Abstract
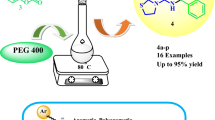
A novel and effective route for the synthesis of 2-monosubstituted pyrido[2,3-d]pyrimidines is reported starting from the corresponding 2-halogeno- and 2-methylsulfanyl-pyrido[2,3-d]pyrimidine through a palladium-catalyzed cross-coupling has been described. The reaction involved original halogen discrimination under visible light as an alternative source of energy. Alternatively, monoarylated compounds were elaborated from 2-halogeno pyrido[2,3-d]pyrimidine by C2 chlorine further displacement and from 2-methylsulfanyl-pyrido[2,3-d]pyrimidine by C-alkylsulfanyl release. We have described the first synthesis of mono substituted pyrido[2,3-d]pyrimidine series using a two strategies using visible light as a powerful synthetic tool for the preparation of focused libraries of biologically active compounds. The use of available starting materials, easy reaction conditions and catalytic processes combined with high yields of products are the main outcome of this method.
Graphical abstract




Similar content being viewed by others
References
Agarwal A, Ashutosh R, Goyal N, Chauhan PMS, Gupta S (2005) Dihydropyrido[2,3-d]pyrimidines as a new class of antileishmanial agents. Bioorg Med Chem 13:6678–6684. https://doi.org/10.1016/j.bmc.2005.07.043
Beatty JW, Stephenson CRJ (2015) Amine functionalization via oxidative photoredox catalysis: methodology development and complex molecule synthesis. Acc Chem Res 48:1474–1484. https://doi.org/10.1021/acs.accounts.5b00068
Bennett LR, Blankley CJ, Fleming RW, Smith RD, Tessman DK (1981) Antihypertensive activity of 6-arylpyrido[2,3-d]pyrimidin-7-amine derivatives. J Med Chem 24:382–389. https://doi.org/10.1021/jm00357a015
Bulicz J, Daniela CG, Bertarelli DCG, Baumert D, Fülle F, Christa EM, Heber D (2006) Synthesis and pharmacology of pyrido[2,3-d]pyrimidinediones bearing polar substituents as adenosine receptor antagonists. Bioorg Med Chem 14:2837–2849. https://doi.org/10.1016/j.bmc.2005.12.008
Chen JR, Hu XQ, Lu LQ, Xiao WJ (2016) Visible light photoredox-controlled reactions of N-radicals and radical ions. Chem Soc Rev 45:2044–2056. https://doi.org/10.1039/C5CS00655D
Degraw JI, Christie PH, Colwell WT, Sirotnak FM (1992) Synthesis and antifolate properties of 5,10-ethano-5,10-dideazaaminopterin. J Med Chem 35:320–324. https://doi.org/10.1021/jm00080a017
Duda ML, Michael FE (2013) Palladium-catalyzed cross-coupling of N-sulfonylaziridines with boronic acids. J Am Chem Soc 135:18347–18349. https://doi.org/10.1021/ja410686v
Fares M, Abou-Seri SM, Abdel-Aziz HA, Abbas SE, Youssef MM, Eladwy RA (2014) Synthesis and antitumor activity of pyrido[2,3-d]pyrimidine and pyrido[2,3-d][1,2,4]triazolo[4,3-a]pyrimidine derivatives that induce apoptosis through G1 cell-cycle arrest. Eur J Med Chem 83:155–166. https://doi.org/10.1016/j.ejmech.2014.06.027
Font M, González Á, Palop JA, Sanmartín C (2011) New insights into the structural requirements for pro-apoptotic agents based on 2,4-diaminoquinazoline, 2,4-diaminopyrido[2,3-d]pyrimidine and 2,4-diaminopyrimidine derivatives. Eur J Med Chem 46:3887–3899. https://doi.org/10.1016/j.ejmech.2011.05.060
Ghilsoo N, Cheol MY, Euikyung K, Chung KR, Joong HK, Jung HS, Sung HK (2001) Syntheses and evaluation of pyrido[2,3-d]pyrimidine-2,4-diones as PDE 4 inhibitors. Bioorg Med Chem Lett 11:611–614. https://doi.org/10.1016/S0960-894X(00),00681-8
Grivsky EM, Lee S, Sigel CW, Duch DS, Nichol CA (1980) Synthesis and antitumor activity of 2,4-diamino-6-(2,5-dimethoxybenzyl)-5-methylpyrido[2,3-d]pyrimidine. J Med Chem 23:327–329. https://doi.org/10.1021/jm00177a025
Kalinin VN (1992) Carbon-carbon bond formation in heterocycles using Ni- and Pd-catalyzed reactions. Synthesis. https://doi.org/10.1055/s-1992-26125
Liu KK, Huang X, Bagrodia S, Chen JH, Greasley S, Cheng H, Sun S, Knighton D, Rodgers C, Rafidi K, Zou A, Xiao J, Yan S (2011) Quinazolines with intra-molecular hydrogen bonding scaffold (iMHBS) as PI3K/mTOR dual inhibitors. Bioorg Med Chem Lett 21:1270–1274. https://doi.org/10.1016/j.bmcl.2010.12.026
Liu B, Lim CH, Miyake GM (2017) Visible-light-promoted C–S cross-coupling via intermolecular charge transfer. J Am Chem Soc 139:13616–13619. https://doi.org/10.1021/jacs.7b07390
Luo K, Chen YZ, Yang WC, Zhu J, Wu L (2016) Cross-coupling hydrogen evolution by visible light photocatalysis toward C(sp2)–P formation: metal-free C–H functionalization of thiazole derivatives with diarylphosphine oxides. Org Lett 18:452–455. https://doi.org/10.1021/acs.orglett.5b03497
Mahmoud MR, El-Bordany EAA, Hassan NF, Abu El-Azm FSM (2007) Utility of nitriles in synthesis of pyrido[2,3-d]pyrimidines, thiazolo[3,2-a]pyridines, pyrano[2,3-b]benzopyrrole, and Pyrido[2,3-d]benzopyrroles. Phosphorus Sulfur Silicon Relat Elem 182:2507–2521. https://doi.org/10.1080/10426500701506465
Maingot L, Dehbi O, Buron F, Aadil M, Akssira M, Routier S, Guillaumet G (2012) Regioselective syntheses of 2,7-(Het)arylpyrido[2,3-d]pyrimidines by an orthogonal cross-coupling strategy. Synlett 23:2449–2452. https://doi.org/10.1055/s-0032-1317180
Miller JR, Dunham S, Mochalkin I, Banotai C, Bowman M, Buist S, Dunkle B, Hanna D, Harwood HJ, Huband MD, Karnovsky A, Kuhn M, Limberakis C, Liu JY, Mehrens S, Mueller WT, Narasimhan L, Ogden A, Ohren J, Prasad JV, Shelly JA, Skerlos L, Sulavik M, Thomas VH, Vanderroest S, Wang L, Wang Z, Whitton A, Zhu T, Stover CK (2009) A class of selective antibacterials derived from a protein kinase inhibitor pharmacophore. Proc Natl Acad Sci USA 106:1737–1742. https://doi.org/10.1073/pnas.0811275106
Miller DC, Choi GJ, Orbe HS, Knowles RR (2015) Catalytic olefin hydroamidation enabled by proton-coupled electron transfer. J Am Chem Soc 137:13492–13495. https://doi.org/10.1021/jacs.5b09671
Monge A, Martinez-Merino V, Sanmartin C, Fernandez FJ, Ochoa MC, Berllver C, Artigas P, Fernandez-Alvarez E (1989) 2-Arylamino-4-oxo-3,4-dihydropyrido[2,3-d]pyrimidines: synthesis and diuretic activity. Euro J Med Chem 24:209–216. https://doi.org/10.1016/0223-5234(89),90001-9
Nofal ZM, Fahmy HH, Zarea ES, El-Eraky W (2011) Novel benzimidazole derivatives as expected anticancer agents. Acta Pol Pharm 68:519–534
Parks EL, Sandford G, Christopher JA, Miller DD (2008) Perhalogenated pyrimidine scaffolds. Reactions of 5-chloro-2,4,6-trifluoropyrimidine with nitrogen centred nucleophiles. Beilstein J Org Chem 4:22–28. https://doi.org/10.3762/bjoc.4.22
Peng Z, Hu G, Qiao H, Xu P, Gao Y, Zhao Y (2014) Palladium-catalyzed suzuki cross-coupling of arylhydrazines via C–N bond cleavage. J Org Chem 79:2733–2738. https://doi.org/10.1021/jo500026g
Pouliot M, Lefranc DGCF, Quaranta L, Lamberth C, Srinivas N (2011) Novel microbicides. PCT Int Appl WO 2011104183, Chem Abstr 155:380363
Riadi Y, Massip S, Leger JM, Jarry C, Lazar S, Guillaumet G (2012) Convenient synthesis of 2,4-disubstituted pyrido[2,3-d]pyrimidines via regioselective palladium-catalyzed reactions. Tetrahedron 68:5018–5024. https://doi.org/10.1016/j.tet.2012.04.051
Riadi Y, Geesi M, Dehbi O, Bakht MA, Alshammari M, Viaud-Massuarde M-C (2017) Novel animal-bone-meal-supported palladium as a green and efficient catalyst for Suzuki coupling reaction in water, under sunlight. Green Chem Lett Rev 10:101–106. doi:10.1080/17518253.2017.1300687
Ribble W, Hill WE, Ochsner UA, Jarvis TC, Guiles JW, Janjic N, Bullard JM (2010) Discovery and analysis of 4H-pyridopyrimidines, a class of selective bacterial protein synthesis inhibitors. Antimicrob Agents Chemother 54:4648–4657. https://doi.org/10.1128/AAC.00638-10
Rosowsky A, Mota CE, Queener SF (1995) Synthesis and antifolate activity of 2,4-diamino-5,6,7,8-tetrahydropyrido[4,3-d]pyrimidine analogues of trimetrexate and piritrexim. J Heterocyclic Chem 32:335–340. https://doi.org/10.1002/jhet.5570320155
Sako M, Yamamoto Y (2004) Science of synthesis: Houben–Weyl methods of molecular transformations. Thieme, Stuttgart, New York 16:1269–1290
Saurat T, Buron F, Rodrigues N, de Tauzia ML, Colliandre L, Bourg S, Bonnet P, Guillaumet G, Akssira M, Corlu A, Guillouzo C, Berthier P, Rio P, Jourdan ML, Bénédetti H, Routier S (2014) Design, synthesis, and biological activity of pyridopyrimidine scaffolds as novel PI3K/mTOR dual inhibitors. J Med Chem 57:613–631. https://doi.org/10.1021/jm401138v
Schröter S, Stock C, Bach T (2005) Regioselective cross-coupling reactions of multiple halogenated nitrogen-, oxygen-, and sulfur-containing heterocycles. Tetrahedron 61:2245–2267. https://doi.org/10.1016/j.tet.2004.11.074
Shaw MH, Shurtleff VW, Terrett JA, Cuthbertson JD, MacMillan DWC (2016) Native functionality in triple catalytic cross-coupling: sp3 C–H bonds as latent nucleophiles. Science 352:1304–1308. https://doi.org/10.1126/science.aaf6635
Singh G, Singh G, Yadav AK, Mishra AK (2002) Synthesis and antimicrobial evaluation of some new pyrido[2,3-d]pyrimidines and their ribofuranosides. Indian J. Chem Sect B Org Chem Incl Med Chem 41:430–432
Taylor EC, Palmer DC, George TJ, Fletcher SR, Tseng CP, Harrington PJ, Beardsley GP (1983) Synthesis and biological activity of l-5-deazafolic acid and l-deazaaminopterin: synthetic strategies to 5-deazapteridines. J Org Chem 48:4852–4860. https://doi.org/10.1021/jo00173a014
Theeramunkong S, Caldarelli A, Massarotti A, Aprile S, Caprioglio D, Zaninetti R, Teruggi A, Pirali T, Grosa G, Tron GC, Genazzani AA (2011) Regioselective Suzuki coupling of dihaloheteroaromatic compounds as a rapid strategy to synthesize potent rigid combretastatin analogues. J Med Chem 54:4977–4986. https://doi.org/10.1021/jm200555r
Wei L, Malhotra SV (2012) Synthesis and cytotoxicity evaluation of novel pyrido[3,4-d]pyrimidine derivatives as potential anticancer agents. Med Chem Comm 10:1250–1257. https://doi.org/10.1039/C2MD20097
Xuan J, Zeng TT, Feng ZJ, Deng QH, Chen JR, Lu LQ, Xiao WJ, Alper H (2015) Redox-neutral α-allylation of amines by combining palladium catalysis and visible-light photoredox catalysis. Angew Chem Int Ed 54:1625–1628. https://doi.org/10.1002/anie.201409999
Acknowledgements
The author wishes to express the gratitude to the staff of Prince Sattam Bin Abdulaziz University for providing necessary facility to carry out the research work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Riadi, Y., Geesi, M. Photochemical route for the synthesis of novel 2-monosubstituted pyrido[2,3-d]pyrimidines by palladium-catalyzed cross-coupling reactions. Chem. Pap. 72, 697–701 (2018). https://doi.org/10.1007/s11696-017-0325-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-017-0325-2