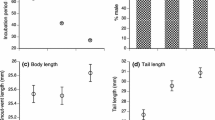
Abstract
Two hypotheses have prevailed to explain the evolution of viviparity in reptiles: the first proposed that viviparity evolved in response to cold-climates because the possibility of pregnant females to thermoregulate at higher temperatures than embryos could experience in a nest in nature. The second hypothesis posits that the advantage of viviparity is based on the possibility of females to maintain stable body temperatures during development, enhancing offspring fitness. With the aim to contribute to understanding the origins of viviparity in reptiles, we experimentally subjected pregnant females of the austral lizard Liolaemus sarmientoi to two temperature treatments until parturition: one that simulated environmental temperatures for a potential nest (17–25 °C) and another that allowed females to thermoregulate at their preferred body temperature (17–45 °C). Then, we analysed newborn body conditions and their locomotor performance to estimate their fitness. In addition, we measured the body temperature in the field and the preferred temperature in the laboratory of pregnant and non-pregnant females. Pregnant females thermoregulated to achieve higher temperatures than the environmental temperatures, and also thermoregulated within a narrower range than non-pregnant females. This could have allowed embryos to develop in higher and more stable temperatures than they would experience in a nest in nature. Thus, offspring developed at the female preferred temperature showed greater fitness and were born earlier in the season than those developed at lower environmental temperatures. Herein, we show that results are in agreement with the two hypotheses of the origin of viviparity for one of the southernmost lizards of the world.




Similar content being viewed by others
References
Aguilar, C., Wood, P. L. Jr., Cusi, J. C., Guzman, A., Huari, F., Lundberg, M., et al. (2013). Integrative taxonomy and preliminary assessment of species limits in the Liolaemus walkeri complex (Squamata, Liolaemidae) with descriptions of three new species from Peru. ZooKeys, 364, 47–91.
Angilletta, M. J. Jr., Niewiarowski, P. H., & Navas, C. A. (2002). The evolution of thermal physiology in ectotherms. Journal of Thermal Biology, 27, 249–268.
Aparicio, J., & Ocampo, M. (2010). Liolaemus grupo montanus Etheridge, 1995 (Iguania-Liolaemidae). Cuadernos de Herpetología, 24, 133–135.
Atkins, N., Swain, R., Wapstra, E., & Jones, S. M. (2007). Late stage deferral of parturition in the viviparous lizard Niveoscincus occellatus (Gray, 1845): Implications for offspring quality and survival. Biological Journal of the Linnean Society, 90, 735–746.
Bauwens, D., Hertz, P. E., & Castilla, A. M. (1996). Thermoregulation in a lacertid lizard: The relative contributions of distinct behavioral mechanisms. Ecology, 77, 1818–1830.
Blackburn, D. G. (1982). Evolutionary origins of viviparity in the Reptilia. I. Sauria. Amphibia-Reptilia, 3, 185–205.
Blackburn, D. G. (2000). Reptilian viviparity: Past research, future directions, and appropriate models. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology, 127, 391–409.
Blackburn, D. G. (2015). Evolution of viviparity in Squamate reptiles: Reversibility reconsidered. Journal of Experimental Zoology Part B: Molecular and Developmental Evolution, 324(6), 473–486.
Booth, D. T., Thompson, M. B., & Herring, S. (2000). How incubation temperature influences the physiology and growth of embryonic lizards. Journal of Comparative Physiology B, 170, 269–276.
Bottari, C. V. (1975). Sobre la presencia de Liolaemus magellanicus en Tierra del Fuego, Argentina (Reptilia Iguanidae). PHYSIS, 34, 211–213.
Braña, F., & Ji, X. (2007). The selective basis for increased egg retention: Early incubation temperature determines hatchling phenotype in wall lizards (Podarcis muralis). Biological Journal of the Linnean Society, 92, 441–447.
Breitman, M. F., Minoli, I., Avila, L. J., Medina, C. D., Sites, J. W. Jr., & Morando, M. (2014). Lagartijas de la provincia de Santa Cruz, Argentina: Distribución geográfica, diversidad genética y estado de conservación. Cuadernos de Herpetología, 28, 83–110.
Cadby, C. D., Jones, S. M., & Wapstra, E. (2014). Geographical differences in maternal basking behaviour and offspring growth rate in a climatically widespread viviparous reptile. Journal of Experimental Biology, 217, 1175–1179.
Cei, J. M. (1986). Reptiles del Centro-oeste y Sur de la Argentina. Herpetofauna de las Zonas Aridas y Semiáridas, first ed. Torino: Museo Regionale di Scienze Naturali, Monografía IV.
Charland, M. B. (1995). Thermal consequences of reptilian viviparity: Thermoregulation in gravid and nongravid garter snakes (Thamnophis). Journal of Herpetology, 29, 383–390.
Charland, M. B., & Gregory, P. T. (1990). The influence of female reproductive status on thermoregulation in a viviparous snake, Crotalus viridis. Copeia, 4, 1089–1098.
Chiaraviglio, M. (2006). The effects of reproductive condition on thermoregulation in the Argentina Boa Constrictor (Boa constrictor occidentalis) (Boidae). Herpetological Monographs, 20, 172–177.
Crews, D., & Gans, C. (1992). The interaction of hormones, brain, and behavior: An emerging discipline in herpetology. In C. Gans & D. Crews (Eds.), Biology of the Reptilia (pp.<background-color:#96C864;> </background-color:#96C864;>1–23). Chicago: The University of Chicago Press.
Cruz, F. B., Belver, L., Acosta, J. C., Villavicencio, H. J., Blanco, G., & Cánovas, M. G. (2009). Thermal biology of Phymaturus lizards: Evolutionary constraints or lack of environmental variation? Zoology, 112, 425–432.
Dayananda, B., Gray, S., Pike, D., & Webb, J. K. (2016). Communal nesting under climate change: Fitness consequences of higher incubation temperatures for a nocturnal lizard. Global Change Biology, 22, 2405–2414.
Deeming, D. C. (2004). Reptilian incubation: Environment, evolution and behaviour. Nottingham: Nottingham University Press.
Donoso-Barros, R. (1973). Una nueva lagartija magallánica (Reptilia, Iguanidae). Neotropica, 19, 163–164.
Du, W.-G., & Ji, X. (2003). The effects of incubation thermal environments on size, locomotor performance and early growth of hatchling soft-shelled turtles, Pelodiscus sinensis. Journal of Thermal Biology, 26, 279–286.
Elphick, M. J., & Shine, R. (1998). Longterm effects of incubation temperatures on the morphology and locomotor performance of hatchling lizards (Bassiana duperreyi, Scincidae). Biological Journal of the Linnean Society, 63, 429–447.
Fernández, J. B., Medina, S. M., Kubisch, E. L., Manero, A. A., Scolaro, J. A., & Ibargüengoytía, N. R. (2015). Female reproductive biology of the lizards Liolaemus sarmientoi and L. magellanicus from the southern end of the world. Herpetological Journal, 25, 101–108.
Fernández, J. B., Smith, J. Jr., Scolaro, A., & Ibargüengoytía, N. R. (2011). Performance and thermal sensitivity of the southernmost lizards in the world, Liolaemus sarmientoi and Liolaemus magellanicus. Journal of Thermal Biology, 36, 15–22.
Gao, J. F., Qu, Y. F., Luo, L. G., & Ji, X. (2010). Evolution of reptilian viviparity: A test of the maternal manipulation hypothesis in a temperate snake, Gloydius brevicaudus (Viperidae). Zoological Science, 27, 248–255.
Guillette, J. L. Jr., DeMarco, V., & Palmer, B. D. (1991). Exogenous progesterone or indomethacin delays parturition in the viviparous lizard Sceloporus jarrovi. General and Comparative Endocrinology, 81, 105–112.
Hertz, P. E., Huey, R., & Stevenson, R. D. (1993). Evaluating temperature regulation by field-active ectotherms: The fallacy of the inappropriate question. American Naturalist, 142, 796–818.
Huey, R. B., Deutsh, C. A., Tewksbury, J. J., Vitt, L. J., Hertz, P. E., Álvarez-Pérez, H. J., & Garland, T. (2009). Why tropical forest lizards are vulnerable to climate warming. Proceedings of the Royal Society of London B: Biological Sciences, 276, 1939–1948.
Ibargüengoytía, N. R., Medina, S. M., Fernández, J. B., Gutiérrez, J. A., Tappari, F., & Scolaro, A. (2010). Thermal biology of the southernmost lizards in the world: Liolaemus sarmientoi and Liolaemus magellanicus from Patagonia, Argentina. Journal of Thermal Biology, 35, 21–27.
Ji, X., Chen, F., Du, W. G., & Chen, H. L. (2003). Incubation temperature affects hatchling growth but not sexual phenotype in the Chinese soft-shelled turtle, Pelodiscus sinensis (Trionychidae). Journal of Zoology, 261, 409–416.
Ji, X., Gao, J. F., & Han, J. (2007a). Phenotypic responses of hatchlings to constant versus fluctuating incubation temperatures in the multi-banded krait, Bungarus multicintus (Elapidae). Zoological Science, 24, 384–390.
Ji, X., Lin, C. X., Lin, L. H., Qiu, Q. B., & Du, Y. (2007b). Evolution of viviparity in warm-climate lizards: An experimental test of the maternal manipulation hypothesis. Journal of Evolutionary Biology, 20, 1037–1045.
Kubisch, E. L., Corbalán, V., Ibargüengoytía, N. R., & Sinervo, B. (2016). Local extinction risk of three species of lizards from Patagonia as a result of global warming. Canadian Journal of Zoology, 94, 49–59.
Kubisch, E. L., Fernández, J. B., & Ibargüengoytía, N. R. (2011). Is locomotor performance optimized at preferred body temperature? A study of Liolaemus pictus argentinus from northern Patagonia, Argentina. Journal of Thermal Biology, 36, 328–333.
Lambert, S. M., & Wiens, J. J. (2013). Evolution of viviparity: A phylogenetic test of the cold climate hypothesis in phrynosomatid lizards. Evolution, 67, 2614–2630.
Legendre, P. (2015). Lmodel2: Model II Regression. R package version 1.7-2. https://cran.rproject.org/web/packages/lmodel2/index.html/.
Lemus, D., Illanes, J., Fuenzalida, M., Paz-De la Vega, Y., & García, M. (1981). Comparative analysis of the development of the lizard, Liolaemus tenuis tenuis. II A series of normal postlaying stages in embryonic development. Journal of Morphology, 169, 337–349.
Li, H., Qu, Y. F., Hu, R. B., & Ji, X. (2009). Evolution of viviparity in cold-climate lizards: Testing the maternal manipulation hypothesis. Evolutionary Ecology, 23, 777–790.
Lin, C. X., Zhang, L., & Ji, X. (2008). Influence of pregnancy on locomotor and feeding performances of the skink, Mabuya multifasciata: Why do females shift thermal preferences when pregnant? Zoology, 111, 188–195.
Lindgren, J. (2004). UV-lamps for terrariums: Their spectral characteristics and efficiency in promoting vitamin D3 synthesis by UVB irradiation. Herpetomania, 2004, 13–20.
Lorioux, S., DeNardo, D. F., Gorelick, R., & Lourdais, O. (2012). Maternal influences on early development: Preferred temperature prior to oviposition hastens embryogenesis and enhances offspring traits in the Children’s python, Antaresia childreni. Journal of Experimental Biology, 215, 1346–1353.
Lourdais, O., Shine, R., Bonnet, X., Guillon, M., & Naulleau, G. (2004). Climate affects offspring phenotypes in a viviparous snake. Oikos, 104, 551–560.
Mathies, T., & Andrews, R. M. (1997). Influence of pregnancy on the thermal biology of the Lizard, Scleroporus jarrovi: Why do pregnant females exhibit low body temperatures? Functional Ecology, 11, 498–507.
Medina, M., Gutiérrez, J., Scolaro, A., & Ibargüengoytía, N. R. (2009). Thermal responses to environmental constraints in two populations of the oviparous lizard Liolaemus bibronii in Patagonia, Argentina. Journal of Thermal Biology, 37, 579–586.
Medina, M., Scolaro, A., Méndez-De la Cruz, F. R., Sinervo, B. R., Miles, D. B., & Ibargüengoytía, N. R. (2012). Thermal biology of genus Liolaemus: A phylogenetic approach reveals advantages of the genus to survive climate change. Journal of Thermal Biology, 37, 579–586.
Mell, R. (1929). Beiträge zur fauna sinica. IV. Grundzüge einer okologie der chinesischen reptilien und einer herpetologischen tiergeographie Chinas. Berlin: Walter de Gruyter.
Moreno-Azócar, D. L., Vanhooydonck, B., Bonino, M. F., Perotti, M. G., Abdala, C. S., Schulte, J. A., & Cruz, F. B. (2013). Chasing the Patagonian sun: Comparative thermal biology of Liolaemus lizards. Oecologia, 171, 773–788.
O’Donnell, R. P., & Arnold, S. J. (2005). Evidence for selection on thermoregulation: Effects of temperature on embryo mortality in the garter snake Thamnophis elegans. Copeia, 2005, 929–933.
Olsson, M., Shine, R., & Bak-Olsoon, E. (2000). Locomotor impairment of gravid lizards: Is the burden physical or physiological? Journal of Evolutionary Biology, 13, 263–268.
Packard, G. C., Tracy, C. R., & Roth, J. J. (1977). The physiological ecology of reptilian eggs and embryos, and the evolution of viviparity within the class Reptilia. Biological Reviews, 52, 71–105.
Paranjpe, D. A., Bastiaans, E., Patten, A., Cooper, R. D., & Sinervo, B. (2013). Evidence of maternal effects on temperature preference in side-blotched lizards: Implications for evolutionary response to climate change. Ecology and Evolution, 3, 1977–1991.
Peig, J., & Green, A. J. (2009). New perspectives for estimating body condition from mass/length data: The scaled mass index as an alternative method. Oikos, 118, 1883–1891.
Peig, J., & Green, A. J. (2010). The paradigm of body condition: A critical reappraisal of current methods based on mass and length. Functional. Ecology, 24, 1323–1332.
Qualls, C. P., & Andrews, R. M. (1999). Cold climates and the evolution of viviparity in reptiles: Cold incubation temperatures produce poor-quality offspring in the lizard, Sceloporus virgatus. Biological Journal of the Linnean Society, 67, 353–376.
Qualls, F. J., & Shine, R. (1998). Lerista bougainvillii, a case study for the evolution of viviparity in reptiles. Journal of Evolutionary Biology, 11, 63–78.
R Core Team. (2015). R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing. http://www.R-project.org/.
Rock, J., & Cree, A. (2003). Intraspecific variation in the effect of temperature on pregnancy in the viviparous gecko Hoplodactylus maculatus. Herpetologica, 59, 8–22.
Rodríguez-Díaz, T., González, F., Ji, X., & Braña, F. (2010). Effects of incubation temperature on hatchling phenotypes in an oviparous lizard with prolonged egg retention: Are the two main hypotheses on the evolution of viviparity compatible? Zoology, 113, 33–38.
Schulte, J. A., Macey, J. R., Espinoza, R. E., & Larson, A. (2000). Phylogenetic relationships in the iguanid lizard genus Liolaemus: Multiple origins of viviparous reproduction and evidence for recurring Andean vicariance and dispersal. Biological Journal of the Linnean Society, 69, 75–102.
Scolaro, J. A., & Cei, J. M. (1997). Systematic status and relationships of Liolaemus species of the archeforus and kingii groups: Morphological and taxonumerical approach (Reptilia: Tropiduridae). Bolletin del Museo Regionale di Scienze Naturali Torino, 15, 369–406.
Shine, R. (1983). Reptilian viviparity in cold climates: Testing the assumptions of an evolutionary hypothesis. Oecologia, 57, 397–405.
Shine, R. (1985). The evolution of viviparity in reptiles: An ecological analysis. Biol. Reptilia, 15, 605–694.
Shine, R. (1995). A new hypothesis for the evolution of viviparity in reptiles. American Naturalist, 145, 809–823.
Shine, R. (2004). Does viviparity evolve in cold climate reptiles because pregnant females maintain stable (not high) body temperatures? Evolution, 58, 1809–1818.
Shine, R., & Bull, J. J. (1979). The evolution of live-bearing in lizards and snakes. American Naturalist, 1979, 905–923.
Shine, R., & Downes, S. J. (1999). Can pregnant lizards adjusts their offspring phenotypes to environmental conditions? Oecologia, 119, 1–8.
Shine, R., & Elphick, M. J. (2001). The effect of short-term weather fluctuations on temperatures inside lizard nests, and on the phenotypic traits of hatchling lizards. Biological Journal of the Linnean Society, 72, 555–565.
Sinervo, B., Hedges, R., & Adolph, S. C. (1991). Decreased sprint speed as a cost of reproduction in the lizard Sceloporus occidentalis: Variation among populations. Journal of Experimental Biology, 155, 323–336.
Stewart, J. R., & Blackburn, D. G. (2014). Viviparity and placentation in lizards. In J. L. Rheubert, D. S. Siegel & S. E. Trauth (Eds.), Reproductive biology and phylogeny of lizards and tuatara (pp. 448–563). Vol 10 of Series: B. G. M. Jamieson (Ed.), Reproductive Biology and Phylogeny. Boca Raton: CRC Press.
Tu, M. C., & Hutchison, V. H. (1994). Influence of pregnancy on thermoregulation of water snakes (Nerodia rhombifera). Journal of Thermal Biology, 19, 255–259.
Van Damme, R., Bauwens, D., & Verheyen, R. F. (1989). Effect of relative clutch mass on sprint speed in the lizard Lacerta vivipara. Journal of Herpetology, 23, 459–461.
Wang, Z., Lu, H. L., Ma, L., & Ji, X. (2014). Viviparity in high-altitude Phrynocephalus lizards is adaptive because embryos cannot fully develop without maternal thermoregulation. Oecologia, 174, 639–649.
Wapstra, E. (2000). Maternal basking opportunity affects juvenile phenotype in a viviparous lizard. Functional Ecolology, 14, 345–352.
Wapstra, E., Uller, T., While, G. M., Olsson, M., & Shine, R. (2010). Giving offspring a head start in life: Field and experimental evidence for selection on maternal basking behaviour in lizards. Journal of Evolutionary Biology, 23, 651–657.
Webb, J. K., Shine, R., & Christian, K. A. (2006). The adaptive significance of reptilian viviparity in the tropics: Testing the maternal manipulation hypothesis. Evolution, 60, 115–122.
Woolrich-Piña, G. A., Smith, G. R., Lemos-Espinal, J. A., & Ramírez-Silva, J. P. (2015). Do gravid female Anolis nebulosus thermoregulate differently than males and non-gravid females? Journal of Thermal Biology, 52, 84–89.
Yan, X. F., Tang, X. L., Yue, F., Zhang, D. J., Xin, Y., Wang, C., & Chen, Q. (2011). Influence of ambient temperature on maternal thermoregulation and neonate phenotypes in a viviparous lizard, Eremias multiocellata, during the gestation period. Journal of Thermal Biology, 36, 187–192.
Acknowledgements
We thank A. Desimone, R. Fernández and D. D’Arielli for logistical support and for their help in the release of lizards to their capture site. We also thank to M. Cabral, L. Rodriguez and A. Scolaro for providing the access to the study site. This research was supported by the Universidad Nacional del Comahue (CRUB) and the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET, PIP 100271 and RD2702-12).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Fernández, J.B., Kubisch, E.L. & Ibargüengoytía, N.R. Viviparity Advantages in the Lizard Liolaemus sarmientoi from the End of the World. Evol Biol 44, 325–338 (2017). https://doi.org/10.1007/s11692-017-9410-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11692-017-9410-2