Abstract
Introduction
Bariatric surgery has seen a sharp rise in India in the last decade. India is one of the 10 most obese nations of the world, ranking second in number of type 2 diabetics.
Aims
To evaluate clinical outcomes of bariatric surgery after 3 years of follow-up in terms of weight loss, co-morbidity resolution, complaints of gastroesophageal reflux disease and weight regain.
Methodology
All patients who underwent bariatric surgery from January to December 2013 with a minimum follow-up of 3 years were included in the study. Their demographic, preoperative, and postoperative data were prospectively maintained on Microsoft Office Excel and analyzed statistically.
Results
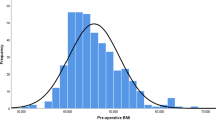
One hundred seventy-eight patients (157 lap. sleeve gastrectomy and 21 patients lap. RYGB) completed 3 years of follow-up. In the LSG group, patients had a pre-operative BMI 44.8 ± 8.33 kg/sq. m (mean ± S.D.) and excess body weight 52.3 ± 23.0 kg. In the RYGB group, pre-operative BMI was 42.7 ± 8.82 kg/sq. m and excess body weight 45 ± 18.7 kg. In the LSG group, % excess weight loss (EWL) at 1 year was 87.6 ± 24.4% and 3 years was 71.8 ± 26.7%. In the RYGB group, % EWL at 1 year was 97.2 ± 27.3% and at 3 years was 85.8 ± 25.3%. Diabetes resolution was seen in 32 (80%) in LSG group and 11 (91.7%) in RYGB group (Figs. 1, 2, 3, and 4).
Conclusion
Our study reflects that there is no statistically significant difference between outcomes of sleeve gastrectomy and Roux-en-Y gastric bypass surgery in terms of weight loss and diabetes resolution at 3 years.




Similar content being viewed by others
References
Remedios C, Bhasker AG, Dhulla N, et al. Bariatric nutrition guidelines for the Indian population. Obes Surg. 2016;26:1057–68.
Khorgami Z, Shoar S, Andalib A, et al. Trends in utilization of bariatric surgery, 2010–2014: sleeve gastrectomy dominates. Surg Obes Relat Dis. 2017;13:774–8.
Prasad P, Tantia O, Patle N, et al. An analysis of 1–3-year follow-up results of laparoscopic sleeve gastrectomy: an Indian perspective. Obes Surg. 2012;22:507–14.
Melissas J., Stavroulakis, K., Tzikoulis, V. et al. Sleeve gastrectomy vs Roux-en-Y gastric bypass. Data from IFSO-European Chapter Center of Excellence Program. Obes Surg (2017) 27: 847.
Vidal P, Ramón JM, Goday A, et al. Laparoscopic gastric bypass versus laparoscopic sleeve gastrectomy as a definitive surgical procedure for morbid obesity. Mid-term results. Obes Surg. 2013;23:292–9.
Misra A, Chowbey P, Makkar BM, et al. Consensus statement for diagnosis of obesity, abdominal obesity and the metabolic syndrome for Asian Indians and recommendations for physical activity, medical and surgical management. JAPI. 2009;57:163–70.
Lauti M, Kularatna M, Hill AG, et al. Weight regain following sleeve gastrectomy—a systematic review. Obes Surg. 2016;26:1326–34.
Chowbey PK, Dhawan K, Khullar R, et al. Laparoscopic sleeve gastrectomy: an Indian experience—surgical technique and early results. Obes Surg. 2010;20:1340–7.
Park JY, Kim YJ. Laparoscopic gastric bypass vs sleeve gastrectomy in obese Korean patients. World J Gastroenterol. 2015;21(44):12612–9.
Li P, Fu P, Chen J, et al. Laparoscopic Roux-en-Y gastric bypass vs. laparoscopic sleeve gastrectomy for morbid obesity and diabetes mellitus: a meta-analysis of sixteen recent studies. Hepato-Gastroenterology 2013;60(121):132–137.
Jacobs M, Bisland W, Gomez E, et al. Laparoscopic sleeve gastrectomy: a retrospective review of 1 and 2 years results. Surg Endosc. 2010;24:781–5.
DeMaria EJ, Pate V, Warthen M, et al. Baseline data from American Society for Metabolic and Bariatric Surgery-designated Bariatric Surgery Centers of Excellence using the bariatric outcomes longitudinal database. Surg Obes Relat Dis. 2010;6(4):347–55.
Gupta PK, Franck C, Miller WJ, et al. Development and validation of a bariatric surgery morbidity risk calculator using the prospective, multicenter NSQIP dataset. J Am Coll Surg. 2011;212(3):301–9.
Rubin M, Yehoshua RT, Stein M, et al. Laparoscopic sleeve gastrectomy with minimal morbidity. Early results in 120 morbidly obese patients. Obes Surg. 2008;18(12):1567–70.
Weiner RA, El-Sayes IA, Theodoridou S, et al. Early post-operative complications: incidence, management, and impact on length of hospital stay. A retrospective comparison between laparoscopic gastric bypass and sleeve gastrectomy. Obes Surg. 2013;22(12):2004–12.
Trelles N, Gagner M. Updated review of sleeve gastrectomy. The open Gastroenterol J. 2008;2:41–9.
Fischer L, Hildebrandt C, Bruckner T, et al. Excessive weight loss after sleeve gastrectomy: a systematic review. Obes Surg. 2012;22:721–31.
Zerrweck C, Sepúlveda EM, Maydón HG, et al. Laparoscopic gastric bypass vs. sleeve gastrectomy in the super obese patient: early outcomes of an observational study. Obes Surg. 2014;24(5):712–7.
Yaghoubian A, Tolan A, Stabile BE, et al. Laparoscopic Roux-en-Y gastric bypass and sleeve gastrectomy achieve comparable weight loss at 1 year. Am Surg. 2012;78(12):1325–8.
Cutolo PP, Nosso G, Vitolo G, et al. Clinical efficacy of laparoscopic sleeve gastrectomy vs laparoscopic gastric bypass in obese type 2 diabetic patients: a retrospective comparison. Obes Surg. 2012;22(10):1535–9.
Yang J, Wang C, Cao G, et al. Long-term effects of laparoscopic sleeve gastrectomy versus roux-en-Y gastric bypass for the treatment of Chinese type 2 diabetes mellitus patients with body mass index 28-35 kg/m2. BMC Surg. 2015;15:88.
Himpens J, Dobbeleir J, Peeters G. Long term results of laparoscopic sleeve gastrectomy for obesity. Ann Surg. 2010;252(2):319–24.
Karmali S, Brar B, Shi X, et al. Weight recidivism post-bariatric surgery: a systematic review. Obes Surg. 2013;23:1922–33.
Peterli R, Borbely Y, Kern B, et al. Early results of the Swiss multicentre bypass or sleeve study (SM-BOSS): a prospective randomized trial comparing laparoscopic sleeve gastrectomy and Roux-en-Y gastric bypass. Ann Surg. 2013;258:690–5.
Christou NV, Look D, Maclean LD. Weight gain after short- and long-limb gastric bypass in patients followed for longer than 10 years. Ann Surg. 2006;244:734–40.
Cooper TC, Simmons EB, Webb K, et al. Trends in weight regain following Roux-en-Y gastric bypass (RYGB) bariatric surgery. Obes Surg. 2015;25:1474–81.
Nicoletti CF, de Oliveira BA, de Pinhel MA, et al. Influence of excess weight loss and weight regain on biochemical indicators during a 4-year follow-up after Roux-en-Y gastric bypass. Obes Surg. 2015;25:279–84.
Liu SYW, Wong SKH, Lam CCH, et al. Long-term results on weight loss and diabetes remission after laparoscopic sleeve gastrectomy for a morbidly obese Chinese population. Obes Surg. 2015;25(10):1901–8.
Abdallah E, El Nakeeb A, Yousef T, et al. Impact of extent of antral resection on surgical outcomes of sleeve gastrectomy for morbid obesity (a prospective randomized study). Obes Surg. 2014;24(10):1587–94.
Bohdjalian A, Langer FB, Shakeri-Leiden Muhler S, et al. Sleeve gastrectomy as sole and definitive bariatric procedure: 5-year results for weight loss and ghrelin. Obes Surg. 2010;20(5):535–40.
Braghetto I, Csendes A, Lanzarini E, et al. Is laparoscopic sleeve gastrectomy an acceptable primary bariatric procedure in obese patients? Early and 5-year postoperative results. Surg Laparosc Endosc Percutan Tech. 2012;22(6):479–86.
Casella G, Soricelli E, Giannotti D, et al. Long-term results after laparoscopic sleeve gastrectomy in a large monocentric series. Surg Obes Relat Dis. 2016;12(4):757–62.
Auclair A, Biertho L, Marceau S, et al. Bariatric surgery-induced resolution of hypertension and obstructive sleep apnea: impact of modulation of body fat, ectopic fat, autonomic nervous activity, inflammatory and adipokine profiles. Obes Surg. 2017;27:3156.
King WC, Jia-Yuh Chen SH. Belle et al. change in pain and physical function following bariatric surgery for severe obesity. JAMA. 2016;315(13):1362–71.
Schauer P, Bhatt D, Kirwan J, et al. Et al. bariatric surgery versus intensive medical therapy for diabetes—3-year outcomes. N Engl J Med. 2014;370:2002–13.
Neff KJ, Baud G, Raverdy V, et al. Renal function and remission of hypertension after bariatric surgery: a 5-year prospective cohort study. Obes Surg. 2017;27:613–9.
Sharma A, Aggarwal S, Ahuja V, et al. Evaluation of gastroesophageal reflux before and after sleeve gastrectomy using symptom scoring, scintigraphy, and endoscopy. Surg Obes Relat Dis. 2014;10(4):600–5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of Interest
The authors declare that they have no conflict of interest.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Nasta, A.M., Goel, R., Dharia, S. et al. Weight Loss and Comorbidity Resolution 3 Years After Bariatric Surgery—an Indian Perspective. OBES SURG 28, 2712–2719 (2018). https://doi.org/10.1007/s11695-018-3218-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-018-3218-8