Abstract
Purpose
The purpose of this study is to investigate the safety and 9-month effectiveness of transcatheter left gastric artery embolization (LGAE) for treating patients with obesity.
Materials and Methods
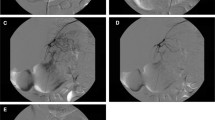
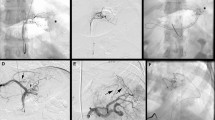
The protocol of this study was approved by the Institutional Ethics Review Board. Five obese patients (3 men and 2 women) with mean weight of 102.0 ± 16.19 kg (range, 82.1–125.5 kg) and mean body mass index (BMI) of 38.1 kg/m2 ± 3.8 (range, 32.9–42.4 kg/m2) underwent LGAE with polyvinyl alcohol (PVA) particles in diameter of 500–710 μm. The primary endpoint was the safety by grading the adverse events (AEs) according to the Common Terminology Criteria for Adverse Events (CTCAE v4.0) within 30 days after LGAE. The secondary endpoints were measured with serum ghrelin and leptin levels, body weight, waist circumference, waist-to-height ratio, and abdominal fat quantity on MRI at the day immediately before LGAE and every 3 months after LGAE.
Results
LGAE was successfully performed in all patients. A superficial linear ulceration below the cardia was seen in 1 patient 3 days after LGAE and healed within 30 days. No other serious AEs (grade III or above) occurred. Average body weight loss at 3, 6, and 9 months was 8.28 ± 7.3 kg (p = 0.074), 10.42 ± 8.21 kg (p = 0.047), and 12.9 ± 14.66 kg (p = 0.121), respectively. The level of serum ghrelin decreased by 40.83% (p = 0.009), 31.94% (p = 0.107), and 24.82% (p = 0.151) at 3, 6, and 9 months after LGAE, respectively. There was minimal reduction of leptin levels at 3 and 6 months following LGAE (decreased by 0.26%, p = 0.929, and 4.33%, p = 0.427, respectively), but it declined obviously 9 months after LGAE (decreased by 11.22%, p = 0.295).
Both waist circumference and waist-to-height ratio decreased after LGAE. MRI showed the area of subcutaneous adipose tissue decreased from the baseline of 400.90 ± 79.25 to 320.36 ± 68.06 cm2 (decreased by 20.09%, p = 0.006) at 3 months, to 328.31 ± 52.67 cm2 (decreased by 18.11%, p = 0.020) at 6 months, and to 286.40 ± 55.72 cm2 (decreased by 28.52%, p = 0.101) at 9 months after LGAE, respectively. But the decrease of abdominal fat loss at 9 months after LGAE was largely due to the reduction in visceral adipose tissue.
Conclusions
Our study with 9-month data in 5 patients indicates that bariatric embolization of the LGA is a safe and may be a promising strategy to suppress the production of ghrelin and results in weight loss and abdominal fat reduction.
Trial Registration
ClinicalTrials.gov (NCT02786108).



Similar content being viewed by others
References
Au N, Johnston DW. Too much of a good thing? Exploring the impact of wealth on weight. Health Econ. 2015;24(11):1403–21.
Asiseh F, Yao J. Family income and body mass index—what have we learned from China. Heal Econ Rev. 2016;6(1):52.
Bays HE, Toth PP, Kris-Etherton PM, et al. Obesity, adiposity, and dyslipidemia: a consensus statement from the National Lipid Association. J Clin. 2013;7(4):304–83.
Mokdad AH, Ford ES, Bowman BA, et al. Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001. JAMA. 2003;289(1):76–9.
Jung Y. Role of endoscopic gastroplasty techniques in the management of obesity. Clin Endosc. 2017;50(1):21–5.
Schauer PR, Bhatt DL, Kirwan JP, et al. Bariatric surgery versus intensive medical therapy for diabetes—3-year outcomes. N Engl J Med. 2014;370:2002–13.
Varban OA, Cassidy RB, Bonham A, et al. Factors associated with achieving a body mass index of less than 30 after bariatric surgery. JAMA Surg JAMA Surg. 2017; https://doi.org/10.1001/jamasurg.2017.2348.
Chang SH, Stoll CR, et al. The effectiveness and risks of bariatric surgery: an updated systematic review and meta-analysis, 2003-2012. JAMA Surg. 2014;149:275–87.
Allen JW. Laparoscopic gastric band complications. Med Clin N Am. 2007;91:485–97.
Abu Dayyeh BK, Edmundowicz S, Thompson CC. Clinical practice update: expert review on endoscopic bariatric therapies. Gastroenterology. 2017;152(4):716–29.
Neylan CJ, Dempsey DT, Tewksbury CM, et al. Endoscopic treatments of obesity: a comprehensive review. Surg Obes Relat Dis. 2016;12(5):1108–15.
Koehestanie P, de Jonge C, Berends FJ, et al. The effect of the endoscopic duodenal-jejunal bypass liner on obesity and type 2 diabetes mellitus, a multicenter randomized controlled trial. Ann Surg. 2014;260(6):984–92.
Arepally A, Barnett BP, Montgomery E, et al. Catheter-directed gastric artery chemical embolization for modulation of systemic ghrelin levels in a porcine model: initial experience. Radiology. 2007;244(1):138–43.
Arepally A, Barnett BP, Patel TH, et al. Catheter-directed gastric artery chemical embolization suppresses systemic ghrelin levels in porcine model. Radiology. 2008;249(1):127–33.
Bawudun D, Xing Y, Liu WY, et al. Ghrelin suppression and fat loss after left gastric artery embolization in canine model. Cardiovasc Intervent Radiol. 2012;35(6):1460–6.
Paxton BE, Alley CL, Crow JH, et al. Histopathologic and immunohistochemical sequelae of bariatric embolization in a porcine model. J Vasc Interv Radiol. 2014;25(3):455–61.
Paxton BE, Kim CY, Alley CL, et al. Bariatric embolization for suppression of the hunger hormone ghrelin in a porcine model. Radiology. 2013;266(2):471–9.
Kipshidze N, Archvadze A, Bertog S, et al. Endovascular bariatrics: first-inman of left gastric artery embolization for weight loss. JACC Cardiovasc Interv. 2015;8(12):1641–4.
Syed MI, Morar K, Shaikh A, et al. Gastric artery embolization trial for the lessening of appetite nonsurgically (GET LEAN): six-month preliminary data. J Vasc Interv Radiol. 2016;27(10):1502–8.
Weiss CR, Akinwande O, Paudel K, et al. Clinical safety of bariatric arterial embolization: preliminary results of the BEAT obesity trial. Radiology. 2017;283(2):598–608.
Mihalache L, Gherasim A, Niță O, et al. Effects of ghrelin in energy balance and body weight homeostasis. Hormones (Athens). 2016;15(2):186–96.
Esler WP, Rudolph J, Claus TH, et al. Small-molecule ghrelin receptor antagonists improve glucose tolerance, suppress appetite, and promote weight loss. Endocrinology. 2007;148(11):5175–85.
Lu SC, Xu J, Chinookoswong N, et al. An acyl-ghrelin-specific neutralizing antibody inhibits the acute ghrelin-mediated orexigenic effects in mice. Mol Pharmacol. 2009;75(4):901–7.
Andrade S, Pinho F, Ribeiro AM, et al. Immunization against active ghrelin using virus-like particles for obesity treatment. Curr Pharm Des. 2013;19(36):6551–8.
Morais T, Andrade S, Pereira SS, et al. Vaccines for metabolic diseases: current perspectives. Vaccine (Auckl). 2014;4:55–72.
Prochaska JM, Flye MW, Johnsrude IS. Left gastric artery embolization for control of gastric bleeding: a complication. Radiology. 1973;107:521–2.
Gunn AJ, OkluR. A preliminary observation of weight loss following left gastric artery embolization in humans. J Obes. 2014;2014:185349.
Expert Panel on Detection Evaluation and Treatment of High Blood Cholesterol In Adults. Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). JAMA. 2001;285:2486–2497.
Kangas S, Timonen P, Knuuttila M, et al. Waist circumference and waist-to-height ratio are associated with periodontal pocketing—results of the Health 2000 Survey. BMC Oral Health. 2017;17(1):48.
Levitt MR, Morton RP, Haynor DR, et al. Angiographic perfusion imaging: real-time assessment of endovascular treatment for cerebral vasospasm. J Neuroimaging. 2014;24:387–92.
Meeks MDME, Mayer J, Desgranges P, et al. Evaluation of infrapopliteal blood flow changes during endovascular revascularization using 2D X-ray perfusion software: a pilot study. Poster presented at CIRSE 2014, Glasgow, UK. Abstract nr 296.
Ravussin Y, Leibel RL, Ferrante Jr AW. A missing link in body weight homeostasis: the catabolic signal of the over fed state. Cell Metab. 2014;20(4):565–72.
Hursting SD. Obesity, energy balance, and cancer: a mechanistic perspective. Cancer Treat Res. 2014;159:21–33.
Jensen MD. Role of body fat distribution and the metabolic complications of obesity. J Clin Endocrinol Metab. 2008;93:S57–63.
Moller JB, Pedersen M, Tanaka H, et al. Body composition is the main determinant for thedifference in type 2 diabetes pathophysiology between Japanese and Caucasians. Diabetes Care. 2014;37(3):796–804.
Hwang YC, Hayashi T, Fujimoto Y, et al. Visceral abdominal fat accumulation predicts the conversion of metabolically healthy obese subjects to an unhealthy phenotype. Int J Obes. 2015;39(9):1365–70.
Fischer K, Pick JA, Moewes D, et al. Qualitative aspects of diet affecting visceral and subcutaneous abdominal adipose tissue: a systematic review of observational and controlled intervention studies. Nutr Rev. 2015;73(4):191–215.
Baum T, Cordes C, Dieckmeyer M, et al. MR-based assessment of body fat distribution and characteristics. Eur J Radiol. 2016;85(8):1512–8.
Matsuzawa Y, Shimomura I, Nakamura T, et al. Pathophysiology and pathogenesis of visceral fat obesity. Obes Res. 1995; Suppl 2:187S–194S.
Fujimoto WY, Abbate SL, Kahn SE, et al. The visceral adiposity syndrome in Japanese-American men. Obes Res. 1994;2(4):364–71.
Banerji MA, Faridi N, Atluri R, et al. Body composition, visceral fat, leptin, and insulin resistance in Asian Indian men. J Clin Endocrinol Metab. 1999;84:137–44.
Galanakis CG, Daskalakis M, Manios A, et al. Computed tomography-based assessment of abdominal adiposity changes and their impact on metabolic alterations following bariatric surgery. World J Surg. 2014;39:417–23.
Toro-Ramos T, Goodpaster BH, Janumala I, et al. Continued loss in visceral and intermuscular adipose tissue in weight-stable women following bariatric surgery. Obesity. 2015;23:62–9.
Kim MK, Kim W, Kwon HS, et al. Effects of bariatric surgery on metabolic and nutritional parameters in severely obese Korean patients with type 2 diabetes: a prospective 2-year follow up. J Diabetes Investig. 2014;5:221–7.
Yoon DY, Kim HK, Kim JA, et al. Changes in the abdominal fat distribution after gastrectomy: computed tomography assessment. ANZ J Surg. 2007;77:121–5.
Funding
This study was supported by Jiangsu Provincial Special Program of Medical Science (BL2013029), China; National Basic Research Program of China (973 Program # 2013CB733800, 2013733803), and National Natural Science Foundation of China (81230034, 81520108015).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The protocol of this study was approved by our Institutional Ethics Review Board (IERB)
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Bai, ZB., Qin, YL., Deng, G. et al. Bariatric Embolization of the Left Gastric Arteries for the Treatment of Obesity: 9-Month Data in 5 Patients. OBES SURG 28, 907–915 (2018). https://doi.org/10.1007/s11695-017-2979-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-017-2979-9