Abstract
A three-dimensional (3D) parallel process model simulating ironmaking blast furnaces (BFs) has been developed using computational fluid dynamics (CFD). It explicitly describes the layered burden and cohesive zone (CZ), gas and liquid re-distribution near raceways, trickling liquid flow in the CZ and dripping zone, and stockline variation. The applicability of the model is confirmed by the reasonable agreement between predicted and measured in-furnace states and global performance under experimental and industrial conditions. Using this model, the 3D characteristics of in-furnace states for a 5000 m3 commercial BF with 40 tuyeres are revealed. Also, it is used to assess the commonly used slot, axisymmetric, sector and full 3D models, which may treat burden distribution as well as gas and liquid flows around raceways differently. The results reveal that the sector and full 3D models are nearly the same; the slot model over-predicts the coke rate up to 13 kg/tHM, and the axisymmetric model gives slightly higher productivity and liquid temperature. These differences are clarified by analyzing model simplifications and their impacts on in-furnace states.
Similar content being viewed by others
Introduction
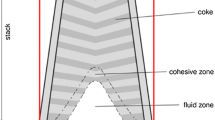
Blast furnace (BF) ironmaking is the most important technology for reducing hot metal (HM) from ferrous materials. In modern BF ironmaking, iron ore and coke particles are often charged alternatively into a furnace via a top charging system, forming layered burden structures within the throat. Meanwhile, the oxygen-rich hot air introduced from tuyeres at the lower part creates void zones known as raceways, where the reducing gas resulting from the combustion of coke and pulverized coal is re-distributed before flowing upward. During the burden descent, the ore is reduced and melts in the cohesive zone (CZ), forming liquid slag and iron by the ascending reducing gas. The liquids then percolate through the coke bed in the form of droplets/rivulets in the dripping zone to the hearth for periodic drainage. To design and operate BFs efficiently and reliably, it is necessary to fully understand these complicated local phenomena and their impacts on BF performance.
In the past years, various methods have been developed to study the BF ironmaking process, such as theoretical analysis, dissection studies, in situ measurements, physical experiments and mathematical modeling.[1,2] BFs are operated under an extremely harsh environment, involving intensive interactions among gas, solid and liquid in terms of flow and heat and mass transfer at high temperature and pressure. Therefore, in-furnace states of industrial BFs are extremely difficult if not impossible to access via physical measurements. To some extent, this problem can be overcome using a small experimental BF (e.g., ~ 9 m3 BFs,[3,4]) which in principle functions in a similar way to a real BF. However, it is unaffordable for most investigators to build up, run and maintain such an experimental platform. Moreover, the scale-up issue from experimental BFs to industrial ones has not been fully resolved yet. All these problems can in principle be overcome by numerical modeling and simulation.
Numerical BF models, as reviewed by different investigators,[2,5,6,7,8,9] can be discrete or continuum based according to the solid phase and are represented by the discrete element model (DEM) and two-fluid (or multi-fluid) model (TFM), respectively. The latter is solved by traditional computational fluid dynamics (CFD) over computational cells that are larger than particles but still very small compared with process equipment. Therefore, it is computationally convenient and efficient and has been mainly used to predict the primary phenomena related to gas, solid, liquid and powder and the global performance of BFs, especially at an industrial scale. Some examples include the CFD modeling of solid flow and deadman,[10,11,12,13,14] chemical reactions,[15,16] liquid flow in the form of droplets/rivulets,[17,18,19] transient behaviors,[20,21] layered burden and CZ,[22,23,24,25,26] powder flow,[15,27,28] stockline variation,[26] liquid drainage,[29] raceway formation[30] and raceway combustion.[31] With different combinations of these developments, different CFD BF process models have been developed to simulate the region between the burden surface and slag surface.[4,15,20,22,23,26,27,32,33,34,35,36]
Generally, the previous CFD process models focused on some specific primary phenomena. To date, the interactions of trickling liquid flow with layered CZ and raceways have been rarely considered. Also, in many studies the productivity, which varies with BF conditions in accordance with stockline variation, was empirically pre-set other than predicted. As a result, the productivity may not match the BF conditions corresponding to the common BF practice. Additionally, the recognized re-distribution of the reducing gas by raceways was often simplified or ignored because of high computational requirements for explicitly simulating raceways. So far, the majority of CFD BF process models are two-dimensional (2D) slot or axisymmetric models, which neglect the flow, heat and mass in the circumferential direction.
The in-furnace states of BF present somewhat 3D characteristics. One example is the flows of reducing gas and liquids near raceways.[37] To date, it is not clear how the prediction reliability of a 2D BF process model is quantitatively affected by different simplifications in describing 3D characteristics of in-furnace states. Besides, practical BF operations induce the development of 3D in-furnace states because of, for example, non-uniform burden charging in the radial and circumferential directions[38,39,40] and non-uniform operations at tuyeres.[41] This situation also applies to some new technologies such as oxygen BFs, where belly and shaft gas injections are introduced at only a few locations in the circumferential direction,[3,4] inevitably causing 3D transport phenomena. As such, it is useful to develop a CFD process model for analyzing 3D characteristics of in-furnace states and their impacts on BF performance under different BF conditions.
To date, a few 3D CFD BF process models have been developed but with different nontrivial simplifications. Takatani et al.[33] proposed a 3D process model to study different BF profiles, where 1D uniform liquid flow was assumed. de Castro et al.[32] presented a 3D model simulating the flow and thermochemical behaviors of gas, liquid, solid and powder within a BF, which was later extended to model powders of different types.[27] In these 3D models, raceways were not explicitly considered. However, Shen et al.[37] presented a sector model considering one tuyere explicitly and demonstrated that the liquid flow around a raceway is significantly affected by the existence of raceway. In that work, the raceway was represented by a spherical cavity, as done by Austin et al.[15,34] in a 2D model. Nevertheless, to date, 3D BF process models explicitly simulating layered burden and CZ, trickling liquid flow and their interactions cannot be found in the literature.
In this article, a 3D CFD parallel BF process model is developed and validated to describe the flow and thermochemical behaviors and global performance of BF. To be realistic, the model is integrated with the modeling of the layered burden and CZ, trickling flow in the CZ and dripping zone, and gas and liquid re-distribution near raceways; it also treats the productivity as an output corresponding to stockline variation. This integration is useful to describe the interactions among different phenomena and has not been fully realized in all the previous 2D and 3D CFD BF process models. The prediction reliability of the model has been tested under different conditions. On this basis, the 3D characteristics of in-furnace states are demonstrated by simulating a 5000 m3 commercial BF with 40 tuyeres. Finally, using this model, the commonly used slot, axisymmetric, sector and full 3D simulation setups are assessed to clarify their differences.
Model Description
The current 3D model is based on the 2D slot models reported elsewhere.[22,26] In this work, new efforts are made to model the 3D characteristics of the layered burden and CZ, trickling liquid flow, and gas and liquid re-distribution by raceways. For brevity, below we only describe the key features of the model. However, new developments will be emphasized.
Governing Equations
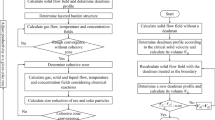
This model is a steady-state multi-fluid model considering the region in a BF from the liquid surface in the hearth up to the burden surface in the throat. The phases considered are gas, solid (i.e., burden materials) and liquid (i.e., hot slag and metal), which are all treated as interpenetrating continua. Each phase consists of one or more components, and each component has its composition and physical properties. The phases are described, respectively, by the separate conservation equations of mass, momentum and enthalpy, with key chemical reactions considered. Tables I and II summarize the governing equations, chemical reactions and associated transport coefficients.
Modeling 3D Layered Burden and Cohesive Zone
The layered structure of the burden materials and CZ determines the permeability, fluid flow, gas usage, thermal and chemical efficiency, and hot-metal quality and thus is of critical importance to the efficient operation of a BF. With the development of computational technology and the numerical method, the consideration of layered burden and CZ in CFD process models has probably become common but only for 2D models.[22,23,24,25,26] In this work, our recent efforts[22,26] in this respect are extended from 2D to 3D cases.
Modeling layered structure requires tracking coke and ore layers. Different methods have been used, including, for example, the timeline tracking model,[22,26] geometric profile model,[24] potential flow model[23,25] and VOF model.[42] In principle, all these models can be used to track the transient motion of coke and ore layers, which is, however, computationally demanding, especially when the temporal variations of in-furnace states are also considered. Generally, transient behaviors become important for unstable operations or during the start-up and transition periods. The previous studies[22,24] show that when the production is stable, the simulations with and without considering the transient motion of the layered burden give nearly the same results. This is in line with the fact that BF performance does not change much during a smooth burden descent. In this study, the timeline tacking model is used. To be computationally efficient in modeling stable production, only the layered burden structure at a specific time is considered. This treatment has been proved valid to reproduce key BF phenomena under different conditions.[4,22,23,24,26,36,43]
Timelines were calculated via dividing cell sizes by their solid velocities in our previous models.[22,26] This calculation is convenient but applicable only to simple grid arrangements in a 2D model. Instead, it is done by solving the equation of timeline (Table I) for 3D models twice; this is valid for complex mesh topology. In the first calculation, timelines at the BF top are initiated with zero. The predicted timelines are then divided by the total batch time \( \left( {t_{\text{batch}} } \right) \) for charging one coke layer and one ore layer, which gives a series of integers to label CFD cells. Here, \( t_{\text{batch}} \) is calculated by \( {{m_{\text{batch}} } \mathord{\left/ {\vphantom {{m_{\text{batch}} } {\rho_{\text{bulk}} u_{\text{feed}} A_{\text{throat}} }}} \right. \kern-0pt} {\rho_{\text{bulk}} u_{\text{feed}} A_{\text{throat}} }} \), where \( m_{\text{batch}} \left( { = m_{\text{batch,ore}} + m_{\text{batch,coke}} } \right) \), \( \rho_{\text{bulk}} \) and \( u_{\text{feed}} \) are the weight of one ore layer and one coke layer, bulk density and feed velocity, respectively; \( A_{\text{throat}} \) is the cross-sectional area of the BF throat. Each integer corresponds to a layer consisting of one coke layer and one ore layer. In the second calculation, the values of timelines at the BF top change to the radially varied batch time of ore. This is done by multiplying the total batch time by the ore volumetric ratios in the radial direction. Then, CFD cells are labeled using another set of integers. The regions with the same integers from two timeline calculations can be identified as coke layers, while the remaining regions are ore layers. Note that based on the proposed method, the transient burden loading can be considered by varying the timelines at the BF top from 0 to \( t_{\text{batch}} \).
With coke and ore layers identified, CZ is modeled following the works reported elsewhere.[22,26] In general, ore particles inside a CZ experience dramatic physicochemical change when transforming from lumpy to softening to half-molten states before finally melting down. Accordingly, ore properties, such as particle size, voidage and other thermochemical parameters, are changed. In this work, CZ is defined to start and finish within the temperature range of 1473 K to 1673 K (1200 °C to 1400 °C). Three ore states of the CZ are specified according to the shrinkage ratio, \( {\text{Sh}}_{r}^{ * } \), which is proportional to the solid temperature and has a value from 0.0 to 1.0. The shrinkage ratio is also applied to determine the particle size and voidage of iron-bearing materials in the CZ. This way allows simulating the effect of the melting ore and “coke window” on gas re-distribution. Moreover, the solid conduction and gas-solid heat transfer coefficients are specified respectively according to different heat and mass transfer mechanisms in each of the ore states, as detailed elsewhere.[22,26] A similar treatment is extended to the lumpy zone (the region above the CZ) consisting of ore and coke layers.
Modeling 3D Trickling Liquid Flow
Liquid slag and iron generated in the CZ trickle down or drop in “icicles” through the coke bed to the hearth. Their discontinuous or discrete characteristics are difficult to describe by a traditional CFD model. Different methods have been proposed to overcome this problem. For example, based on discrete methods, Natsui et al.[44,45,46,47,48,49,50] performed vigorous simulations of the trickling flow through packed beds over a range of conditions. Such studies are useful to understand the liquid flow in the dripping zone, although further dedicated efforts are needed to couple a discrete method with a BF process model. Another method is based on the so-called force balance model,[18,19] which describes the liquid flow as rivulets or droplets under the influences of gravity, gas drag and bed resistance (see Table I). In this model, the stochastic phenomenon related to the liquid dispersion can be also considered. Based on the force balance model with liquid dispersion ignored, Chew et al.[17] correlated the drag forces with liquid and packing properties, which was then incorporated into a process model.[35] This liquid flow model has also been adopted by Dong et al.[22] To date, a force balance model considering liquid dispersion has not been implemented in a process model to predict in-furnace states and global performance of BFs. As the first step towards modeling stochastic liquid trickling flow, the force balance model of Wang et al.[18] is taken, where the coefficients of the drag force model were estimated by fitting experimental data.
In the model, the main flow represented by \( \overline{{\mathbf{U}}}_{\text{l}} \) (see Figure 1(a)) is described by:
where Ug is the gas velocity; \( \rho_{\text{g}} \) and \( \rho_{\text{l}} \) are the gas and liquid densities. \( C_{DG} \) and \( C_{DS} \) are the drag coefficients. Ag−l and As−l are the effective contact areas, and hd is the dynamic holdup and calculated by
Schematic illustration of the liquid dispersion model: (a) previous model (adapted from Ref. [18]); (b and c) present model
Although Eq. [1] or the like can be directly used to determine the liquid velocity,[22,35] a stochastic velocity US is introduced to consider the stochastic characteristic of the droplets or rivulets flowing through the complex pore geometry formed by the packing particles. Thus, the liquid velocity is expressed as:
where US is assumed normal to the main flow direction and varies from − US0 to US0 (see Figure 1(a)). US0 is the maximum stochastic velocity related to bed properties, calculated by \( 0.44\sqrt {{g \mathord{\left/ {\vphantom {g {\phi_{2} }}} \right. \kern-0pt} {\phi_{2} }}} \).[18] To determine US, the mass balance for the liquid flow is applied:
where k is the number of cell faces surrounding the Oth point, Fi and FO are the mass flow rates of the liquid flowing out of the ith and Oth point, respectively, and Pi,O is the proportion of liquid flowing from the ith to Oth point. Similarly, PO,i represents the flow from the Oth to ith point. Both PO,i and Pi,O are determined by integrating \( f(\theta ) \) over the area covered by US. \( f(\theta ) \) is the probability distribution function of stochastic velocity:
where \( \tan \alpha = {{\left| {{\mathbf{U}}_{\text{S0}} } \right|} \mathord{\left/ {\vphantom {{\left| {{\mathbf{U}}_{\text{S0}} } \right|} {\left| {\overline{{\mathbf{U}}}_{\text{l}} } \right|}}} \right. \kern-0pt} {\left| {\overline{{\mathbf{U}}}_{\text{l}} } \right|}} \). For example, when i = 4, \( P_{O , 4} = \int_{{\theta_{3} }}^{{\theta_{4} }} {f(\theta )} {\text{d}}\theta \), where the definitions of \( \theta_{3} \)and \( \theta_{4} \) are shown in Figure 1(a). Correspondingly, the mass flux through the bottom cell face is calculated by\( F_{O} P_{O,4} - F_{4} P_{4,O} \), which is needed for solving the liquid temperature.
It should be noted that the integration of Eq. [5] is difficult to achieve in a 3D model. Thus, a simplified method is proposed, which directly calculates \( P_{{{\text{O,}}i}} \) rather than integrating a probability distribution function:
where \( \lambda_{i} = \left\{ \begin{aligned} \cos \beta_{i} { (}\cos \beta_{i} \ge 0 )\hfill \\ 0{ (}\cos \beta_{i} { < }\, 0 )\hfill \\ \end{aligned} \right. \); \( \beta_{i} \) is the angle between the liquid main flow \( \overline{{\mathbf{U}}}_{\text{l}} \) and the line connecting the Oth to the ith point. The values of \( \alpha \) are dependent on bed properties and liquid main flow velocities, determining the liquid dispersion range. Here, \( \lambda_{i}^{{{{(\pi } \mathord{\left/ {\vphantom {{(\pi } \alpha }} \right. \kern-0pt} \alpha })^{2} }} \) is plotted against \( \beta_{i} \) for different values of \( \alpha \) (see Figure 1(c)). This indicates that a larger \( \beta_{i} \) leads to a stronger liquid flow from the Oth into the ith point. When \( \alpha \) is smaller, the liquid dispersion is less significant. Thus, as done by Eqs. [4] and [6] is linked with different bed properties. More importantly, the latter can straightforwardly extend to 3D from its 2D form.
The treatments at boundaries including CZ layers, walls and liquid outlets are still the same as done by Wang et al.[18] When the liquid from an upper layer of CZ reaches the top boundary of the lower layer, it is assumed to be uniformly distributed in left and right directions in a 2D simulation. This changes to four directions in a 3D case. Also, the liquid phase is assumed not to flow into the raceway because of the high-speed gas flow there. Thus, the raceway boundary is an impermeable wall for the liquid flow.
Numerical Solution
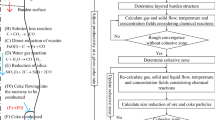
The well-established sequential solution procedure is employed to calculate the coupled multi-phase momentum, heat and mass transfer, and chemical reactions, as shown in Figure 2. Because the fluid flow and heat and mass transfer are solved in sequence, their numerical solutions are in principle similar to those used for a single-phase flow, whose convergence conditions have been well discussed and documented in the literature.[51] However, a general convergence criterion as used in CFD cannot ensure that the current model generates physically meaningful results. Our tests indicated that the convergence condition of the model is determined mainly by the CZ position where both flow and thermochemical behaviors are significantly different from those in other regions of the BF. In particular, a solid temperature range is used to define the shape and position of the CZ, which in turn affects the solid temperature field. Therefore, a new criterion is introduced in this work for simple use. That is, the CZ profile is characterized by the volume ratio of the lumpy zone and dripping zone. The solution is converged only when the difference of the ratios in two consecutive iterations is less than a specific value, which is set to 0.1 pct in this work. With this treatment, CZ and the associated results have trivial variations when the solution is converged.
Another important consideration for solution convergence is productivity. It is established that productivity is one of key process parameters. It varies related to stockline variation and is dependent on the material, geometrical and operational conditions. Corresponding to practical BF operations, productivity is treated as an output in the current model rather than as an input as in most of the previous models. To this effect, the burden charge rate is automatically adjusted according to the carbon balance illustrated in Figure 2 to adapt to given BF conditions for achieving specific in-furnace states. This way is like the consideration of stockline variation. For a converged solution, the difference between the coke combusted in raceways and the coke following into raceway is < 0.1 kg/tHM to meet the coke balance requirement.
Simulation and Boundary Conditions
Table III lists the conditions considered. A 9 m3 experimental BF with 3 tuyeres[3] and a 5000 m3 industrial BF with 40 tuyeres are simulated. Figure 3 shows the computational domain and representative grid arrangement for the industrial BF. The whole domain is divided into around 9 million hexahedra (see Figure 3(b)), the sizes of which are on average 8 × 8 × 8 cm3. This allows using from four to six computational cells to represent each of the ore layers in a CZ in the vertical direction, which is necessary to capture lumpy, softening and melting states of ore. Also, at the lower part, the grid is refined to identify raceway boundaries.
The top burden conditions at the furnace throat are the inputs of a CFD process model, including the ore and coke particle sizes, ore-to-coke ratio and burden surface shape. This information is often difficult to theoretically calculate or directly measure. In this work, the top burden conditions for the industrial BF are provided by our industrial partners, who obtained this information from DEM (discrete element method) simulations under the condition considered. Such inputs are shown in Figure 4 where the ore-to-coke ratio is defined as the volume fraction of iron ore in burden materials. Also, the burden surface given by the DEM simulation is used as the top boundary of the computational domain (see Figure 3(a)). For the experimental BF, the ore-to-coke ratio is derived from the measured CZ structures, whereas uniform coke and ore particle sizes and flat top are used because of the lack of experimental details. In all simulations, the burden distribution and blast conditions are assumed to be uniform in the circumferential direction. The shape of raceways is given according to DEM simulations.[52] Their dimensions are finally determined according to the blast rate, pressure, temperature and coke size in the deadman using Nomura’s empirical correlations.[53] In addition, the iterative method as proposed by Zhang et al.[14] is adopted to determine the deadman profile during a calculation, which is outlined in Figure 2(c). In this calculation, the region with solid velocities being less than the critical velocity is defined as the deadman zone. Then, the deadman profile is treated as a boundary. On this base, a new deadman zone profile is determined in conjunction with boundary conditions at the walls, inlet and outlet. This procedure repeats until the variation of the deadman profile is trivial, which can often be achieved through a few iterations. Here, the critical velocity is defined as one ninth particle diameter per minute, as suggested by Zhang et al.[14]
In a simulation, particle sizes imposed at the furnace top are initially extended to the lower part so that the porosity distribution in the entire furnace can be obtained. Note that the size degradations of coke and ore particles and mixed layers on coke/ore interfaces are ignored because of the lack of sound theoretical methods. Also, the powder flow, which is not a current focus, is not modeled. With these simplifications, CFD process models can reasonably predict the in-furnace states and global performance of BFs under different conditions, as demonstrated by different investigators.[4,22,23,24,33,36,37,43,44] Nevertheless, further model developments are needed for different applications. During the calculation, with the identification of the lumpy zone, CZ, dripping zone and deadman, the particle sizes and porosity in these regions are determined or adjusted based on the following rules: (1) in the lumpy zone, the porosity in the ore or coke layer is a function of particle size (see Table III); (2) in the CZ, the porosity and particle size of iron-bearing materials are a function of the normalized shrinkage ratio as used elsewhere[22]; (3) in the CZ and dripping regions, the porosity in the areas occupied by coke is calculated in the same way as used for coke layers in the lumpy zone; (4) in the deadman, the coke size and porosity are assumed as, respectively, 0.025 m and 0.65 according to normal BF practice; (5) in raceways, the porosity is set to 1 and there are no particles.
Table III lists the composition, temperature and flow rate of the reducing gas generated from raceways, which are used as the inlet conditions of gas phase in simulations. This information is determined according to the local mass and heat balance for given hot blast conditions and coke properties, as done by almost all the previous CFD BF process modeling. In other words, the combustion inside raceways is not explicitly considered. However, the influences of raceways on surrounding gas and liquid flows and associated transport phenomena are considered. Burden materials containing ore, coke and flux are charged into the furnace from the top with an identical downward velocity. The solid outlets are assumed as the intersection faces between raceways and the horizontal plane through all tuyere centers for achieving high numerical stability. Facilitated with these boundaries, the model is solved by an in-house Fortran F90 code, which is developed based on the open-source parallel block structured FVM (finite volume method) single-phase CFD code CAFFA3D.[54] The number of CPUs assigned for a simulation varies from 50 to 500 depending on the cases involved. Each simulation lasts for from 3 to 30 h depending on the model setup considered.
Results and Discussion
Model Applicability
Three cases are considered to examine the validity of the current model. The first one is based on the experiments of Watakabe et al.,[3] who measured both the in-furnace states and global performance of a 9 m3 experimental BF. Table IV shows the predicted and measured adiabatic flame temperature, top gas utilization factor, top gas temperature and liquid temperature are in good agreement. Note that the simulation predicts the liquid temperature at the slag surface; however, the experiment measures the temperature at the taphole. The temperature at two locations is assumed to have a difference of ~ 50 K (50 °C) referring to field experience. Figure 5 also shows that the current model can satisfactorily predict the CZ shape and position, the radial profiles of gas temperature at two different heights as well the axial profile of reduction degree. However, it is noted that the CZ thickness varies monotonously at different heights in simulations but non-monotonously in measurements. Provided that batch weights of coke and iron ore remain unchanged, coke and ore layers are expected to become thinner during their descent to maintain mass balance. This has been reported numerically and experimentally based on cold BF models.[23,24,55,56,57,58] Due to the lack of experimental details, it is difficult to identify the reasons behind the non-monotonous variation of CZ thickness in the experimental BF. More experimental data for further validating CZ predications are needed.
The second case considers the industrial BF. Here, only global performance indicators are available for model validation. Table V shows that a reasonable agreement between measured and predicted results can also be achieved. The third case examines the effects of the blast and coke rate on BF performance based on the industrial BF. Their trends have been well established from routine operations. When the coke rate increases from 300 to 315 kg/tHM, the productivity and top gas utilization factor decrease; however, the top gas temperature and liquid temperature increase (Figure 6(a)). The increase of liquid temperature is due to a higher coke rate raising up the CZ and thus lengthening the distance for the heat exchanges between gas and liquid and between solid and liquid within the dripping zone. By contrast, as the blast rate increases by the same percent (5 pct) as for the coke rate, only the productivity apparently increases while other indicators vary slightly (Figure 6(b)). These trends are in line with the field experience. The thus far obtained results suggest that the current 3D model is reliable for simulating BF, at least qualitatively.
Three-Dimensional Characteristics of In-furnace States
Figure 7 reveals the in-furnace states of the industrial BF. Four regions inside a BF can be successfully depicted by the current model, including the lumpy zone, CZ, dripping zone and deadman (Figure 7(a)). The curved ore and coke layers following the burden surface shape gradually change to be horizontal during the burden descent from the top to the CZ. Expectedly, the CZ presents the “inverse V” shape in accordance with the burden distribution pattern with more coke loaded at the center. As expected, ore particles inside the CZ experience, in turn, the lumpy, softening and melting states and melt to liquid (Figure 7(b)).
Figures 7(c) and (d) shows that the high-speed reducing gas in raceways drastically drops its velocity after entering the coke bed and then flows upward, corresponding to the so-called second gas distribution. The gas velocity is higher in the center compared with the peripheral region because of a load of more coke into the center. This center-developed flow is essential for achieving stable operations.[43] Under the condition considered, the 3D gas flow is mainly observed in the lower part, resulting from the presence of discrete raceways.
Figure 7(e) shows the liquid generated in one layer accumulates and spreads on the top of the lower fused layer. A fraction of this liquid moves inward over a fused layer, and it combines with the liquid source generated, forming a relatively dense downward liquid flow channel. This is the so-called “icicle” flow. The remaining fraction of liquid moves outward and flows down along cohesive layers, forming significant liquid streams at the CZ root. Because of the impermeable boundary of raceways, the liquid passes around raceways and flows down through the coke bed between raceways. This causes the non-uniform distribution of liquid mass flow rates and temperature in the circumferential direction in the lower part, as shown in Figures 7(f) and (g). Clearly, the spatial distributions of liquid flow and temperature are significantly affected by the layered CZ and discrete raceways, although these effects are somewhat neglected in almost all the previous CFD BF process models.
Figure 8 shows the contours of thermochemical properties of the industrial BF. Spatial distributions of gas and solid temperatures show a similar trend. Resulting from the gas and liquid re-distributions near raceways, there the gas, solid and liquid temperatures present 3D distribution characteristics (Figures 8(a) through (c)). However, these characteristics rapidly disappear inside the upper furnace, where axisymmetric distributions are present. It is interesting to note that the iso-surfaces of gas and solid temperatures both present a “cone” shape in the shaft because of the center-developed flow. They change to an “inversed glass” in the lower part, which should be attributed to the existence of a low-permeability deadman. Interestingly, the liquid temperature presents in the form of “needles” near the CZ (see Figure 8(c)) corresponding to the feature of trickling flow. For the gas utilization factor and reduction degree shown in Figures 8(d) through (e), 3D characteristics could not be observed. This is because the reduction of iron ore occurs mainly inside the upper furnace away from the raceways.
Comparison of Different Models
Figure 9 shows four common geometrical configurations considered in the CFD modeling of the BF process. They correspond to the so-called full 3D BF model, sector model, axisymmetric model and slot model, respectively. These models may describe burden distribution and raceways differently and are assessed in detail.
To be comparable, unless otherwise noted, the inputted coke rate (kg/tHM), PCI rate (kg/tHM) and blast rate (Nm3/min), as well as blast, coke and ore components, remain unchanged. The blast volume and burden mass are determined according to the ratio of the computational BF volume to the effective BF volume. The full 3D model considers the whole 360 deg BF with 40 tuyeres, each of which occupies 9 deg. The sector model computes a 36 deg BF section with four tuyeres. The geometry simulated in the 2D axisymmetric model is the same as that in the sector model. However, the raceway of the former is generated by sweeping a 2D raceway along the circumference (Figure 9(c)). A similar raceway is also considered in the 2D slot model (Figure 9(d)). Note that from the viewpoint of geometric similarity, the slot model is different from others. That is, at any height, the horizontal cross-sectional area is proportional to the radius in the slot model, but to the radius squared in other models. Because of this difference, the ore-to-coke radial profile in the slot model needs to be scaled up by 1.068 from that in the other models (see Figure 10) so that the same coke rate can be maintained in four models.
Figure 11 compares the layered burden structures predicted by different models. Here, only the solid flow is simulated. For comparison, the results on the vertical plane crossing the tuyere centerline are considered. It is shown that full 3D, sector and axisymmetric models predict nearly the same layered burden structures. Their layer thickness decreases when burden materials move downward. However, the slot model gives thinner burden layers inside the throat but thicker ones inside the bosh. This difference is caused by the geometric simplification in the slot model.
Table VI compares the global performance indicators predicted by four models at the same coke rate, including the productivity (P), top gas utilization factor (TGUF), top gas temperature (TGT), coke rate (CR) and liquid temperature (LT). To facilitate the following discussion, the slot model considering the same coke rate as by the full 3D model is referred to as slot-CR. Performance indicators are the same for sector and full 3D models. This is because these two models treat burden distributions and raceways in the same way. Also, in this work, the inputs at the BF top and bottom are uniform in the circumferential direction. Compared with the full 3D model, the axisymmetric model over-predicts the productivity by 0.5 pct and liquid temperature by 0.2 pct, but gives nearly the same top gas temperature and utilization factor. Considering that the scale of BF production is large, this difference in productivity is significant. Notably, slot-CR over-predicts the top gas temperature and under-predicts the top gas utilization factor and liquid temperature. Based on the slot model, the coke rate is also adjusted via a series of simulations to achieve the same hot metal temperature as in the full 3D model (referred to as slot-LT). Notably, compared with the full 3D model, slot-LT over-predicts the coke rate by 13 kg/tHM to achieve the same hot metal temperature.
To gain insights into the differences among different models, the in-furnace states are examined in detail. Figure 12 compares the shape and location of CZ. It reveals that the CZs predicted by the full 3D, sector and axisymmetric models are nearly the same. Conversely, slot-CR gives a much lower CZ, where the distance for heating the liquid is hence shortened. This accounts for its under-predicted liquid temperature. To achieve a higher liquid temperature, the coke rate needs to be increased, as done in the case of slot-LT. The CZ from slot-LT apparently arises near the central region, though its root position still nearly touches the raceway.
As discussed previously, the center-developed flow is established by loading more coke into the central area. This operation is substantially intensified by the slot model (including Slot-CR and Slot-LT) because of its larger cross-sectional areas in the central area compared with those of other models if the same volume is considered. To confirm this, Figure 13 compares the radial profiles of the gas flow rate with respect to different models at different heights. Here, results at each height are normalized by the total mass rate. This treatment helps reflect how the total gas flow rate is distributed in the radial direction when gas passes through a given cross-section. As expected, more gas is observed to pass through the central area in the slot model. At the tuyere level, the difference in gas flow distribution is located mainly around the raceways. This may be caused by the different treatments on both the raceway and burden distribution.
Thermochemical behaviors are also examined with respect to different models, and the results are given in Figure 14. Owing to the center-developed flow, all models predict higher solid temperature in the central area than in the peripheral area, which becomes opposite at the tuyere level because of the presence of the low-permeability deadman and high-temperature raceways (Figure 14(a)). This trend becomes more significant in the 2D slot model compared with other models because of its intensified center-developed flow.
Figure 14(b) shows that compared with other models, the slot model predicts lower indirect reaction rates in areas highlighted by red circles because of the lower temperature there. Thus, it has a more developed endothermal direct reaction, causing more heat consumption. Moreover, when the coke rate is fixed, more coke per tonnage hot metal is consumed by direct reaction and thus less combusted in the raceways, which results in less heat input. Also, the center-developed flow reduces the heat exchange between ore and gas, causing more energy loss from off-gas. The reduced heat input as well as increased off-gas heat loss and direct reduction together account for the low CZ in slot-CR. To compensate for such heat, a higher coke rate is necessary, as demonstrated in the case of slot-LT.
The trickling liquid flow is observed in all the models. Their patterns are somewhat different because of the differences in CZ shape and position. Since the raceway boundary is impermeable and there is no coke bed between raceways in axisymmetric and slot models, their liquid must flow along the raceway boundary into the hearth (Figure 14(c)). Thus, a more significant liquid mass flow rate is observed near raceways in slot and axisymmetric models than in full 3D and sector models. Layered CZ also affects the liquid temperature. However, this effect is mainly up to the CZ position and thus less significant than by liquid flow distribution (Figure 14(d)). As a result, full 3D, sector and axisymmetric models give similar liquid temperature distributions, which are different from those given by slot-CR and slot-LT.
Conclusion
The 3D characteristics of in-furnace states inside ironmaking blast furnaces are studied. The major results of this study can be summarized as follows:
-
(1)
A 3D parallel comprehensive CFD BF process model is developed. It explicitly considers layered burden CZ, gas and liquid re-distribution near raceways, trickling liquid flow in the CZ and dripping zone, and stockline variation. The prediction reliability of this model is tested by simulating an experimental BF and an industrial BF. The measured and predicted in-furnace states and global performance are in reasonable agreement. Also, the model can predict the trends established from routine operations related to the effects of coke and blast rates on BF performance well.
-
(2)
The in-furnace states of a 5000-m3 industrial BF with 40 tuyeres are revealed by the developed model. Results show that layered CZ and raceways significantly affect the spatial distributions of the flow and temperature of gas and liquid phases. Three-dimensional characteristics of in-furnace states are present in this BF operated even with uniform bottom and top inputs. This is observed for gas/liquid flow and temperature but mainly around the discrete raceways formed in the circumferential direction. Also, the liquid temperature distribution appears in the form of “needles” near the CZ because of the trickling liquid flow.
-
(3)
Four commonly used CFD process models are assessed against the modeling of 3D BF. Results show that the sector and full 3D models are nearly the same; the slot model over-predicts the coke rate up to 13 kg/tHM; the axisymmetric model over-predicts productivity by 0.5 pct and liquid temperature by 0.2 pct. The slot model does not maintain the geometric similarity like other models and thus predicts the intensified center-developed flow. Its lower thermochemical energy utilization efficiency, as reflected by the higher top gas temperature and less developed indirect chemical reactions, results in the significant over-prediction of the coke rate. The simplified treatment on raceways in axisymmetric and slot models significantly affects the radial distribution of flow and temperature for both gas and liquid phases. However, its influence on global performance indicators is much less significant than the geometric simplification in the slot model.
Abbreviations
- \( a_{{\text{FeO}}} \) :
-
Activity of molten wustite
- \( A_{\text{c}} \) :
-
Effective surface area of coke for reaction (m2)
- \( A_{\text{throat}} \) :
-
Cross-sectional area of BF throat (m2)
- \( c_{\text{p}} \) :
-
Specific heat \( \left( {\text{J kg}^{ - 1} \text{ K}^{ - 1} } \right) \)
- \( C_{{\text{SiO}_{2} }} \) :
-
Concentration of \( \text{SiO}_{2} \)\( \left( {\text{mol m}^{ - 3} } \right) \)
- \( {\text{CR}} \) :
-
Coke rate \( \left( {\text{kg tHM}^{ - 1} } \right) \)
- \( d \) :
-
Diameter of solid phase (m)
- \( D \) :
-
Diffusion coefficient \( \left( {\text{m}^{2} \,\text{s}^{ - 1} } \right) \)
- \( D_{{\text{s5}}} \) :
-
Intra-particle diffusion coefficient of \( {\text{H}}_{ 2} \)in reduced iron phase \( \left( {\text{m}^{2} \,\text{s}^{ - 1} } \right) \)
- \( E_{\text{f}} \) :
-
Effectiveness factors of solution loss reaction by \( {\text{CO}} \)
- \( E^{\prime}_{f} \) :
-
Effectiveness factors of water-gas reaction
- \( E_{{\text{gl}}} \) :
-
Volumetric enthalpy flux between gas and liquid, \( \left( {\text{W}\,\text{m}^{ - 3} } \right) \)
- \( f_{\text{o}} \) :
-
Fraction conversion of iron ore
- \( F \) :
-
Liquid mass flow rate \( \left( {\text{kg}\,\text{s}^{ - 1} } \right) \)
- \( {\mathbf{F}} \) :
-
Interaction force per unit volume \( \left( {\text{kg m}^{ - 2} \text{ s}^{ - 2} } \right) \)
- \( {\mathbf{g}} \) :
-
Gravitational acceleration \( \left( {\text{m s}^{ - 2} } \right) \)
- \( h_{ij} \) :
-
Heat transfer coefficient between i and j phase \( \left( {\text{W m}^{ - 2} \text{ K}^{ - 1} } \right) \)
- \( H \) :
-
Enthalpy \( \left( {\text{J}\,\text{kg}^{ - 1} } \right) \)
- \( \Delta H \) :
-
Reaction heat \( \left( {\text{J}\,\text{mol}^{ - 1} } \right) \)
- \( k \) :
-
Thermal conductivity \( \left( {\text{W m}^{ - 1} \text{ K}^{ - 1} } \right) \)
- \( k_{1} \) :
-
Rate constant of indirect reduction of iron ore by \( {\text{CO}} \)\( \left( {\text{m}\,\text{s}^{ - 1} } \right) \)
- \( k_{2} \) :
-
Rate constant of direction reduction of molten wustite \( \left( {\text{mol}\,\text{m}^{ - 2} \,\text{s}^{ - 1} } \right) \)
- \( k_{3} \) :
-
Rate constant of solution loss reaction by \( {\text{CO}} \)\( \left( {\text{m}^{3} \text{ kg}^{ - 1} \text{ s}^{ - 1} } \right) \)
- \( k_{5} \) :
-
Rate constant of indirect reduction of iron ore by \( {\text{H}}_{ 2} \)\( \left( {\text{m}\,\text{s}^{ - 1} } \right) \)
- \( k_{6} \) :
-
Rate constant of water gas reaction \( \left( {\text{m}^{3} \text{ kg}^{ - 1} \text{ s}^{ - 1} } \right) \)
- \( k_{8} \) :
-
Rate constant of silica reduction reaction in slag \( \left( {\text{m}\,\text{s}^{ - 1} } \right) \)
- \( k_{\text{f}} \) :
-
Gas-film mass transfer coefficient \( \left( {\text{m}\,\text{s}^{ - 1} } \right) \)
- \( k_{{\text{f5}}} \) :
-
Gas-film mass transfer coefficient in indirect reduction of iron ore by\( {\text{H}}_{ 2} \)\( \left( {\text{m}\,\text{s}^{ - 1} } \right) \)
- \( k_{{\text{f6}}} \) :
-
Gas-film mass transfer coefficient water–gas reaction \( \left( {\text{m}\,\text{s}^{ - 1} } \right) \)
- \( K_{1} \) :
-
Equilibrium constant of indirect reduction of iron ore by \( {\text{CO}} \)
- \( K_{5} \) :
-
Equilibrium constant of indirect reduction of iron ore by \( {\text{H}}_{ 2} \)
- \( LT \) :
-
Liquid temperature (K)
- \( m_{\text{batch}} \) :
-
Weight for one ore layer and one coke layer (kg)
- \( m_{\text{batch,ore}} \) :
-
Weight for one ore layer (kg)
- \( m_{\text{batch,coke}} \) :
-
Weight for one coke layer (kg)
- \( M_{i} \) :
-
Molar mass of \( i{\text{th}} \) species in gas phase
- \( M_{{\text{sm}}} \) :
-
Molar mass of \( {\text{FeO}} \) or flux in solid phase \( \left( {{\text{kg}}\,{\text{mol}}^{ - 1} } \right) \)
- \( N_{{\text{coke}}} \) :
-
Number of coke particles in unit volume of bed \( \left( {\text{m}^{ - 3} } \right) \)
- \( N_{{\text{ore}}} \) :
-
Number of iron oxide particles in unit volume of bed \( \left( {\text{m}^{ - 3} } \right) \)
- \( p \) :
-
Pressure \( \left( {\text{Pa}} \right) \)
- \( P \) :
-
Productivity \( \left( {\text{tHM m}^{ - 3} \text{ day}^{ - 1} } \right) \)
- \( P_{i,j} \) :
-
Proportion of liquid flowing from ith point to jth point
- \( {\text{Pe}} \) :
-
Peclet number
- \( { \Pr } \) :
-
Prandtl number
- \( R \) :
-
Gas constant \( \left(8.314 {\text{ J mol}}^{ - 1} \text{K}^{-1} \right) \)
- \( R_{k}^{*} \) :
-
Reaction rate for \( k\text{th} \)reaction \( \left( {{\text{mol}}\,{\text{m}}^{ - 3} \,{\text{s}}^{ - 1} } \right) \)
- \( {\text{Re}} \) :
-
Reynolds number
- \( S \) :
-
Source term
- \( {\text{Sc}} \) :
-
Schmidt number
- \( Sh_{r}^{ * } \) :
-
Normalized shrinkage ratio
- \( t_{\text{s}} \) :
-
Timeline (s)
- \( t_{batch} \) :
-
Total batch time for one ore layer and one coke layer (s)
- \( T \) :
-
Temperature (K)
- \( {\text{TGT}} \) :
-
Top gas temperature (K)
- \( {\text{TGUF}} \) :
-
Top gas utilization factor (pct)
- \( u_{\text{feed}} \) :
-
Burden feed velocity \( \left( {\text{m}\,\text{s}^{ - 1} } \right) \)
- \( {\mathbf{u}} \) :
-
Velocity \( \left( {\text{m}\,\text{s}^{ - 1} } \right) \)
- \( \overline{{\mathbf{U}}}_{\text{l}} \) :
-
Liquid main velocity \( \left( {\text{m}\,\text{s}^{ - 1} } \right) \)
- \( {\mathbf{U}}_{\text{S}} \) :
-
Stochastic velocity of liquid dispersion flow, \( \left( {\text{m}\,\text{s}^{ - 1} } \right) \)
- \( V_{\text{B}} \) :
-
Bed volume (m3)
- \( V_{{\text{cell}}} \) :
-
Volume of control volume (m3)
- \( y_{i} \) :
-
Mole fraction of ith species in gas phase
- \( y_{{\text{CO}}} ,y_{{\text{H}_{\text{2}} }} \) :
-
Molar fraction of \( {\text{CO}} \) and H2
- \( y_{{\text{CO}}}^{*} ,y_{{\text{H}_{\text{2}} }}^{*} \) :
-
Molar fraction of \( {\text{CO}} \) and \( {\text{H}}_{ 2} \) in equilibrium state for indirect reaction
- \( y_{{\text{CO}_{\text{2}} }} ,y_{{\text{H}_{\text{2}} \text{O}}} \) :
-
Molar fraction of \( {\text{CO}}_{ 2} \) and H2O(g)
- \( \alpha \) :
-
Specific surface area, \( \text{m}^{2} \,\text{m}^{ - 3} \); relaxation factor; liquid dispersion angle, rad
- \( \beta \) :
-
Mass increase coefficient of fluid phase associated with reactions, \( \left( {\text{kg mol}^{ - 1} } \right) \)
- \( \varGamma \) :
-
Diffusion coefficient
- \( \delta \) :
-
Distribution coefficient
- \( \varepsilon \) :
-
Volume fraction
- \( \eta \) :
-
Fractional acquisition of reaction heat
- \( {\mathbf{\rm I}} \) :
-
Identity tensor
- \( \mu \) :
-
Viscosity \( \left( {\text{kg}\,\text{m}^{ - 1} \,\text{s}^{ - 1} } \right) \)
- \( \xi_{\text{ore}} ,\xi_{\text{coke}} \) :
-
Local ore, coke volume fraction
- \( \rho \) :
-
Density \( \left( {\text{kg}\,\text{m}^{ - 3} } \right) \)
- \( \rho_{\text{bulk}} \) :
-
Bulk density of burden at BF throat, \( \left( {\text{kg}\,\text{m}^{ - 3} } \right) \)
- \( \varvec{\tau} \) :
-
Stress tensor (Pa)
- \( \varphi \) :
-
General variable
- \( \omega \) :
-
Mass fraction
- \( \text{e} \) :
-
Effective
- \( \text{g} \) :
-
Gas
- \( i \) :
-
Identifier (g, s or l)
- \( i\text{,}m \) :
-
mth species in i phase
- \( j \) :
-
Identifier (g, s or l)
- \( k \) :
-
kth reaction
- \( \text{l} \) :
-
Liquid
- \( \text{l,d} \) :
-
Dynamic liquid
- \( \text{sm} \) :
-
FeO or flux in solid phase
- \( \text{e} \) :
-
Effective
- \( \text{g} \) :
-
Gas
- \( \text{l} \) :
-
Liquid
- \( \text{s} \) :
-
Solid
- \( T \) :
-
Transpose
References
Y. Omori: Blast furnace phenomena and modelling. (Elsevier Science Pub. Co. Inc.,New York, NY 1987).
X. F. Dong, A. B. Yu, J. Yagi and P. Zulli, ISIJ Int., 2007, vol. 47, pp. 1553-70.
S. Watakabe, K. Miyagawa, S. Matsuzaki, T. Inada, Y. Tomita, K. Saito, M. Osame, P. Sikström, L. S. Ökvist and J.-O. Wikstrom, ISIJ Int, 2013, vol. 53, pp. 2065-71.
Z. Y. Li, S. B. Kuang, A. Y. Yu, J. J. Gao, Y. H. Qi, D. L. Yan, Y. T. Li and X. M. Mao, Metall and Materi Trans B, 2018, vol. 49, pp. 1995-2010.
J. Yagi, ISIJ Int., 1993, vol. 33, pp. 619-39.
S. Ueda, S. Natsui, H. Nogami, J. Yagi and T. Ariyama, ISIJ Int., 2010, vol. 50, pp. 914-23.
T. Ariyama, S. Natsui, T. Kon, S. Ueda, S. Kikuchi and H. Nogami, ISIJ Int., 2014, vol. 54, pp. 1457-71.
S. B. Kuang, Z. Y. Li and A. B. Yu, Steel Res. Int., 2018, vol. 89, p. 1700071.
T. Okosun, A. K. Silaen and C. Q. Zhou, Steel Res Int, 2019, vol. 90, p. 1900046.
H. Nogami and J. Yagi, ISIJ Int., 2004, vol. 44, pp. 1826-34.
H. Nogami, P. R. Austin, J.-i. Yagi and K. Yamaguchi, ISIJ Int, 2004, vol. 44, pp. 500-09.
J. Chen, T. Akiyama, H. Nogami, J. Yagi and H. Takahashi, ISIJ Int., 1993, vol. 33, pp. 664-71.
Z. Y. Zhou, A. B. Yu and P. Zulli, Prog. Comput. Fluid. Dy., 2004, vol. 4, pp. 39-45.
S. J. Zhang, A. B. Yu, P. Zulli, B. Wright and U. Tuzun, ISIJ Int., 1998, vol. 38, pp. 1311-19.
P. R. Austin, H. Nogami and J. Yagi, ISIJ Int., 1997, vol. 37, pp. 748-55.
H. Nogami, Y. Kashiwaya and D. Yamada, ISIJ Int., 2012, vol. 52, pp. 1523-27.
S. J. Chew, P. Zulli and A. B. Yu, ISIJ Int., 2001, vol. 41, pp. 1112-21.
G. X. Wang, S. J. Chew, A. B. Yu and P. Zulli, Metall. Mater. Trans. B, 1997, vol. 28, pp. 333-43.
G. X. Wang, J. D. Litster and A. B. Yu, ISIJ Int., 2000, vol. 40, pp. 627-36.
J. A. de Castro, H. Nogami and J. Yagi, ISIJ Int., 2000, vol. 40, pp. 637-46.
Y. Hashimoto, Y. Sawa, Y. Kitamura, T. Nishino and M. Kano, ISIJ Int, 2018, vol. 58, pp. 2210-18.
X. F. Dong, A. B. Yu, S. J. Chew and P. Zulli, Metall. Mater. Trans. B, 2010, vol. 41, pp. 330-49.
K. Yang, S. Choi, J. Chung and J. Yagi, ISIJ Int, 2010, vol. 50, pp. 972-80.
D. Fu, Y. Chen, Y. F. Zhao, J. D’Alessio, K. J. Ferron and C. Q. Zhou, Appl. Therm. Eng., 2014, vol. 66, pp. 298-308.
P. Zhou, H. L. Li, P. Y. Shi and C. Q. Zhou, Appl. Therm. Eng., 2016, vol. 95, pp. 296-302.
S. B. Kuang, Z. Y. Li, D. L. Yan, Y. H. Qi and A. B. Yu, Miner. Eng., 2014, vol. 63, pp. 45-56.
J. A. de Castro, A. J. da Silva, Y. Sasaki and J. Yagi, ISIJ Int., 2011, vol. 51, pp. 748-58.
X. F. Dong, S. J. Zhang, D. Pinson, A. B. Yu and P. Zulli, Powder Technol., 2004, vol. 149, pp. 10-22.
L. Shao and H. Saxén, ISIJ Int., 2013, vol. 53, pp. 988-94.
D. Rangarajan, T. Shiozawa, Y. S. Shen, J. S. Curtis and A. B. Yu, Ind. Eng. Chem. Res., 2014, vol. 53, pp. 4983-90.
M. Y. Gu, G. Chen, M. C. Zhang, D. Huang, P. Chaubal and C. Q. Zhou, Appl. Math. Model., 2010, vol. 34, pp. 3536-46.
J. A. de Castro, H. Nogami and J. Yagi, ISIJ Int., 2002, vol. 42, pp. 44-52.
K. Takatani, T. Inada and Y. Ujisawa, ISIJ Int., 1999, vol. 39, pp. 15-22.
P. R. Austin, H. Nogami and J. Yagi, ISIJ Int., 1997, vol. 37, pp. 458-67.
S. J. Chew, P. Zulli and A. B. Yu, ISIJ Int., 2001, vol. 41, pp. 1122-30.
Z. Y. Li, S. B. Kuang, D. L. Yan, Y. H. Qi and A. B. Yu, Metall. Mater. Trans. B, 2017, vol. 48, pp. 602-18.
Y. S. Shen, B. Y. Guo, S. Chew, P. Austin and A. B. Yu, Metall Mater Trans B, 2015, vol. 46, pp. 432-48.
H. Zhao, M. Zhu, P. Du, S. Taguchi and H. Wei, ISIJ Int., 2012, vol. 52, pp. 2177-85.
G. Zhao, S. Cheng, W. Xu and C. Li, ISIJ Int, 2015, vol. 55, pp. 2566-75.
J. Xu, S. Wu, M. Kou, L. Zhang and X. Yu, Appl Math Model, 2011, vol. 35, pp. 1439-55.
A. Polinov, A. Pavlov, O. Onorin, N. Spirin and I. Gurin, Metallurgist, 2018, 62, 418-24.
K. Nishioka, Y. Ujisawa and K. Takatani, Nippon Steel & Sumitomo Metal Technical Report No. 120 2018.
Z. Y. Li, S. B. Kuang, S. D. Liu, J. Q. Gan, A. B. Yu, Y. T. Li and X. M. Mao, Powder Technol., 2019, vol. 353, pp. 385-97.
S. Natsui, T. Kikuchi and R. O. Suzuki, Metall and Materi Trans B, 2014, vol. 45, pp. 2395-413.
T. Kon, S. Natsui, S. Ueda and H. Nogami, ISIJ Int., 2015, vol. 55, pp. 1284-90.
S. Natsui, T. Kikuchi, R. O. Suzuki, T. Kon, S. Ueda and H. Nogami, ISIJ Int., 2015, vol. 55, pp. 1259-66.
S. Natsui, K.-i. Ohno, S. Sukenaga, T. Kikuchi and R. O. Suzuki, ISIJ Int., 2018, vol. 58, pp. 282-91.
S. Natsui, A. Sawada, T. Kikuchi and R. O. Suzuki, ISIJ Int., 2018, vol. 58, pp. 1742-44.
S. Natsui, A. Sawada, K. Terui, Y. Kashihara, T. Kikuchi and R. O. Suzuki, ETSU TO HAGANE, 2018, vol. 104, pp. 347-57.
S. Natsui, A. Sawada, K. Terui, Y. Kashihara, T. Kikuchi and R. O. Suzuki, Chem Eng Sci, 2018, vol. 175, pp. 25-39.
J. Ferziger and M. Peric: Computational Methods for Fluid Dynamics. 3rd ed. (Springer, New York, 2002).
Y. S. Shen, B. Y. Guo, A. B. Yu, P. R. Austin and P. Zulli, Fuel, 2011, vol. 90, pp. 728-38.
S.-i. Nomura, T Iron Steel I Jpn, 1986, vol. 26, pp. 107-13.
G. Usera, A. Vernet and J. Ferré, Flow Turbul and Combust, 2008, vol. 81, pp. 471-95.
S. Natsui, S. Ueda, Z. Fan, N. Andersson, J. Kano, R. Inoue and T. Ariyama, ISIJ Int., 2010, vol. 50, pp. 207-14.
H. Takahashi, M. Tanno and J. Katayama, ISIJ Int., 1996, vol. 36, pp. 1354-59.
Z. Zhou, H. Zhu, A. Yu, B. Wright, D. Pinson and P. Zulli, ISIJ Int., 2005, vol. 45, pp. 1828-37.
S. J. Zhang, A. B. Yu, P. Zulli, B. Wright and P. Austin, Appl. Math. Model., 2002, vol. 26, pp. 141-54.
I. Muchi, Trans. ISIJ, 1967, vol. 7, pp. 223-37.
Acknowledgments
The authors are grateful to the Australian Research Council (ARC) and the Baosteel Australia Research and Development Centre (BAJC) for the financial support of this work; the National Computational Infrastructure (NCI), Sunway TaihuLight, for the use of their high-performance computational facilities; and CAFFA3D for making a useful code available for free use and adaptation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Manuscript submitted June 24, 2019.
Rights and permissions
About this article
Cite this article
Jiao, L., Kuang, S., Yu, A. et al. Three-Dimensional Modeling of an Ironmaking Blast Furnace with a Layered Cohesive Zone. Metall Mater Trans B 51, 258–275 (2020). https://doi.org/10.1007/s11663-019-01745-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-019-01745-3