system. Desalination 131:329–344. https://doi.org/10.1016/S0011-9164(00)90017-1
Eugénio S, Silva TM, Carmezim MJ, Duarte RG, Montemor MF (2014) Electrodeposition and characterization of nickel–copper metallic foams for application as electrodes for supercapacitors. J Appl Electrochem 44:455–465. https://doi.org/10.1007/s10800-013-0646-y
Fan C, Piron DL, Sleb A, Paradis P (1994) Study of electrodeposited nickel–molybdenum, nickel–tungsten, cobalt–molybdenum, and cobalt–tungsten as hydrogen electrodes in alkaline water electrolysis. J Electrochem Soc 141:382–387. https://doi.org/10.1149/1.2054736
Florea A, Anicai L, Costovici S, Golgovici F, Visan T (2010) Ni and Ni alloy coatings electrodeposited from choline chloride-based ionic liquids—electrochemical synthesis and characterization. Surf Interface Anal 42:1271–1275
Fujimura K, Matsui T, Habazaki H, Kawashima A, Kumagai N, Hashimoto K (2000) The durability of manganese-molybdenum oxide anodes for oxygen evolution in seawater electrolysis. Electrochim Acta 45:2297–2303. https://doi.org/10.1016/S0013-4686(00)00316-9
Gao M, Yang C, Zhang Q, Zeng J, Li X, Hua Y, Xu C, Dong P (2017) Facile electrochemical preparation of self-supported porous Ni–Mo alloy microsphere films as efficient bifunctional electrocatalysts for water splitting. J Mater Chem A 5:5797–5805. https://doi.org/10.1039/C6TA10812A
Gong M, Wang D-Y, Chen C-C, Hwang B-J, Dai H (2016) A mini review on nickel-based electrocatalysts for alkaline hydrogen evolution reaction. Nano Res 9(1):28–46. https://doi.org/10.1007/s12274-015-0965-x
González-Buch C, Herraiz-Cardona I, Ortega E, García-Antón J, Pérez-Herranz V (2016) Study of the catalytic activity of 3D macroporous Ni and NiMo cathodes for hydrogen production by alkaline water electrolysis. J Appl Electrochem 46:791–803. https://doi.org/10.1007/s10800-016-0970-0
Grigoriev SA, Fateev VN (2017) Hydrogen production by water electrolysis. In: Sankir M, Sankir ND (eds) Hydrogen production technologies. Wiley, Hoboken. https://doi.org/10.1002/9781119283676.ch6
Han Q, Cui S, Pu N, Chen J, Liu K, Wei X (2010) A study on pulse plating amorphous NieMo alloy coating used as HER cathode in alkaline medium. Int J Hydrogen Energy 35:5194–5201. https://doi.org/10.1016/j.ijhydene.2010.03.093
Holladay JD, Hu J, King DL, Wang Y (2009) An overview of hydrogen production technologies. Catal Today 139:244–260. https://doi.org/10.1016/j.cattod.2008.08.039
Jiang N, Meng H-M, Song L-J, Yu H-Y (2010) Study on Ni–Fe–C cathode for hydrogen evolution from seawater electrolysis. Int J Hydrogen Energy 35:8056–8062. https://doi.org/10.1016/j.ijhydene.2010.01.092
Jović BM, Lačnevac C, Krstajić NV, Jović VD (2014) Service life test of the NiSn coatings as cathodes for hydrogen evolution in industrial chlor-alkali electrolysis. Int J Hydrogen Energy 39(17):8947–8958. https://doi.org/10.1016/j.ijhydene.2014.04.015
Kalamaras CM, Efstathiou AM (2013) Hydrogen production technologies: current state and future developments. Conf Papers Energy Artic 2013:690627. https://doi.org/10.1155/2013/690627
Karunadasa HI, Chang CJ, Long JR (2010) A molecular molybdenum-oxo catalyst for generating hydrogen from water. Nature 464(7293):1329e33
Kato Z, Izumiya K, Kumagai N, Hashimoto K (2009) Energy-saving seawater electrolysis for hydrogen production. J Solid State Electrochem 13:219–224. https://doi.org/10.1007/s10008-008-0548-9
Kubisztal J, Budniok A, Lasia A (2007) Study of the hydrogen evolution reaction on nickel-based composite coatings containing molybdenum powder. Int J Hydrogen Energy 32:1211–1218. https://doi.org/10.1016/j.ijhydene.2006.11.020
Marozzi CA, Chialvo AC (2000) Development of electrode morphologies of interest in electrocatalysis. Part 1: electrodeposited porous nickel electrodes. Electrochim Acta 45:2111–2120. https://doi.org/10.1016/S0013-4686(99),00422-3
Metikos-Hukovic M, Jukic A (2000) Correlation of electronic structure and catalytic activity of Zr–Ni amorphous alloys for the hydrogen evolution reaction. Electrochim Acta 45:4159–4170. https://doi.org/10.1016/S0013-4686(00)00550-8
Navarro-Flores E, Chong Z, Omanovic S (2005) Characterization of Ni, NiMo, NiW and NiFe electroactive coatings as electrocatalysts for hydrogen evolution in an acidic medium. J Mol Catal A: Chem 226:179–197. https://doi.org/10.1016/j.molcata.2004.10.029
Norskov JK, Bligaard T, Logadottir A, Kitchin JR, Chen JG, Pandelov S, Stimming U (2005) Trends in the exchange current for hydrogen evolution. J Electrochem Soc 152(3):J23–J26. https://doi.org/10.1149/1.1856988
Oniciu L, Grunwald E (1980) Galvanotehnica (in Romanian), Ed. Stiintifica si Enciclopedica Bucharest
Petica A, Brincoveanu O, Golgovici F, Manea AC, Enachescu M, Anicai L (2018) Pd surface functionalization of 3D electroformed Ni and Ni-Mo alloy metallic nanofoams for hydrogen production. J Phys 987:012003. https://doi.org/10.1088/1742-6596/987/1/012003
Podlaha EJ, Landolt D (1996a) Induced codeposition. I. An experimental investigation of Ni-Mo alloys. J Electrochem Soc 143:885–892. https://doi.org/10.1149/1.1836553
Podlaha EJ, Landolt D (1996b) Induced codeposition. II. A mathematical model describing the electrodeposition of Ni-Mo alloys. J Electrochem Soc 143:893–899. https://doi.org/10.1149/1.1836554
Prioteasa P, Anicai L, Visan T (2010a) Synthesis and corrosion characterization of electrodeposited Ni-Mo alloys obtained from aqueous solutions. UPB Sci Bull Series B 72:11–24
Prioteasa P, Golgovici F, Sbircea G, Anicai L, Visan T (2010b) Electrodeposition, structure and corrosion behavior of Ni-Mo alloys from citrate aqueous electrolyte. Rev Chim (Bucharest) 61:1046–1053
Sabalova M, Orinakova R, Orinak A, Smoradova I, Kupkova M, Streckova M (2017) Electrocatalytic hydrogen evolution in acidic media using electrodeposited Ag/PPy and Ni/PPy hybrid materials. Chem Pap 71:513–523. https://doi.org/10.1007/s11696-016-0079-2
Safizadeh F, Ghali E, Houlachi G (2015) Electrocatalysis developments for hydrogen evolution reaction in alkaline solutions-A review. Int J Hydrogen Energy 40:256–274. https://doi.org/10.1016/j.ijhydene.2014.10.109
Saitou M (2014) Characterization of electrodeposited Ni and Ni–Mo thin films by X-ray diffraction. Int J Electrochem Sci 9:6033–6042
Sapountzi FM, Gracia JM, Weststrate CJ, Fredriksson HOA, Niemantsverdriet JW (2017) Electrocatalysts for the generation of hydrogen, oxygen and synthesis gas. Prog Energ Combust 58:1–35. https://doi.org/10.1016/j.pecs.2016.09.001
Shetty S, Sadiq MMJ, Bhat DK, Hedge AC (2017) Electrodeposition and characterization of Ni-Mo alloy as an electrocatalyst for alkaline water electrolysis. J Electroanal Chem 796:57–65. https://doi.org/10.1016/j.jelechem.2017.05.002
Shin HC, Dong J, Liu M (2003) Nanoporous structures prepared by an electrochemical deposition process. Adv Mater 15:1610–1614. https://doi.org/10.1002/adma.200305160
Srivastava M, Yoganandan G, William Grips VK (2012) Electrodeposition of Ni and Co coatings from ionic liquid. Surf Eng 28:424–429
Tian J, Cheng N, Liu Q, Sun X, He Y, Asiri AM (2015) Self-supported NiMo hollow nanorods array: an efficient 3D bifunctional catalytic electrode for overall water splitting. J Mater Chem A 3:20056–20059. https://doi.org/10.1039/C5TA04723D
Vijayakumar J, Mohan S, Anand Kumar S, Suseendiran SR, Pavithra S (2013) Electrodeposition of Ni-Co-Sn alloy from choline chloride-based deep eutectic solvent and characterization as cathode for hydrogen evolution in alkaline solution. Int J Hydrogen Energy 38:10208–10214
Villars P, Cenzual K (2007) Pearson’s Crystal Data: Crystal Structure Database for Inorganic Compounds (on CD-ROM), Version 1.0, Release 2007/8, ASM InternationalR, Materials Park,Ohio, USA
Wang H, Huang B, Zhang W, Liu X, Yao S (2014) Electrodeposition of Ni–Mo alloy from deep eutectic solvents and its catalytic properties for hydrogen generation. CIESC J 65(11):4529
Wang M, Wang Z, Yu X, Guo Z (2015) Facile one-step electrodeposition preparation of porous NiMo film as electrocatalyst for hydrogen evolution reaction. Int J Hydrogen Energy 40:2173–2181. https://doi.org/10.1016/j.ijhydene.2014.12.022
Xu C, J-b Zhou, Zeng M, X-l Fu, X-j Liu, J-m Li (2016) Electrodeposition mechanism and characterization of Ni-Mo alloy and its electrocatalytic performance for hydrogen evolution. Int J Hydrogen Energy 41:13341–13349. https://doi.org/10.1016/j.ijhydene.2016.06.205
Zeng K, Zhang DK (2010) Recent progress in alkaline water electrolysis for hydrogen production and applications. Prog Energy Combust Sci 36:307–326
Zheng J (2017) Seawater splitting for high-efficiency hydrogen evolution by alloyed PtNix electrocatalysts. Appl Surf Sci 413:360–365. https://doi.org/10.1016/j.apsusc.2017.03.285
Acknowledgements
Part of this work was supported by the NANOFOAM project, Contract No. 37/2016, M-Era.NET Program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Golgovici, F., Pumnea, A., Petica, A. et al. Ni–Mo alloy nanostructures as cathodic materials for hydrogen evolution reaction during seawater electrolysis. Chem. Pap. 72, 1889–1903 (2018). https://doi.org/10.1007/s11696-018-0486-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-018-0486-7