Abstract
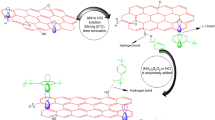
In the present work, a set of polyaniline–graphene oxide (PANI–GO) nanocomposites which exhibit superior properties in terms of shelf life, processability and conductivity due to the synergistic effect of GO and PANI, have been synthesized by varying the concentration of highly non-conducting GO with respect to aniline. The obtained materials were characterized by UV–Vis, FTIR, XRD, Raman, TGA as well as FESEM, TEM analysis. The results reveal that nanocomposites show better dispersibility, crystallinity, thermal stability, and conductivity. Further, the synthesized composites have been tested for their anti-corrosion properties. The potentiodynamic results reveal that PANI nanocomposites with 1% GO exhibited long-term anti-corrosion behavior with a corrosion rate of 6.5 × 10−5 mm year−1, which is much lower than its individual components and commercial-grade red oxide. Also, it possesses highest impedance modulus ~33 kΩ cm2 and real impedance ~32 kΩ cm2, maximum coating resistance ~14.81 × 103 Ω cm2 and minimum coating capacitance after 96 h of immersion in 3.5% mass NaCl than those exhibited by all other coated samples. Higher concentration of GO could not retard the corrosion rate confirming that hydrophilicity of GO play an important role in the redox mechanism of PANI.



















Similar content being viewed by others
References
Athawale A, Bhagwat SV, Katre P (2006) Nanocomposite of Pd–polyaniline as a selective methanol sensor. Sensors Actuators B 114:263–267. doi:10.1016/j.snb.2005.05.009
Chang CH, Huang TC, Peng CW, Yeh TC, Lu HI, Hung WI, Weng CJ, Yang TI, Yeh JM (2012) Novel anticorrosion coatings prepared from polyaniline/graphene composites. Carbon 50:5044–5051. doi:10.1016/j.carbon.2012.06.043
Conradi M, Kocijan A (2016) Surface and anticorrosion properties of hydrophobic and hydrophilic TiO2 coatings on a stainless-steel substrate (2016). Mat Technol 50(6):967–970. DOI:10.17222/mit.2016.068
Deberry DW (1985) Modification of the electrochemical and corrosion behavior of stainless steel with electroactive coating. J Electrochem Soc 132:1022–1026. doi:10.1149/1.2114008
Deshpande PP, Jadhav NG, Gelling VJ, Sazou D (2014) Conducting polymers for corrosion protection: a review. J Coating Technol Res 11:473–494. doi:10.1007/s11998-014-9586-7
Ganguly A, Sharma S, Papakonstantinou P, Hamilton J (2011) Probing the thermal deoxygenation of graphene oxide using high-resolution in situ X-ray-based spectroscopies. J Phys Chem C 115:17009–17019. doi:10.1021/jp203741y
Guo Y, Sun X, Liu Y, Wang W, Qiu H, Gao J (2012) One pot preparation of reduced graphene oxide (RGO) or Au (Ag) nanoparticle-RGO hybrids using chitosan as a reducing and stabilizing agent and their use in methanol electro oxidation. Carbon 50:2513–2523. doi:10.1016/j.carbon.2012.01.074
He Y, Yu Z, Di H, Ma Y, Liang L, Pan Y, Zhang C (2015) Fabrication of graphene oxide–alumina hybrids to reinforce the anti-corrosion performance of composite epoxy coatings. Appl Surf Sci 351:986–996. doi:10.1016/j.apsusc.2015.06.026
He Y, Yu Z, Liang L, Ma Y, Di H (2016) Covalent modification of graphene oxide by metronidazole for reinforced anti-corrosion properties of epoxy coatings. RSC Adv 6:18217–18226. doi:10.1039/C5RA23595B
Hsiao MC, Liao SH, Yen MY, Liu PI, Pu NW, Wang CA, Ma CCM (2010) Preparation of covalently functionalized graphene using residual oxygen-containing functional groups. ACS App Mater Interfaces 2:3092–3099. doi:10.1021/am100597d
Kendova A, Vesely D, Stejskal J (2008) Organic coatings containing polyaniline and inorganic pigments as corrosion inhibitors. Prog Organ Coating 62:105–116. doi:10.1016/j.porgcoat.2007.10.001
Krishna BM, Saha A, Okram GS, Soni A, Purakayastha S, Ghosh B (2009) Electrical properties of polyaniline doped with metal ions. J Phys D Appl Phys 42:095404–095410. doi:10.1088/0022-3727/42/9/095404
Krishnamoorthy K, Jeyasubramanian K, Premanathan M, Subbiah G, Shin HS, Kim SJ (2014) Graphene oxide nanopaint. Carbon 7(2):328–337
Lai Q, Zhu S, Luo X, Zou M, Huang S (2012) Ultraviolet-visible spectroscopy of graphene oxides. AIP Advances 2(32146):1–6. doi:10.1063/1.4747817
Lee V, Whittaker L, Jaye C, Baroudi KM, Fischer DA, Banerjee S (2009) Large-area chemically modified graphene films: electrophoretic deposition and characterization by soft x-ray absorption spectroscopy. Chem Mater 21:3905–3916. doi:10.1021/cm901554p
Liu P, Liu W, Xue Q (2004) In situ chemical oxidative graft polymerization of aniline from silica nanoparticles. Mater Chem Phy 87:109–113. doi:10.1016/j.matchemphys.2004.05.001
Liu WR, Chang KC, Ji WF, Lai MC, Hsiao YR, Hsu CH, Chuang TL, Wei Y, Yeha JM (2014) Synergistic effects of hydrophobicity and gas barrier properties on the anticorrosion property of PMMA nanocomposite coatings embedded with graphene nanosheets. Polym Chem 5:1049–1056. doi:10.1039/C3PY01178J
Lu WK, Elsenbaumer RL, Wessling B (1995) Corrosion protection of mild steel by coating containing polyaniline. Synth Met 71:2163–2166. doi:10.1016/0379-6779(94)03204-J
Lu JX, Moon KS, Wong CP (2007) High dielectric constant polyaniline/epoxy composites via in situ polymerization for embedded capacitor applications. Polymer 48:1510–1516. doi:10.1016/j.polymer.2007.01.057
Marcel C, Tarascon JM (2001) An all-plastic WO3.H2O/polyaniline electrochromic device. Solid State Ionics 143:89–101. doi:10.1016/S0167-2738(01)00837-2
Mathew H, Vrinda S, Kuriakose PS, SyamalaKumari B, Manuel A (2013) Synthesis and electrical characterization of polyaniline-multiwalled carbon nanotube composites with different dopants. Int J Sci Res Publ 8:1–10
Meroufel A, Deslouis C, Touzain S (2008) Electrochemical and anticorrosion performances of zinc-rich and polyaniline powder coatings. Electrochim Acta 53:2331–2338. doi:10.1016/j.electacta.2007.09.056
Mooss VA, Athawale AA (2016) Polyaniline-graphene oxide nanocomposites: Influence of nonconducting graphene oxide on the conductivity and oxidation-reduction mechanism of polyaniline. J Polym Sci A Polym Chem 54:3778–3786. doi:10.1002/pola.28277
Mostafaei A, Nasirpouri F (2014) Epoxy/polyaniline–ZnO nanorods hybrid nanocomposite coatings: Synthesis, characterization and corrosion protection performance of conducting paints. Prog Org Coat 77:146–159. doi:10.1016/j.porgcoat.2013.08.015
Navarro CG, Meyer JC, Sundaram RS, Chuvilin A, Kurasch S, Burghard M, Kern K, Kaiser U (2010) Atomic structure of reduced graphene oxide. Nano Lett 10:1144–1148. doi:10.1021/nl9031617
Olad A, Naseri B (2010) Preparation, characterization, and anticorrosive properties of a novel polyaniline/clinoptilolite nanocomposite. Prog Organic Coating 67:233–238. doi:10.1016/j.porgcoat.2009.12.003
Pimenta MA, Dresselhaus G, Dresselhaus MS, Cancado LG, Jorioa A, Saito R (2007) Studying disorder in graphite-based systems by Raman spectroscopy. Phys Chem Chem Phys 9:1276–1291. doi:10.1039/B613962K
Radhakrishnan S, Siju CR, Mahanta D, Patil S, Madras G (2009) Conducting polyaniline–nano–TiO2 composites for smart corrosion resistant coatings. Electrochim Acta 54:1249–1254. doi:10.1016/j.electacta.2008.08.069
Rana U, Malik S (2012) Graphene oxide/polyaniline nanostructures: transformation of 2D sheet to 1D nanotube and in situ reduction. Chem Commun 48:10862–10864. doi:10.1039/C2CC36052G
Rohwerder M, Michalik A (2007) Conducting polymers for corrosion protection: what makes the difference between failure and success? Electrochim Acta 53:1300–1313. doi:10.1016/j.electacta.2007.05.026
Sahoo S, Khurana GS, Barik K, Dussan S, Barrionuevo D, Katiyar RS (2013) In situ Raman studies of electrically reduced graphene oxide and its field-emission properties. J Phys Chem C 117:5485–5491. doi:10.1021/jp400573w
Samui AB, Patankar AS, Rangarajan J, Deb PC (2003) Study of polyaniline containing paint for corrosion prevention. Prog Org Coat 47:1–7. doi:10.1016/S0300-9440(02)00117-0
Sathiyanarayanan S, Muthukrishnan S, Venkatachari G (2006) Performance of polyaniline pigmented vinyl acrylic coating on steel in aqueous solutions. Prog Org Coat 55:5–10. doi:10.1016/j.porgcoat.2005.09.002
Sawangphruk M, Suksomboon M, Kongsupornsak K, Khuntilo J, Srimuk P, Sanguansak Y, Klunbud P, Suktha P, Chiochan P (2013) High-performance supercapacitors based on silver nanoparticle–polyaniline–graphene nanocomposites coated on flexible carbon fiber paper. J Mater Chem A 1:9630–9636. doi:10.1039/C3TA12194A
Shao D, Hou G, Li J, Wen T, Ren X, Xinagke, Wang (2014) PANI/GO as a super adsorbent for the selective adsorption of uranium (VI). Chem Eng J 255:604–612. doi:10.1016/j.cej.2014.06.063
Stankovich S, Pine RD, Chen X, Wu N, Nguyen SBT, Ruoff RS (2006) Stable aqueous dispersions of graphitic nanoplatelets via the reduction of exfoliated graphite oxide in the presence of poly (sodium 4-styrenesulfonate). J Mater Chem 16:155–158. doi:10.1039/B512799H
Valles C, Muñoz PJE, Benito AM, Maser WK (2011) Simultaneous reduction of graphene oxide and polyaniline: doping-assisted formation of a solid state charge-transfer complex. J Phys Chem C 115:10468–10474. doi:10.1021/jp201791h
Venugopal G, Krishnamoorthy K, Mohan R, Kim S (2012) An investigation of the electrical transport properties of graphene oxide thin films. Mater Chem Phys 132:29–33. doi:10.1016/j.matchemphys.2011.10.040
Wang W, Gumfekar SP, Jia Q, Zhao B (2013) Ferrite-grafted polyaniline nanofibers as electromagnetic shielding materials. J Mater Chem 1:2851–2859. doi:10.1039/C3TC00757J
Wessling B (1996) Corrosion prevention with an organic metal (polyaniline): Surface ennobling, passivation, corrosion test results. Mater Corros 47:439–445. doi:10.1002/maco.19960470804
Williams G, McMurray HN (2005) Factors affecting acid-base stability of the interface between polyaniline emeraldine salt and oxide covered metal. Electrochem Solid-State Lett 8:B42–B45. doi:10.1149/1.1959978
Yang D, Lu W, Goering R, Mattes BR (2009) Investigation of polyaniline processibility using GPC/Uv–vis analysis. Synth Met 159:666–674. doi:10.1016/j.synthmet.2008.12.013
Yeh JM, Chang KC, Hsu MH, Lu HI, Lai MC, Liu PJ, Hsu CH, Ji WF, Chuang TL, Wei Y, Liu WR (2014) Room-temperature cured hydrophobic epoxy/graphene composites as corrosion inhibitor for cold-rolled steel. Carbon 66:144–153. doi:10.1016/j.carbon.2013.08.052
Yu H, Tian Z, Wang L, Saleem M, Ren F, Ren P, Chen Y, Sun R, Sun Y, Huang L (2014). RSC Adv 4:28195–28208. doi:10.1039/c4ra03146f
Zhang X, Xie D, Zhong L, Cai W, Sheng X (2016) Synthesis of functionalized graphene/polyaniline nanocomposites with effective synergistic reinforcement on anticorrosion. Ind Eng Chem Res 55:8576–8585. doi:10.1021/acs.iecr.6b01975
Acknowledgements
The authors thank Department of Chemistry and Physics for characterization techniques. One of the authors VM thanks SPPU for the financial support. The authors also thank Prof. N.B. Dhokey, Head, Department of Metallurgy and Materials Science, College of Engineering, Pune, 411005 (M.S.), India, for providing facilities for anti-corrosion studies and Prof. B.B. Ahuja, Director, College of Engineering, Pune, 411005 (M.S.), India, for his encouragement.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mooss, V.A., Bhopale, A.A., Deshpande, P.P. et al. Graphene oxide-modified polyaniline pigment for epoxy based anti-corrosion coatings. Chem. Pap. 71, 1515–1528 (2017). https://doi.org/10.1007/s11696-017-0146-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-017-0146-3