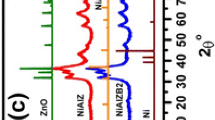
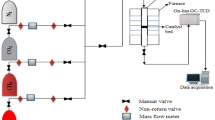
Abstract
The catalytic hydrogenation of carbon dioxide is known for over 100 years. In recent years, the importance of this reaction has significantly increased due to synthetic natural gas production (SNG). Synthetic natural gas is produced from renewable energy sources (such as wind, hydro or solar power). Excess electric energy can be used for the production of hydrogen via electrolysis. This work focuses on the synthesis and characterization of a laboratory prepared nickel-based catalyst. This catalytic activity is compared to that of a commercial nickel-based catalyst. This manuscript also describes the methanation pilot unit where the catalytic tests took place. Energy is supplied to the pilot unit from a small photovoltaic power plant. Catalytic tests were performed at different pressures and temperatures.







Similar content being viewed by others
References
Aksoylu AE, Isli A, Zi Onsan (1999) Interaction between nickel and molybdenum in Ni–Mo/Al2O3 catalysts: effect of impregnation strategy. Appl Catal A Gen 183:357–364. https://doi.org/10.1016/S0926-860X(99)00075-7
Ananikov VP (2015) Nickel: the “Spirited Horse” of transition metal catalysis. ACS Catal 5:1964–1971. https://doi.org/10.1021/acscatal.5b00072
Atkins P, Julio P (2006) Atkin’s physical chemistry, 8th edn. Freeman WH and Company, Oxford, pp 210–211
Aziz MAA, Jalil AA, Triwagyono S, Mukti RR, Taufiq-Yap YH, Sazegar MR (2014) Highly active Ni-promoted mesostructured silica nanoparticles for CO2 methanation. Appl Catal B Environ 147:359–368. https://doi.org/10.1016/j.apcatb.2013.09.015
Aziz MAA, Jalil AA, Triwagyono S, Ahmed A (2015) CO2 methanation over heterogeneous catalyst recent progress and future prospect. Green Chem 17:2647–2663. https://doi.org/10.1039/C5GC00119F
Bartholomew CH (2001) Mechanisms of catalyst deactivation. Appl Catal A Gen 212:17–60. https://doi.org/10.1016/S0926-860X(00)00843-7
Brooks KP, Hu L, Zhu H, Kee RJ (2007) Methanation of carbon dioxide by hydrogen reduction using the Sabatier process in microchannel reactors. Chem Eng Sci 62:1161–1170. https://doi.org/10.1016/j.ces.2006.11.020
Buerkhardt M, Busch G (2013) Methanation of hydrogen and carbon dioxide. Appl Energy 111:74–79. https://doi.org/10.1016/j.apenergy.2013.04.080
Chen M, Guo Z, Zheng J, Jing F, Chu W (2016) CO2 selective hydrogenation to synthetic natural gas (SNG) over four nano-sized Ni/ZrO2 samples: ZrO2 crystalline phase and treatment impact. J Energy Chem 25:1070–1077. https://doi.org/10.1016/j.jechem.2016.11.008
Civan F (2004) Natural gas transportation and storage. Elsevier, Amsterdam, pp 273–282
Darensbourg DJ, Holtcamp MW, Struck GE, Zimmer MS, Niezgoda SA, Rainey P, Robertson JB, Draper JD, Reibenspies JH (1998) Catalytic activity of a series of Zn (II) phenoxides for the copolymerization of epoxides and carbon dioxide. J Am Chem Soc 121:107–116. https://doi.org/10.1021/ja9826284
Dry ME (2002) The Fischer–Tropsch process: 1950–2000. Catal Today 71:227–241. https://doi.org/10.1016/S09250-5861(01)00453-9
Falconer JL, Zağli AE (1980) Adsorption and methanation of carbon dioxide on a nickel/silica catalyst. J Catal 62:280–285. https://doi.org/10.1016/0021-9517(80)90456-X
Gao J, Wang Y, Ping Y, Hu D, Xu G, Gu F, Su F (2012) A thermodynamic analysis of methanation reactions of carbon oxides for the production of synthetic natural gas. RSC Adv 2:2358–2368. https://doi.org/10.1039/C2RA00632D
Gao J, Liu Q, Gu F, Liu B, Zhong Z, Su F (2015) Recent advances in methanation catalysts for the production of synthetic natural gas. RSC Adv 5:22759–22776. https://doi.org/10.1039/C4RA16114A
Garbarino G, Riani P, Magistri L, Busca G (2014) A study of the methanation of carbon dioxide on Ni/Al2O3 catalysts at atmospheric pressure. Int J Hydrog Energy 39:11557–11565. https://doi.org/10.1016/j.ijhydene.2014.05.111
Graça I, Gonzáles LV, Bacariza MC, Frenandes A, Henriques C, Lopes JM, Riberio MF (2014) CO2 hydrogenation into CH4 on NiHNaUSY zeolites. Appl Catal B Environ 147:101–110. https://doi.org/10.1016/j.apcatb.2013.08.010
Green Car Congress (2013) Audi opens power-to-gas facility in Werlte/Emsland; e-gas from water, green electricity and CO2. Retrieved October 3, 2016 from http://www.greencarcongress.com/2013/06/audi-20130625.html
Hou PY, Wise H (1985) Kinetic studies with a sulphur-tolerant methanation catalyst. J Catal 93:409–416. https://doi.org/10.1016/0021-9517(85)90188-5
International Energy Agency (2014) World Energy Outlook 2014. Executive summary. Retrieved October 2, 2016 from http://www.iea.org/Textbase/npsum/WEO2014SUM.pdf
Jasquemin M, Beuls A, Ruiz P (2010) Catalytic production of methane from CO2 and H2 at low temperature: insight on the reaction mechanism. Appl Catal 157:462–466. https://doi.org/10.1016/j.cattod.2010.06.016
Lin C, Wang H, Li Z, Wang B, Ma X, Qin S, Sun Q (2012) Effect of a promotore on the methanation activity of Mo-based sulphur-resistant catalyst. Front Chem Sci Eng 7:88–94. https://doi.org/10.1007/s11705-013-301-1
Mills GA, Steffgen FW (1974) Catalytic methanation. Catal Rev 8:159–210. https://doi.org/10.1080/01614947408071860
Moore CL (2010) Technology development for human exploration of Mars. Acta Astronaut 67:1170–1175. https://doi.org/10.1016/j.actaastro.2010/06.031
Ross JRH (2012) Heterogenous catalysis: fundamentals and applications. Elsevier, Amsterdam, pp 65–96
Sabatier P, Senderens JB (1902) New synthesis of methane. Compt Rend 134:514–516
Saheldelfar S, Takht RM (2015) Carbon dioxide utilization for methane production: a thermodynamic analysis. J Pet Sci Eng 134:14–22. https://doi.org/10.1016/j.petrol.2017.07.015
Sato S, Arai T, Morikawa T, Uemura K, Suzuki TM, Tanaka H, Kajino T (2011) Selective CO2 conversion to formate conjugated with H2O oxidation utilizing semiconductor/complex hybrid photocatalysts. J Am Chem Soc 133:15240–15243. https://doi.org/10.1021/ja204881d
Schaaf T, Grüning J, Schuster MR, Rothenfluh T, Orth A (2014) Methanation of CO2-storage of renewable energy in a gas distribution system. Energy Sustain Soc 4:1–14. https://doi.org/10.1186/s13705-014-0029-1
Schlesinger MD, Demeter JJ, Greyson M (1956) Catalyst for producing methane from hydrogen and carbon monoxide. Ind Eng Chem Res 48:68–70. https://doi.org/10.1021/ie50553a022
Sing KSW (1997) Physical adsorption: experimental, theory and applications. Kluwer Academic Publishers, Dordrecht, pp 9–16
Šnajdrová V, Hlinčík T, Baraj E (2016) Power-to-gas. Gas 2:35–40
Šnajdrová V, Hlinčík T, Jílková L, Vrbová V, Ciahotný K (2017) Syntéza katalyzátorů pro methanizační reakci. Paliva 4:99–104
Solymosi F, Erdöhelyi A, Kocis M (1981) Methanation of CO2 on supported Ru catalysts. J Chem Soc 77:1003–1012. https://doi.org/10.1039/F19817701003
Sterner M (2009) Bioenergy and renewable power methane in integrated 100% renewable energy systems: limiting global warming by transforming energy systems. Kassel University Press, Kassel
Tada S, Schimizu T, Kameyama H, Haneda T, Kikuchi R (2012) Ni/CeO2 catalysts with high CO2 methanation activity and high CH4 selectivity at low temperatures. Int J Hydrog Energy 37:5527–5531. https://doi.org/10.1016/j.ijhydene.2011.12.122
Trueba M, Trassati SP (2005) γ-Alumina as a support for catalysts: a review of fundamental aspects. Eur J Inorg Chem 17:3393–3403. https://doi.org/10.1002/ejic.200500348
Tsuji M, Kodama T, Yoshida T, Kitayama Y, Tamaura Y (1996) Preparation and CO2 methanation activity of an ultrafine Ni (II) ferrite catalyst. J Catal 164:315–321. https://doi.org/10.1006/j.cat.1996.0387
Vance CK, Bartholomew CH (1983) Hydrogenation of carbon dioxide on group viii metals: effects of support on activity/selectivity and adsorption properties of nickel. Appl Catal 7:169–177. https://doi.org/10.1016/0166-9834(83)80005-0
Vannice MA (1976) The catalytic synthesis of hydrocarbons from carbon monoxide and hydrogen. Catal Rev 14:153–191. https://doi.org/10.1080/03602457608073410
Wang W, Gang J (2011) Methanation of carbon dioxide: an overview. Front Chem Sci Eng 5:2–10. https://doi.org/10.1007/s11705-010-0528-3
Wang BW, Yao YQ, Liu SH, Hu ZY, Li ZH, Ma XB (2015) Effects of MoO3 loading and calcination temperature on the catalytic performance of MoO3/CeO2 toward sulfur-resistant methanation. Fuel Sci Technol 138:263–270. https://doi.org/10.1016/j.fuproc.2015.06.009
Wang W, Chu W, Wang N, Yang W, Jiang CH (2016) Mesoporous nickel catalyst supported on multi-walled carbon nanotubes for carbon dioxide methanation. Int J Hydrog Energy 41:967–975. https://doi.org/10.1016/j.ijhydene.2015.11.133
Wentrcek PR, Wood BJ, Wise H (1976) The role of surface carbon in catalytic methanation. J Catal 43:363–366. https://doi.org/10.1016/0021-9517(76)90324-9
Yamaguchi T (1994) Application of ZrO2 as a catalyst and a catalyst support. Catal Today 20:199–214. https://doi.org/10.1016/0920-5861(94)80003-0
Yu KMK, Curcic I, Gabriel J, Tsang SCE (2008) Recent advances in CO2 capture and utilization. Chem Sus Chem 1:893–899. https://doi.org/10.1002/CSSC.200800169
Zheng J, Wang Ch, Chu W, Zhou Y, Köhler K (2016) CO2 methanation over supported Ru/Al2O3 catalysts: mechanistic studies by in situ infrared spectroscopy. ChemistrySelect 1:3197–3203. https://doi.org/10.1002/slct.201600651
Acknowledgements
The presented work was financially supported by TACR-Epsilon Project TH02020767 and IGA VSCHT Project A2_FTOP_2017_027.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Šnajdrová, V., Hlinčík, T., Ciahotný, K. et al. Pilot unit of carbon dioxide methanation using nickel-based catalysts. Chem. Pap. 72, 2339–2346 (2018). https://doi.org/10.1007/s11696-018-0456-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-018-0456-0