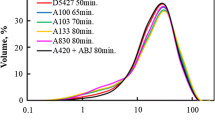
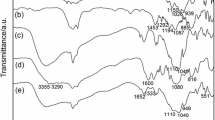
Abstract
The goal of this article is focused on the preparation and the basic characterization of heterogeneous weak acid cation exchange membranes. The six weak cation exchange resins were chosen for the preparation of the membranes. The strong acid cation exchange membrane was prepared for the comparison of properties and preparation parameters. Heterogeneous membranes were prepared in the several steps. The last operations were the extrusion and hydraulic hot-pressing. The torque and pressure in the extrusion head were observed while the extrusion of heterogeneous membranes. According to the type of the ion exchange resin, parameters varied greatly. The strong acid cation exchange membrane and weak acid cation exchange membranes with carboxylic groups had the lowest values of the torque and pressure while the homogenization and extrusion. All membranes were characterized. Electrochemical (permselectivity, ion exchange capacity, resistance), physical (relative water content, swelling in the different solutions), and mechanical properties were measured. Properties varied distinctly according to the type of the ion exchange resins. Swelling dimensional changes of ion exchange membranes in sodium hydroxide and water were bigger than changes in hydrochloric acid. Ion exchange capacity of membranes related with ion exchange capacity of used ion exchange resins. Weak acid cation exchange membranes with carboxylic functional groups compared to the strong membrane had the best specific and areal resistance.








Similar content being viewed by others
Abbreviations
- A :
-
Activities of anions or cations in the solution, mol dm−3
- F:
-
Faraday’s constant, 96,485 °C mol−1
- I :
-
Current, A
- IEC:
-
Ion exchange capacity, meq/g or eq/l
- P :
-
Permselectivity, %
- R :
-
Gas constant, 8.314 J K−1 mol−1
- R A :
-
Areal resistance, Ω cm2
- R s :
-
Specific resistance, Ω cm
- S :
-
Active area, cm2
- T :
-
Temperature, K
- Th:
-
Thickness, cm
- U :
-
Potential, V
- Δlg:
-
Dimensional changes in length, %
- Δth:
-
Dimensional changes in thickness, %
- Δwd:
-
Dimensional changes in width, %
- A:
-
Anion
- C:
-
Cation
- IEM:
-
Ion exchange membrane
- Meas.:
-
Measured
- Sol:
-
Solution
- Theor.:
-
Theoretical
References
Agel E, Bouet J, Fauvarque JF (2001) Characterization and use of anionic membranes for alkaline fuel cells. J Power Sources 101:267–274. doi:10.1016/S0378-7753(01)00759-5
Bohling JCH, Lundquist EG (2012) Preparation of weak acid cation exchange resins. EP1266690 B2. March 21, 2012
Bulejko P, Stránská E, Weinertová K (2017) Electrochemical and mechanical stability of ion-exchange membranes in alkaline solution. Chem Pap 71(7):1303–1309. doi:10.1007/s11696-016-0122-3
Chen GQ, Eschbach FII, Weeks M, Gras SL, Kentish SE (2016) Removal of lactic acid from acid whey using electrodialysis. Sep Purif Technol 158:230–237. doi:10.1016/j.seppur.2015.12.016
Cui W, Kerres J, Eigenberge G (1998) Development and characterization of ion-exchange polymer blend membranes. Sep Purif Technol 14:145–154. doi:10.1016/S1383
Demisch HU, Pusch W (1979) Electric and electrokinetic transport properties of homogeneous weak ion exchange membranes. J Colloid Interface Sci 69(2):247–270. doi:10.1016/0021-9797(79)90154-1
Długołęcki P, Ogonowski P, Metz SJ, Saakes M, Nijmeijer K, Wessling M (2010) On the resistances of membrane, diffusion boundary layer and double layer in ion exchange membrane transport. J Membr Sci 349:369–379. doi:10.1016/j.memsci.2009.11.069
Gohil GS, Shahi VK, Rangarajan RJ (2004) Comparative studies on electrochemical characterization of homogeneous and heterogeneous type of ion-exchange membrane. J Membr Sci 240:211–219. doi:10.1016/j.memsci.2004.04.022
Herrada RA, Medel A, Manríquez F, Siréz I, Bustos E (2016) Preparation of IrO2-Ta2O5|Ti electrodes by immersion, painting and electrophoretic deposition for the electrochemical removal of hydrocarbons from water. J Hazard Mater 319:102–110. doi:10.1016/j.jhazmat.2016.02.076
Jelínek L (2008) Desalinační a separační metody v úpravě vody. Vydavatelství VŠCHT, Praha. ISBN 978-80-7080-705-7
Ji ZY, Chen QB, Yuan JS, Liu J, Zhao YY, Feng WX (2017) Preliminary study on recovering lithium from high Mg2+/Li+ ratio brines by electrodialysis. Sep Purif Technol 172:168–177. doi:10.1016/j.seppur.2016.08.006
Křivčík J, Vladařová J, Hadrava J, Černín A, Brožová L (2012) The effect of an organic ion-exchange resin on properties of heterogeneous ion-exchange membranes. Desalin Water Treat 14:179–184. doi:10.5004/dwt.2010.1025
Křivčík J, Neděla D, Weinertová K, Stránská E (2017) Non-laminated ion-exchange membranes. Desal Water Treat 75:368–375. doi:10.5004/dwt.2017.20592
Kunin R, Barry RE (1949) Carboxylic, weak acid type, cation exchange resin. Ind Eng Chem 41(6):1269–1272. doi:10.1021/ie50474a028
Lasanta C, Gómez J (2012) Tartrate stabilization of wines. Trends Food Sci Technol 28(1):52–59. doi:10.1016/j.tifs.2012.06.005
Lopez AM, Hestekin JA (2015) Improved organic acid purification through wafer enhanced electrodeionization utilizing ionic liquids. J Membr Sci 493:200–205. doi:10.1016/j.memsci.2015.06.008
Luiz A, McClure DD, Lim K, Leslie G, Coster HGL, Barton GW, Kavanagh JM (2017) Potential upgrading of bio-refinery streams by electrodialysis. Desalination 415:20–28. doi:10.1016/j.desal.2017.02.023
Meyers P (ResinTech Inc.) Application of weak acid cation resin in waste treatment. Presented at the AESF Conference, June 1999
Nayar KG, Sundararaman P, O’Connor CL, Schacherl JD, Heath ML, Orozco MG, Shah SR, Wright NC (2017) Feasibility study of an electrodialysis system for in-home water desalination in urban India. Dev Eng 2:38–46. doi:10.1016/j.deveng.2016.12.001
Nojavan S, Zare K, Mohammadi K (2017) Application of fuel cell and electrolyzer as hydrogen energy storage system in energy management of electricity energy retailer in the presence of the renewable energy sources and plug. Energy Convers Manag 136:404–417. doi:10.1016/j.enconman.2017.01.017
Oren Y, Freger V, Linder C (2004) Highly conductive ordered heterogeneous ion-exchange membranes. J Membr Sci 239:17–26. doi:10.1016/j.memsci.2003.12.031
Pesavento M, Biesuz R (1997) Sorption of divalent metal ions on an iminodiacetic resin from artificial seawater. Anal Chim Acta 346(3):381–391. doi:10.1016/S0003
Pesavento M, Biesuz R, Cortina JL (1994) Sorption of metal ions on a weak acid cation exchange resin containing carboxylic groups. Anal Chim Acta 298(2):225–232. doi:10.1016/0003
Sata T (2004) Ion exchange membranes: preparation, characterization, modification and application. The Royal Society of Chemistry, Cambridge
Serre E, Rozoy E, Pedneault C, Lacour S, Bazinet L (2016) Deacidification of cranberry juice by electrodialysis: impact of membrane types and configurations on acid migration and juice physicochemical characteristics. Sep Purif Technol 163:228–237. doi:10.1016/j.seppur.2016.02.044
Slagt M (2015) Ion exchange resin selection. Dow Water & Process Solutions. http://www.soci.org
Stránská E (2015) Relationships between transport and physical-mechanical properties of ion exchange membranes. Desalin Water Treat 56(1–2):3220–3227. doi:10.1080/19443994.2014.981413
Suarez G, Nguyen NTK, Rendtorff NM, Sakka Y, Uchikoshi K (2016) Electrophoretic deposition for obtaining dense lanthanum silicate oxyapatite (LSO). Ceram Int 42(16):19283–19288. doi:10.1016/j.ceramint.2016.09.095
Sun X, Lu H, Wang J (2016) Brackish water desalination using electrodeionization reversal. Chem Eng Process Process Intensif 104:262–270. doi:10.1016/j.cep.2016.03.014
Sun X, Lu H, Wang J (2017) Recovery of citric acid from fermented liquid by bipolar membrane electrodialysis. J Clean Prod 143:250–256. doi:10.1016/j.jclepro.2016.12.118
Tavares T, Malcata FX (2016) Whey and whey powders, principles and applications of dialysis. Encycl Food Health. doi:10.1016/B978-0-12-384947-2.00750-9 (Elsevier)
WaterTech (2004) Role of weak acid cation resin in water treatment. No. 2 March 2004. http://www.ionindia.com/pdf/water_tech/watertech_mar%2004.pdf
Zaganiaris EJ (2013) Ion exchange resins and adsorbents in chemical processing. Book on Demand GmbH, Paris
Zainol Z, Nicol MJ (2009a) Comparative study of chelating ion exchange resins for the recovery of nickel and cobalt from laterite leach tailings. Hydrometallurgy 96(4):283–287. doi:10.1016/j.hydromet.2008.11.005
Zainol Z, Nicol MJ (2009b) Ion-exchange equilibria of Ni2+, Co2+, Mn2+ and Mg2+ with iminodiacetic acid chelating resin Amberlite IRC 748. Hydrometallurgy 99(3):175–180. doi:10.1016/j.hydromet.2009.08.004
Zhang Y, Wang L, Xuan S, Lin X, Luo X (2014) Variable effects on electrodeionization for removal of Cs ions from simulated wastewater. Desalination 344:212–218. doi:10.1016/j.desal.2014.03.034
Zhang W, Miao M, Pan J, Sotto A, Shen J, Gao C, der Bruggen BV (2017) Separation of divalent ions from seawater concentrate to enhance the purity of coarse salt by electrodialysis with monovalent-selective membranes. Desalination 411:28–37. doi:10.1016/j.desal.2017.02.008
Acknowledgements
The work was carried out within the framework of project No. LO1418 “Progressive development of Membrane Innovation Centre” supported by the program NPU I Ministry of Education Youth and Sports of the Czech Republic, using the infrastructure of the Membrane Innovation Centre.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stránská, E., Weinertová, K., Neděla, D. et al. Preparation and basic characterization of heterogeneous weak acid cation exchange membrane. Chem. Pap. 72, 89–98 (2018). https://doi.org/10.1007/s11696-017-0260-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-017-0260-2