Abstract
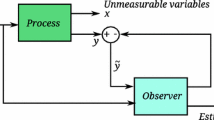
The aim of this paper is to present an alternative state observer structure for online estimation purposes of the key dynamical variables in a class of batch culture for plasmid production; the latter has been extremely attractive to be used as DNA vaccines or gene therapy. A mathematical model for culture of Escherichia coli DH5α-harboring plasmid was considered a benchmark system for the application of the proposed estimation methodology. Local observability analysis revealed that the system is partially observable for plasmid concentration considering only the biomass concentration in the batch culture as the measured variable. The proposed observer is designed with a simple proportional–integral feedback of the measured biomass concentration, where under the proposed design, the observer gain´s array can compensate the main nonlinearities of the estimation error dynamics. The convergence of estimated variables to the real ones is mathematically analyzed, reaching an asymptotic behavior. Numerical experiments were performed, where a comparison with a standard extended Luenberger observer was done and the proposed estimation methodology revealed a satisfactory performance.









Similar content being viewed by others
Abbreviations
- X :
-
Biomass concentration (g/L)
- S :
-
Glucose concentration (g/L)
- G :
-
Glycerol concentration (g/L)
- A :
-
Acetate concentration (g/L)
- P :
-
Plasmid concentration (g/L)
- k S :
-
Affinity constant for glucose (g/L)
- K g :
-
Affinity constant for glycerol (g/L)
- K a :
-
Affinity constant for acetate (g/L)
- K igs :
-
Inhibition constant of growth on glycerol by glucose (g/L)
- K ias :
-
Inhibition constant of growth on acetate by glucose (g/L)
- K isa :
-
Inhibition constant of growth on glucose by acetate (g/L)
- K iga :
-
Inhibition constant of growth on glycerol by acetate (g/L)
- K ixa :
-
Inhibition constant of biomass growth by acetate (g/L)
- K ixp :
-
Inhibition constant of biomass growth by plasmid production (g/L)
- Y Pa/s :
-
Acetate yield on glucose
- Y Pa/g :
-
Acetate yield on glycerol
- Y x/s :
-
Biomass yield on glucose
- Y X/g :
-
Biomass yield on glycerol
- Y P/Xs :
-
Plasmid yield on biomass growth on glucose
- Y P/Xg :
-
Plasmid yield on biomass growth on glycerol
- Y P/Xa :
-
Plasmid yield on biomass growth on acetate
- t :
-
Time (h)
- g 1 :
-
Gain of the observer (g/L)
- g 2 :
-
Gain of the observer (1/h)
- \( \mu_{\text{s}} \) :
-
Specific growth rate on glucose (1/h)
- \( \mu_{\text{g}} \) :
-
Specific growth rate on glycerol (1/h)
- \( \mu_{\text{a}} \) :
-
Specific growth rate on acetate (1/h)
- \( \mu_{{\text{max\,s}}} \) :
-
Maximum specific growth rate on glucose (1/h)
- \( \mu_{{\text{max\,g}}} \) :
-
Maximum specific growth rate on glycerol (1/h)
- \( \mu_{{\text{max\,a}}} \) :
-
Maximum specific growth rate on acetate (1/h)
- \( \xi \) :
-
Estimation error
References
Aguilar-López R, Neria-González MI, Ruiz Camacho B, Rangel E, Santos-Sánchez OJ, López Pérez PA (2017) State estimation based on nonlinear observer for hydrogen production in a photocatalytic anaerobic bioreactor. Int J Chem React Eng. https://doi.org/10.1515/ijcre-2017-0004
Aguilar-López R, Neria-González MI, Martínez-Guerra R, Mata-Machuca JL (2014) Nonlinear estimation in a class of gene transcription process. Appl Math Comput 226:131–144. https://doi.org/10.1016/j.amc.2013
Ali JM, Hoang NH, Hussain MA, Dochain D (2015) Review and classification of recent observers applied in chemical process systems. Comput Chem Eng 76:27–41. https://doi.org/10.1016/j.compchemeng.2015.01.019
Bennemo M, Blom H, Emilsson A, Lemmens R (2009) A chromatographic method for determination of supercoiled plasmid DNA concentration in complex solutions. J Chromatogr B Analyt Technol Biomed Life Sci 877:2530–2536. https://doi.org/10.1016/j.jchromb.2009.06.037
Boker AM, Khalil HK (2013) Nonlinear observers comprising high-gain observers and extended Kalman filters. Automatica 49:3583–3590. https://doi.org/10.1016/j.automatica.2013.08.031
Chen SY (2012) Kalman filter for robot vision: a survey. IEEE Trans Ind Electron 59(11):4409–4420. https://doi.org/10.1109/TIE.2011.2162714
Corriou JP (2018) Nonlinear control of reactors with state estimation. In: Process control. Springer, Cham, pp 769–791. https://doi.org/10.1007/978-3-319-61143-3_19
Didi I, Dib H, Cherki B (2015) A Luenberger-type observer for the AM2 model. J Process Control 32:117–126. https://doi.org/10.1016/j.jprocont.2015.04.010
Dochain D, Chen L (1992) Local observability and controllability of stirred tank reactors. J Proc Control 2:139–144. https://doi.org/10.1016/0959-1524(92)85003-F
Dudret S, Beauchard K, Ammouri F, Rouchon P (2012) Stability and asymptotic observers of binary distillation processes described by nonlinear convection/diffusion models. In: American control conference Fairmont Queen Elizabeth, Montréal, QC, Canada. https://doi.org/10.1109/acc.2012.6315036
Freudenau I, Lutter P, Baier R, Schleef M, Bednarz H, Lara AR, Niehaus K (2015) ColE1-plasmid production in Escherichia coli: mathematical simulation and experimental validation. Front Bioeng Biotechnol 3:127. https://doi.org/10.3389/fbioe.2015.00127.eCollection
Gálvez RM, Pablos TE, Sigala JC, Lara AR (2014) Co-utilization of glucose and xylose increases growth rate without affecting plasmid DNA yield of engineered E. coli. Rev Mex Ing Quim 13(2):387–391. http://www.redalyc.org/pdf/620/62031508003.pdf
Gupta MK, Tomar NK, Bhaumik S (2014) Detectability and observer design for linear descriptor systems. In: 22nd mediterranean conference on control and automation, palermo, pp 1094–1098. https://doi.org/10.1109/med.2014.6961520
Guysinsky M, Hasselblatt B, Rayskin V (2003) Differentiability of the Hartman–Grobman linearization. Discrete Cont Dyn Syst 9(4):979–984. http://emerald.tufts.edu/as/math/Preprints/HasselblattGuysinskyRayskin.pdf
Hoang NH, Couenne F, Le Gorrec Y, Chen CL, Ydstie BE (2013) Passivity-based nonlinear control of CSTR via asymptotic observers. Annu Rev Control 37(2):278–288. https://doi.org/10.1016/j.arcontrol.2013.09.007
Imanaka T, Tanaka T, Tsunekawa H, Aiba S (1981) Cloning of the genes for penicillinase, penP and penI, of Bacillus licheniformis in some vector plasmids and their expression in Escherichia coli, Bacillus subtilis and Bacillus licheniformis. J Bacteriol 147(3):776–786
Jenzsch M, Simutis R, Eisbrenner G, Stückrath I, Lübbert A (2006) Estimation of biomass concentrations in fermentation processes for recombinant protein production. Bioprocess Biosyst Eng 29:19–27. https://doi.org/10.1007/s00449-006-0051-6
Kentzoglanakis K, García López D, Brown SP, Goldstein RA (2013) The evolution of collective restraint: policing and obedience among non-conjugative plasmids. PLoS Comput Biol 9(4):e1003036. https://doi.org/10.1371/journal.pcbi.1003036
Khalil HK, Praly L (2014) High-gain observers in nonlinear feedback control. Int J Robust Nonlinear Control 24:993–1015. https://doi.org/10.1002/rnc.3051
Kiviharju K, Salonen K, Moilanen U, Eerikäinen T (2008) Biomass measurement online: the performance of in situ measurement and software sensors. J Ind Microbiol Biotechnol 35:657–665. https://doi.org/10.1007/s10295-008-0346-5
Klumpp S (2011) Growth-rate dependence reveals design principles of plasmid copy number control. Plos One 6(5):ARTN e20403
Kostov G, Lyubenova V, Shopska V, Petelkov I, Ivanov K, Iliev V, Denkova R, Ignatova M (2015) Software sensors for monitoring the biomass concentration and the kinetics of continuous beer fermentation with immobilized cells. Comptes rendus de l’Académie bulgare des sciences: sciences mathématiques et naturelles 68(11):1439–1448
Krone SM, Lu R, Fox R, Suzuki H, Top EM (2007) Modelling the spatial dynamics of plasmid transfer and persistence. Microbiology-Sgm 153:2803–2816. https://doi.org/10.1099/mic.0.2006/004531-0
Lee J, Ramirez WF (1992) Mathematical modeling of induced foreign protein production by recombinant bacteria. Biotechnol Bioeng 15:635–646. https://doi.org/10.1002/bit.260390608
Lee C, Jaai K, Seung GS, Seokhwan H (2006) Absolute and relative qPCR quantification of plasmid copy number in Escherichia coli. J Biotechnol 123:273–280. https://doi.org/10.1016/j.jbiotec.2005.11.014
Lopes MB, Martins G, Calado CRC (2014) Kinetic modeling of plasmid bioproduction in Escherichia coli DH5α cultures over different carbon sources compositions. J Biotechnol 186:38–48. https://doi.org/10.1016/j.jbiotec.2014.06.022
Lopes MB, Calado CRC, Figueiredo MAT, Bioucas-Dias JM (2017) Does nonlinear modeling play a role in plasmid bioprocess monitoring using fourier transform infrared spectra? Appl Spectrosc 71(6):1148–1156. https://doi.org/10.1177/0003702816670913
López Pérez PA, Maya-Yescas R, Gómez Acata RV, Peña Caballero V, Aguilar López R (2013) Software sensors design for the simultaneous saccharification and fermentation of starch to ethanol. Fuel 110:219–226. https://doi.org/10.1016/j.fuel.2012.11.020
Lyubenova V (2016) Monitoring the kinetics of bioprocess variables–theory and applications. Inf Technol Control 14(1):2–12. https://doi.org/10.1515/itc-2016-0016
Madrid RE, Felice CJ (2005) Microbial biomass estimation. Crit Rev Biotechnol 25:97–112. https://doi.org/10.1080/07388550500248563
Mairet F, Moisan M, Bernard O (2014) Estimation of neutral lipid and carbohydrate quotas in microalgae using adaptive interval observers. Bioprocess Biosyst Eng 37:51–61. https://doi.org/10.1007/s00449-013-0913-7
Martinez-Fonseca N, Chairez I, Poznyak A (2014) Uniform step-by-step observer for aerobic bioreactor based on super-twisting algorithm. Bioprocess Biosyst Eng 37(12):2493–2503. https://doi.org/10.1007/s00449-014-1227-0
Mata-Machuca JL, Martínez-Guerra R, Aguilar-López R (2010) Monitoring in a predator-prey systems via a class of high order observer design. Biosystems 100:65–69. https://doi.org/10.1016/j.biosystems.2010.01.003
Nuñez S, Garelli F, De Battista H (2016) Product-based sliding mode observer for biomass and growth rate estimation in Luedeking–Piret like processes. Chem Eng Sci 105:24–30. https://doi.org/10.1016/j.cherd.2015.10.030
Ponciano JM, De Gelder L, Top EM, Joyce P (2007) The population biology of bacterial plasmids: a Hidden Markov model approach. Genetics 176:957–968. https://doi.org/10.1534/genetics.106.061937
Raïssi T, Efimov D, Zolghadri A (2012) Interval state estimation for a class of nonlinear systems. IEEE Trans Automat Control 57(1):260–265. https://doi.org/10.1109/TAC.2011.2164820
Rodriguez A, Quiroz G, Femat R, Mendez-Acosta HO, de Leon J (2015) An adaptive observer for operation monitoring of anaerobic digestion wastewater treatment. Chem Eng J 269:186–193. https://doi.org/10.1016/j.cej.2015.01.038
Schmidt-Hager J, Ude C, Findeis M, Gernot JT, Scheper T, Sascha B (2014) Noninvasive online biomass detector system for cultivation in shake flasks. Eng Life Sci 14:467–476. https://doi.org/10.1002/elsc.201400026
Singer A, Eiteman MA, Altman E (2009) DNA plasmid production in different host strains of Escherichia coli. J Ind Microbiol Biotechnol 36:521–530. https://doi.org/10.1007/s10295-008-0522-7
Vargas A, Moreno JA, Vande-Wouwer A (2015) Super-twisting estimation of a virtual output for extremum-seeking output feedback control of bioreactors. J Process Control 35:41–49. https://doi.org/10.1016/j.jprocont.2015.08.003
Wunderlich M, Taymaz-Nikerel H, Gosser G, Ramírez OT, Lara AR (2014) Effect of growth rate on plasmid DNA production and metabolic performance of engineered Escherichia coli strains. J Biosci Bioeng 117:336–342. https://doi.org/10.1016/j.jbiosc.2013.08.007
Xenopoulos A, Pattnaik P (2014) Production and purification of plasmid DNA vaccines: is there scope for further innovation? Expert Rev Vaccines 13(12):1537–1551. https://doi.org/10.1586/14760584.2014.968556
Acknowledgements
FGH is grateful with Consejo Nacional de Ciencia y Tecnología (CONACyT, Mexico) for the financial support via a postgraduate scholarship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grijalva-Hernández, F., Caballero, V.P., López-Pérez, P.A. et al. Estimation of plasmid concentration in batch culture of Escherichia coli DH5α via simple state observer. Chem. Pap. 72, 2589–2598 (2018). https://doi.org/10.1007/s11696-018-0478-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-018-0478-7