Abstract
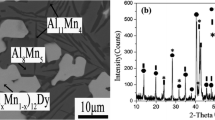
The isothermal section of the Dy-Mn-As ternary system at 773 K has been investigated by using x-ray diffraction, scanning electron microscopy and energy dispersive spectroscopy. This isothermal section consists of 12 single-phase regions, 21 two-phase regions and 10 three-phase regions. The highest solid solubility of Dy in MnAs is about 0.65 at.%Dy, and that of Mn in DyAs is less than 0.79 at.%Mn. The maximum solubility of As in DyMn2 is less than 0.33 at.%As. No ternary compound was found to exist in this section at 773 K.















Similar content being viewed by others
References
N.K. Sun, W.B. Cui, D. Li, D.Y. Geng, F. Yang, and Z.D. Zhang, Giant Room-Temperature Magnetocaloric Effect in Mn1-xCrxAs, Appl. Phys. Lett., 2008, 92(7), p 072504
W.B. Cui, W. Liu, X.H. Liu, S. Guo, Z. Han, X.G. Zhao, and Z.D. Zhang, Beneficial Effect of Minor Al Substitution on the Magnetocaloric Effect of Mn1−xAlxAs, Mater. Lett., 2009, 63(6), p 595-597
D.L. Rocco, A. de Campos, A.M.G. Carvalho, L. Caron, A.A. Coelho, S. Gama et al., Ambient Pressure Colossal Magnetocaloric Effect in Mn1−xCuxAs Compounds, Appl. Phys. Lett., 2007, 90(24), p 242507
M. Balli, D. Fruchart, D. Gignoux, C. Dupuis, A. Kedous-Lebouc, and R. Zach, Giant Magnetocaloric Effect in Mn1-x(Ti0.5V0.5)xAs: Experiments and Calculations, J. Appl. Phys., 2008, 103(10), p 103908
X.X. Zhang, F.W. Wang, and G.H. Wen, Magnetic Entropy Change in RCoAl (R = Gd, Tb, Dy, and Ho) Compounds: Candidate Materials for Providing Magnetic Refrigeration in the Temperature Range 10 K to 100 K, J. Phys. Condens. Matter, 2001, 13(31), p L747
K.A. Gschneidner, Jr., and V.K. Pecharsky, Rare Earths and Magnetic Refrigeration, J. Rare Earths, 2006, 24(6), p 641-647
A.K. Pathak, I. Dubenko, S. Stadler, and N. Ali, Magnetic, Magnetocaloric, and Magnetotransport Properties of RCo1.8Mn0.2 (R = Er, Ho, Dy, and Tb) Compounds, J. Magn. Magn. Mater., 2011, 323(20), p 2436-2440
H. Landolt and R. Börnstein, Phase Equilibria, Crystallographic and Thermodynamic Data of Binary Alloys, Vol 3, Springer, Berlin, 1993
H.R. Kirchmayr and W. Lugscheider, Constitution of Binary Alloys of Gadolinium, Dysprosium, Holmium, and Erbium with Manganese, Z. Metallkd., 1967, 58, p 185
H. Okamoto, Dy-Mn (Dysprosium-Manganese), J. Phase Equilib. Diffus., 2011, 32, p 167
H. Okamoto and T.B. Massalski, Binary Alloy Phase Diagrams Requiring Further Studies, J. Phase Equilib., 1994, 15(5), p 500
M. Hansen and K. Anderko, Constitution of Binary Alloys, McGraw-Hill, New York, 1958
K.A. Gschneidner, Jr., and F.W. Calderwood, The Arsenic-Rare Earth Systems, Bull. Alloy Phase Diagr., 1986, 7, p 277
R. Hanks and M.M. Faktor, Quantitative Application of Dynamic Differential Calorimetry. Part 2—Heats of Formation of the Group 3 A Arsenides, Trans. Faraday Soc., 1967, 63, p 1130
Pierre Villars and Lauriston D. Calvert, Pearson’s Handbook of Crystallographic Data for Intermetallic Phases, Vol 2, American Society for Metals, Metals Park, OH, 1985
P. Villars, Pearson’s Handbook Desk Edition: Crystallographic Data for Intermetallic Phases, ASM International, Materials Park, OH, 1997
H. Landolt and R. Börnstein, Phase Equilibria, Crystallographic and Thermodynamic Data of Binary Alloys, Springer, Berlin, 1993
M.F. Hagedorn and W. Jeitschko, Synthesis and Crystal Structure of Mn4As3 and Its Relation to Other Manganese Arsenides, J. Solid State Chem., 1995, 119(2), p 344-348
L.Η. Dietrich, W. Jeitschko, and Μ.H. Möller, The Crystal Structure of Mn3As2 (I), Cryst. Mater., 1989, 190(1-4), p 259-270
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant Nos. 50261002, 50661002 and 51161005) and National Basic Research Program of China (No. 2014CB643703).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gu, Z.F., Yu, S.L., Xu, C.F. et al. Experimental Investigation of the Phase Relations of the Dy-Mn-As System at 773 K. J. Phase Equilib. Diffus. 36, 306–316 (2015). https://doi.org/10.1007/s11669-015-0384-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11669-015-0384-4