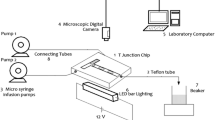
Abstract
This research has been conducted to study extraction of an anionic dye, Alizarin Red S (ARS), from the aqueous phase into the organic phase in a T-junction microchannel. The organic phase included Aliquat 336 and 1-octanol. Equal volumetric flow rates of aqueous and organic phases were adjusted in all the experiments. Designing the experiments and analyzing of the parameters that affect the extraction percentage of ARS were carried out using response surface methodology. The parameters were feed pH, feed concentration, extractant concentration and flow rate of aqueous and organic phases. The maximum extraction percentage of 98.7 was obtained at the feed pH of 3, feed concentration of 5000 mg L−1, extractant concentration of 4 vol.% and flow rate of 2.5 mL min−1. Under the optimum conditions obtained from the experimental design analysis, ARS extraction was performed in a batch system, too. The two-phase contact times to reach the extraction percentage of 98.7 in the microchannel and batch system were 2.4 s and 5.5 min, respectively.





Similar content being viewed by others
Abbreviations
- C aq, in :
-
Concentration of ARS in the inlet of the aqueous phase mg L−1
- C aq, out :
-
Concentration of ARS in the outlet of the aqueous phase mg L−1
- E :
-
Extraction percentage of ARS
- PC:
-
Percentage contribution
- Q aq :
-
Volumetric flow rate of the aqueous phase m3 s−1
- Q org :
-
Volumetric flow rate of the organic phase m3 s−1
- SS:
-
Sum of squares
- t m :
-
Residence time of mixture two-phase s
- V :
-
Total volume of mixing channel m3
- aq:
-
Aqueous phase
- org:
-
Organic phase
- in:
-
Inlet
- m:
-
Mixture of the liquid–liquid two-phase
- out:
-
Outlet
References
Aghamohammadi N, Aziz HB, Isa MH, Zinatizadeh AA (2007) Powdered activated carbon augmented activated sludge process for treatment of semi-aerobic landfill leachate using response surface methodology. Bioresour Technol 98(18):3570–3578. doi:10.1016/j.biortech.2006.11.037
Aghdasinia H, Khataee A, Sheikhi M, Takhtfiroozeh P (2017) Pilot plant fluidized-bed reactor for degradation of basic blue 3 in heterogeneous fenton process in the presence of natural magnetite. Environ Prog Sustain Energy. doi:10.1002/ep.12569
Aksu Z, Tezer S (2000) Equilibrium and kinetic modelling of biosorption of Remazol Black B by Rhizopus arrhizus in a batch system: effect of temperature. Process Biochem 36(5):431–439. doi:10.1016/S0032-9592(00)00233-8
Aoki N, Fukuda T, Maeda N, Mae K (2013) Design of confluence and bend geometry for rapid mixing in microchannels. Chem Eng J 227:198–202. doi:10.1016/j.cej.2012.03.061
Arzani M, Mahdavi HR, Bakhtiari O, Mohammadi T (2016) Preparation of mullite ceramic microfilter membranes using response surface methodology based on central composite design. Ceram Int 42(7):8155–8164. doi:10.1016/j.ceramint.2016.02.022
Assmann N, von Rohr PR (2011) Extraction in microreactors: intensification by adding an inert gas phase. Chem Eng Process 50(8):822–827. doi:10.1016/j.cep.2011.05.009
Assmann N, Ładosz A, Rudolf von Rohr P (2013) Continuous micro liquid–liquid extraction. Chem Eng Technol 36(6):921–936. doi:10.1002/ceat.201200557
Bayat A, Mahdavi HR, Kazemimoghaddam M, Mohammadi T (2016) Preparation and characterization of γ-alumina ceramic ultrafiltration membranes for pretreatment of oily wastewater. Desalination Water Treat 57(51):24322–24332. doi:10.1080/19443994.2016.1146922
Boubakri A, Hafiane A, Bouguecha SAT (2014) Application of response surface methodology for modeling and optimization of membrane distillation desalination process. J Ind Eng Chem 20(5):3163–3169. doi:10.1016/j.jiec.2013.11.060
Ceylan H, Kubilay S, Aktas N, Sahiner N (2008) An approach for prediction of optimum reaction conditions for laccase-catalyzed bio-transformation of 1-naphthol by response surface methodology (RSM). Bioresour Technol 99(6):2025–2031. doi:10.1016/j.biortech.2007.03.018
Chagas N, Quinaia S, Anaissi F, Santos J, Felsner M, Justi K (2014) Clay and charcoal composites: characterisation and application of factorial design analysis for dye adsorption. Chem Pap 68(4):553. doi:10.2478/s11696-013-0472-z
Chaouchi S, Hamdaoui O (2014) Acetaminophen extraction by emulsion liquid membrane using Aliquat 336 as extractant. Sep Purif Technol 129:32–40. doi:10.1016/j.seppur.2014.03.021
Chen X, Cui D-F (2009) Microfluidic devices for sample pretreatment and applications. Microsyst Technol 15(5):667–676. doi:10.1007/s00542-009-0783-8
Chen X, Li T (2016a) A novel design for passive misscromixers based on topology optimization method. Biomed Microdevice 18(4):57. doi:10.1007/s10544-016-0082-y
Chen X, Li T (2016b) A novel passive micromixer designed by applying an optimization algorithm to the zigzag microchannel. Chem Eng J. doi:10.1016/j.cej.2016.11.052
Chen X, Shen J (2017) Numerical analysis of mixing behaviors of two types of E-shape micromixers. Int J Heat Mass Transf 106:593–600. doi:10.1016/j.ijheatmasstransfer.2016.09.034
Chen X, Zhao Z (2017) Numerical investigation on layout optimization of obstacles in a three-dimensional passive micromixer. Anal Chim Acta. doi:10.1016/j.aca.2017.01.066
Chen X, Liu C, Xu Z, Pan Y, Liu J, Du L (2013) An effective PDMS microfluidic chip for chemiluminescence detection of cobalt(II) in water. Microsyst Technol 19(1):99–103. doi:10.1007/s00542-012-1551-8
Chen X, Li T, Hu Z (2016a) A novel research on serpentine microchannels of passive micromixers. Microsyst Technol. doi:10.1007/s00542-016-3060-7
Chen X, Li T, Zeng H, Hu Z, Fu B (2016b) Numerical and experimental investigation on micromixers with serpentine microchannels. Int J Heat Mass Transf 98:131–140. doi:10.1016/j.ijheatmasstransfer.2016.03.041
Chen X, Shen J, Hu Z, Huo X (2016c) Manufacturing methods and applications of membranes in microfluidics. Biomed Microdevice 18(6):104. doi:10.1007/s10544-016-0130-7
Chung K-T, Cerniglia CE (1992) Mutagenicity of azo dyes: structure-activity relationships. Mutat Res Rev Genet Toxicol 277(3):201–220. doi:10.1016/0165-1110(92)90044-A
Daas A, Hamdaoui O (2010) Extraction of anionic dye from aqueous solutions by emulsion liquid membrane. J Hazard Mater 178(1–3):973–981. doi:10.1016/j.jhazmat.2010.02.033
Design-Expert-Software (2015) Trial version. Www.Statease.Com
Dessimoz A-L, Cavin L, Renken A, Kiwi-Minsker L (2008) Liquid–liquid two-phase flow patterns and mass transfer characteristics in rectangular glass microreactors. Chem Eng Sci 63(16):4035–4044. doi:10.1016/j.ces.2008.05.005
El-Ashtoukhy ESZ, Fouad YO (2015) Liquid–liquid extraction of methylene blue dye from aqueous solutions using sodium dodecylbenzenesulfonate as an extractant. Alex Eng J 54(1):77–81. doi:10.1016/j.aej.2014.11.007
Fan L, Zhang Y, Li X, Luo C, Lu F, Qiu H (2012) Removal of alizarin red from water environment using magnetic chitosan with Alizarin Red as imprinted molecules. Colloids Surf B Biointerfaces 91:250–257. doi:10.1016/j.colsurfb.2011.11.014
Fatoba OS, Akanji OL, Aasa AS (2014) Optimization of carburized UNS G10170 steel process parameters using Taguchi approach and response surface model (RSM). J Miner Mater Charact Eng 2:566–578. doi:10.4236/jmmce.2014.26058
Filiz M, Sayar AA (2006) Extraction of Titanium(IV) from aqueous hydrochloric acid solutions into Alamine 336-m-xylene mixtures. Chem Eng Commun 193(9):1127–1141. doi:10.1080/00986440500354457
Gautam RK, Mudhoo A, Chattopadhyaya MC (2013) Kinetic, equilibrium, thermodynamic studies and spectroscopic analysis of Alizarin Red S removal by mustard husk. J Environ Chem Eng 1(4):1283–1291. doi:10.1016/j.jece.2013.09.021
Isanejad M, Arzani M, Mahdavi HR, Mohammadi T (2017) Novel amine modification of ZIF-8 for improving simultaneous removal of cationic dyes from aqueous solutions using supported liquid membrane. J Mol Liq 225:800–809. doi:10.1016/j.molliq.2016.11.007
Kakavandi FH, Rahimi M, Jafari O, Azimi N (2016) Liquid–liquid two-phase mass transfer in T-type micromixers with different junctions and cylindrical pits. Chem Eng Process 107:58–67. doi:10.1016/j.cep.2016.06.011
Kansal SK, Lamba R, Mehta SK, Umar A (2013) Photocatalytic degradation of Alizarin Red S using simply synthesized ZnO nanoparticles. Mater Lett 106:385–389. doi:10.1016/j.matlet.2013.05.074
Karami HR, Keyhani M, Mowla D (2016) Experimental analysis of drag reduction in the pipelines with response surface methodology. J Petrol Sci Eng 138:104–112. doi:10.1016/j.petrol.2015.11.041
Kashid M, Harshe Y, Agar D (2007) Liquid–liquid slug flow in a capillary: an alternative to suspended drop or film contactors. Ind Eng Chem Res 46(25):8420–8430. doi:10.1021/ie070077x
Kashid MN, Gupta A, Renken A, Kiwi-Minsker L (2010) Numbering-up and mass transfer studies of liquid–liquid two-phase microstructured reactors. Chem Eng J 158(2):233–240. doi:10.1016/j.cej.2010.01.020
Kuban P, Berg J, Dasgupta PK (2003) Vertically stratified flows in microchannels. Computational simulations and applications to solvent extraction and ion exchange. Anal Chem 75(14):3549–3556. doi:10.1021/ac0340713
Marták J, Schlosser Š (2006) Phosphonium ionic liquids as new, reactive extractants of lactic acid. Chem Pap 60(5):395. doi:10.2478/s11696-006-0072-2
Marták J, Schlosser Š (2008) Liquid–liquid equilibria of butyric acid for solvents containing a phosphonium ionic liquid. Chem Pap 62(1):42. doi:10.2478/s11696-007-0077-5
Muthuraman G, Teng TT (2009) Extraction and recovery of rhodamine B, methyl violet and methylene blue from industrial wastewater using D2EHPA as an extractant. J Ind Eng Chem 15(6):841–846. doi:10.1016/j.jiec.2009.09.010
Namasivayam C, Kavitha D (2002) Removal of Congo Red from water by adsorption onto activated carbon prepared from coir pith, an agricultural solid waste. Dyes Pigments 54(1):47–58. doi:10.1016/S0143-7208(02)00025-6
Nandagopal MSG, Antony R, Selvaraju N (2014) Comparative study of liquid–liquid extraction in miniaturized channels over other conventional extraction methods. Microsyst Technol 22(2):349–356. doi:10.1007/s00542-014-2391-5
Owolabi RU, Usman MA, Kehinde AJ (2016) Modelling and optimization of process variables for the solution polymerization of styrene using response surface methodology. J King Saud Univ Eng. doi:10.1016/j.jksues.2015.12.005
Padervand M, Tasviri M, Gholami M (2011) Effective photocatalytic degradation of an azo dye over nanosized Ag/AgBr-modified TiO2 loaded on zeolite. Chem Pap 65(3):280. doi:10.2478/s11696-011-0013-6
Přibyl M, Knapkova V, Šnita D, Marek M (2005) Analysis of reaction-transport phenomena in a microfluidic system for the detection of IgG. Chem Pap 59(6a):434–440
Rahimi M, Azimi N, Parsamogadam MA, Rahimi A, Masahy MM (2016) Mixing performance of T, Y, and oriented Y-micromixers with spatially arranged outlet channel: evaluation with Villermaux/Dushman test reaction. Microsyst Technol. doi:10.1007/s00542-016-3118-6
Shakeel F, Haq N, Alanazi FK, Alsarra IA (2014) Removal of alizarin red from aqueous solution by ethyl acetate green nanoemulsions. Water Sci Technol 70(9):1569–1574. doi:10.2166/wst.2014.400
Shin Y, Cho H, Choi J, Sun Jang Y, Choi YJ, Sohn J, Lee S, Choi J (2015) Application of response surface methodology (RSM) in the optimization of dewetting conditions for flat sheet membrane distillation (MD) membranes. Desalination Water Treat 57:1–11. doi:10.1080/19443994.2015.1038114
Tang J, Zhang X, Cai W, Wang F (2013) Liquid–liquid extraction based on droplet flow in a vertical microchannel. Exp Therm Fluid Sci 49:185–192. doi:10.1016/j.expthermflusci.2013.04.017
Tiwari D, Lalhriatpuia C, Lalhmunsiama, Lee SM, Kong SH (2015) Efficient application of nano-TiO2 thin films in the photocatalytic removal of Alizarin Yellow from aqueous solutions. Appl Surf Sci 353:275–283. doi:10.1016/j.apsusc.2015.06.131
Vijayaraghavan R, Vedaraman N, Surianarayanan M, MacFarlane DR (2006) Extraction and recovery of azo dyes into an ionic liquid. Talanta 69(5):1059–1062. doi:10.1016/j.talanta.2005.12.042
Yang L, Zhao Y, Su Y, Chen G (2013) An experimental study of copper extraction characteristics in a T-junction microchannel. Chem Eng Technol 36(6):985–992. doi:10.1002/ceat.201200464
Yin S, Zhang L, Peng J, Li S, Ju S, Zhang L (2015) Microfluidic solvent extraction of La(III) with 2-ethylhexyl phosphoric acid-2-ethylhexyl ester (P507) by a microreactor. Chem Eng Process 91:1–6. doi:10.1016/j.cep.2015.03.003
Zhang L, Xie F, Li S, Yin S, Peng J, Ju S (2015) Solvent extraction of Nd(III) in a Y type microchannel with 2-ethylhexyl phosphoric acid-2-ethylhexyl ester. Green Process Synth 4(1):3. doi:10.1515/gps-2014-0095
Zhao Y, Chen G, Yuan Q (2007) Liquid–liquid two-phase mass transfer in the T-junction microchannels. AIChE J 53(12):3042–3053. doi:10.1002/aic.11333
Zularisam AW, Ismail AF, Salim MR, Sakinah M, Matsuura T (2009) Application of coagulation–ultrafiltration hybrid process for drinking water treatment: optimization of operating conditions using experimental design. Sep Purif Technol 65(2):193–210. doi:10.1016/j.seppur.2008.10.018
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hosseini Kakavandi, F., Rahimi, M., Baniamer, M. et al. Performance evaluation of Alizarin extraction from aqueous solutions in a microfluidic system. Chem. Pap. 71, 2521–2532 (2017). https://doi.org/10.1007/s11696-017-0248-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-017-0248-y