Abstract
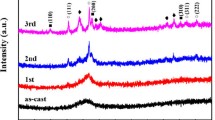
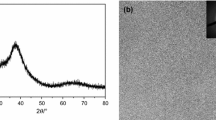
The kinetics of glass transition and crystallization of a novel Zr40Hf10Ti4Y1Al10Cu25Ni7Co2Fe1 bulk metallic glass (BMG) with high mixing entropy have been studied by differential scanning calorimetry (DSC) and X-ray diffraction (XRD). The continuous DSC curves show five stages of crystallization at lower heating rates (≤ 20 K/min). The activation energies of glass transition were determined by Moynihan and Kissinger methods, while the activation energies of crystallization were calculated utilizing Kissinger, Ozawa, and Boswell models. The crystalline phases corresponding to each crystallization step have been found out. The kinetic fragility of Zr40Hf10Ti4Y1Al10Cu25Ni7Co2Fe1 BMG has also been evaluated. Based on the isothermal DSC curves, the Avrami exponent, evaluated from the Johnson–Mehl–Avrami equation, has been analyzed in detail. The current study reveals that the crystallization behavior of Zr40Hf10Ti4Y1Al10Cu25Ni7Co2Fe1 BMG exhibits characteristics of both the high entropy BMGs and traditional BMGs with a single principal element, leading to its high glass-forming ability.











Similar content being viewed by others
References
W.H. Wang, C. Dong, and C.H. Shek: Mater. Sci. Eng. R, 2004, vol. 44, pp. 45-89.
C.A. Schuh, T.C. Hufnagel, and U. Ramamurty: Acta Mater., 2007, vol. 55, pp. 4067-109.
K.F. Shamlaye, K.J. Laws, and J.F. Loffler: Acta Mater., 2017, vol. 128, pp.188-96.
E. Perim, D. Lee, Y. Liu, C. Toher, P. Gong, Y. Li, W. Neal Simmons, O. Levy, JJ. Vlassak, J. Schroers, and S. Curtarolo: Nat. Commun., 2016, vol. 7, pp. 12315
C. Chattopadhyay, and B.S. Murty: Scripta Mater., 2016, vol. 116, pp. 7-10.
J.W. Yeh, S.K. Chen, S.J. Lin, J.Y. Gan, T.S. Chin, C.H. Tsau, and S.Y Chang: Adv. Eng. Mater., 2014, vol. 6, pp. 299-303.
Y. Zhang, T.T. Zuo, Z. Tang, M.C. Gao, K.A. Dahmen, P.K. Liaw, and Z.P. Lu: Prog. Mater. Sci., 2014, vol. 61, pp. 1-93.
S. Guo, C. Ng, J. Lu, and C.T. Liu: J. Appl. Phys., 2011, vol. 109, pp.103505.
Takeuchi A, Chen N, Wada T, Yokoyama Y, Kato H, Inoue A, Yeh JW (2011) Intermetallics, 19:1546-54.
X.Q. Gao, K. Zhao, H.B. Ke, D.W. Ding, W.H. Wang, and H.Y. Bai: J. Non-Cryst. Solids, 2011, vol. 357, pp.3557-60.
H.Y. Ding, and K.F. Yao: J. Non-Cryst. Solids, 2013, vol. 364, 9-12.
S.F. Zhao, Y. Shao, X. Liu, N. Chen, H.Y. Ding, and K.F. Yao: Mater. Des., 2015, vol. 87 pp. 625-31.
T. Qi, Y. Li, A. Takeuchi, G. Xie, H. Miao, and W. Zhang: Intermetallics, 2015, vol. 66, pp. 8-12.
C. Chen, S. Pang, T. Cheng, and T. Zhang: J Non-Cryst. Solids, 2015, vol. 410, pp. 39-42.
Y. Zhang, X. Yang, and P.K. Liaw: JOM, 2012, vol. 64, pp. 830-8.
W.H. Wang: JOM, 2014, vol. 66, pp. 2067-77.
M.H. Tsai, and J.W. Yeh: Mater. Res. Lett., 2014, vol. 2, pp. 107-23.
K.N. Lad, R.T. Savalia, A. Pratap, G.K. Dey, and S. Banerjee: Thermochim. Acta, 2008, vol. 473, pp. 74-80.
J. Cui, J.S. Li, J. Wang, H.C. Kou, J.C. Qiao, S. Gravier, and J.J. Blandin: J. Non-Cryst. Solids, 2014, vol. 404, pp. 7-12.
H.Y. Jung, M. Stoica, S. Yi, D.H. Kim, and J. Eckert: Metall. Mater. Trans. A, 2015, vol. 46, pp. 2415-21.
P. Gong, S. Zhao, X. Wang, and K. Yao: Appl. Phys. A, 2015, vol. 120, pp. 145-153.
A.A. Tsarkov, E.N. Zanaeva, A.Y. Churyumov, S.V. Ketov, and D.V. Louzguine-Luzgin: Mater. Charact., 2016, vol. 111, pp. 75-80.
Z.F. Yao, J.C. Qiao, C. Zhang, J.M. Pelletier, and Y. Yao: J. Non-Cryst. Solids, 2015, vol. 415, pp. 42-50.
P. Gong, K.F. Yao, and H.Y. Ding: Mater. Lett., 2015, vol. 156, pp.146-9.
P. Gong, S. Zhao, H. Ding, K. Yao, and X. Wang: J. Mater. Res., 2015, vol. 30, pp. 2772-82.
M. Lasocka: Mater. Sci., 1976, vol. 23, pp. 173-7.
S.X. Wang, S.G. Quan, and C. Dong: Thermochim. Acta, 2012, vol. 532, pp. 92-5.
C.T. Moynihan: J. Am. Ceram. Soc., 1993, vol. 76, pp.1081-7.
H.E. Kissinger: Anal. Chem. 1957, vol. 29, 1702-6.
T. Ozawa: J. Bull. Chem. Soc. Jpn., 1965, vol. 38, pp.1881-6.
P.G. Boswell: J. Therm. Anal. Calorim., 1980, vol. 18, pp. 353-8.
Y. Li, W. Zhang, C. Dong, J. Qiang, and A. Inoue: Int. J. Miner. Metall. Mater., 2013, vol. 20, pp. 445-9.
N. Hua, W. Chen, X. Liu, and F. Yue: J. Non-Cryst. Solids, 2014, vol. 388, pp. 10-6.
J. Tan, Y. Zhang, M. Stoica, U. Kuhn, N. Mattern, F.S. Pan, and J. Eckert: Intermetallics, 2011, vol. 19, pp. 567-571.
L. Liu, Z.F. Wu, and J. Zhang: J. Alloys Compd., 2002, vol. 339, pp. 90-5.
C. Peng, Z.H. Chen, X.Y. Zhao, A.L. Zhang, L.K. Zhang, and D. Chen: J. Non-Cryst. Solids, 2014, vol. 405, pp.7-11.
W.K. An, A.H. Cai, J.H. Li, Y. Luo, T.L. Li, X. Xiong, Y. Liu, and Y. Pan: J. Non-Cryst. Solids, 2009, vol. 355, pp.1703-6.
A.H. Cai, W.K. An, Y. Luo, T.L. Li, X.S. Li, X. Xiong, and Y. Liu: J. Alloys Compd., 2010, vol. 490, pp. 642-6.
S. Cheng, C. Wang, M. Ma, D. Shan, and B. Guo: Thermochim. Acta, 2014, vol. 587, pp.11-7.
C.A. Angell: Science, 1995, vol. 267, pp. 1924-35.
M. Zhu, J.J. Li, L.J. Yao, Z.Y. Jian, F.E. Chang, and G.C. Yang: Thermochim. Acta, 2013, vol. 565, pp. 132-6.
M.Q. Jiang, and L.H. Dai: Phys. Rev. B, 2007, vol. 76, pp. 054204.
Q. Wang, J.M. Pelletier, L. Xia, H. Xu, and Y.D. Dong: J. Alloys Compd., 2006, vol. 413 pp. 181-7.
D. Wang, H. Tan, and Y. Li: Acta Mater., 2005, vol. 53, pp. 2969-79.
D. Okai, Y. Shimizu, N. Hirano, T. Fukami, T. Yamasaki, and A. Inoue: J. Alloys Compd., 2010, vol. 504, pp. S247-50.
K.K. Song, P. Gargarella, S. Pauly, G.Z. Ma, U. Kuhn, and J. Eckert, J. Appl. Phys., 2012, vol. 112, pp. 063503.
M. Avrami: J. Chem. Phys., 1939, vol. 7, pp. 1103-12.
M. Avrami: J. Chem. Phys., 1940, vol. 8, pp. 212-24.
S. Ranganathan, and M. Von Heimendahl: J. Mater. Sci., 1981, vol. 16, pp. 2401-4.
A. Calka, and A.P. Radinski: J. Mater. Res., 1985, vol. 3, pp. 59-63.
S. Wei, B. Ding, T. Lei, and Z. Hu: Mater. Lett., 1998, vol. 37, pp. 263-7.
E.J. Mittemeijer: J. Mater. Sci., 2004, vol. 39, pp. 1621-34.
M. Yang, X.J. Liu, H.H. Ruan, Y. Wu, H. Wang, and Z.P. Lu: J. Appl. Phys., 2016, vol. 119, pp. 245112.
G. Adam, and J.H. Gibbs: J. Chem. Phys., 1965, vol. 43, pp. 139-46.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Grant No. 51601063), the National Science Foundation for Distinguished Young Scholars of China (Grant No. 51725504), the Basic Research Project of Shenzhen (Grant No. JCYJ20170307155718660), and the Tribology Science Fund of State Key Laboratory of Tribology (Grant No. SKLTKF17B03). The authors are also grateful to the State Key Laboratory of Materials Processing and Die & Mould Technology and the Analytical and Testing Center, Huazhong University of Science and Technology for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted October 23, 2017.
Rights and permissions
About this article
Cite this article
Gong, P., Wang, S., Li, F. et al. Kinetics of Glass Transition and Crystallization of a Zr40Hf10Ti4Y1Al10Cu25Ni7Co2Fe1 Bulk Metallic Glass with High Mixing Entropy. Metall Mater Trans A 49, 2918–2928 (2018). https://doi.org/10.1007/s11661-018-4612-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11661-018-4612-5