Abstract
Objective
To explore the protective effect and the underlying mechanism of Hu-Lu-Ba-Wan (葫芦巴丸, HLBW) on the testis of diabetic rats.
Methods
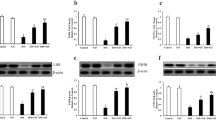
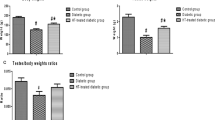
Twenty-four male Wistar rats (160–180 g) were randomly divided into 3 groups according to a random number table, including a control group (n=8), diabetic group (n=8), and HLBW group (n=8). Diabetic rat model was established by high-fat-diet administration and single intravenous injection of streptozotocin (26 mg/kg). Then HLBW granule was administrated for 12 weeks. Fasting blood glucose and insulin levels as well as serum total testosterone level and testicular testosterone content were examined. Oxidative stress markers in both serum and testis were tested. Meanwhile, testicular morphology was observed under hematoxylin and eosin (HE) and the ultrastructure of Leydig cell was observed by electron microscope. The superoxide anion level was detected by DHE, and TUNEL-positive cells of testis was evaluated by TUNEL assay. The gene and protein expression of protein kinase C (PKCα), phosphorylated PKCα (P-PKCα) and P47phox in testicular tissues were determined by quantitative RT-PCR analysis and Western bolt analysis.
Results
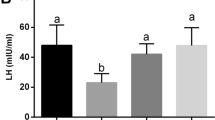
Compared with the diabetic group, HLBW treatment significantly reduced the fasting glucose levels and increased the levels of fasting insulin and testosterone in serum (P<0.01). HLBW administration also reduced the levels of reactive oxygen species (ROS) in plasma and alleviated the damage of oxidative stress in the testis of diabetic rats. Additionally, HLBW down-regulated the protein and mRNA levels of PKCα, P-PKCα and P47phox in testicular tissues.
Conclusion
HLBW may attenuate the oxidative stress in the testis of diabetic rats via PKCα /NAPDH oxidase signaling pathway.
Similar content being viewed by others
References
Maresch CC, Stute DC, Alves MG, Oliveira PF, de Kretser DM, Linn T. Diabetes-induced hyperglycemia impairs male reproductive function: a systematic review. Hum Reprod Update 2018;24:86–105.
Shi GJ, Zheng J, Wu J, Qiao HQ, Chang Q, Niu Y, et al. Beneficial effects of Lycium barbarum polysaccharide onspermatogenesis by improving antioxidant activity andinhibiting apoptosis in streptozotocin-induced diabetic malemice. Food Funct 2017;8:1215–1226
Kong ZL, Johnson A, Ko FC, He JL, Cheng SC. Effect of Cistanche Tubulosa Extracts on Male ReproductiveFunction in StreptozotocinNicotinamide-Induced DiabeticRats. Nutrients 2018;10:1–22.
Liu XX, Zhou HJ, Cai L, Zhang W, Ma JL, Tao XJ, et al. NADPH oxidase-dependent formation of reactive oxygen species contributes to transforming growth factor β1-induced epithelial-mesenchymal transition in rat peritoneal mesothelial cells, and the role of astragalus intervention. Chin J Integr Med 2014;20:667–674.
Rezende F, Moll F, Walter M, Helfinger V, Hahner F, Janetzko P, et al. The NADPH organizers NoxO1 and P47phox are both mediators of diabetes-induced vascular dysfunction in mice. Redox Biol 2018;15:12–21.
Xu SJ, Zhao YB, Jin CY, Yu L, Ding F, Fu GS, et al. PKC/NADPH oxidase are involved in the protective effect of pioglitazone in high homocysteine-induced paracrine dyfunction in endothelial progenitor cells. Am J Transl Res 2017;9:1037–1048.
Xie XR, Chen Y, Liu JC, Zhang WB, Zhang X, Zha LT, et al. High glucose induced endothelial cell reactive oxygen species via OGG1/ PKC/NADPH oxidase pathway. Life Sci 2020;256:117886.
Lu QB, Du Q, Wang HP, Tang ZH, Wang YB, Sun HJ. Salusin-β mediates tubular cell apoptosis in acute kidney injury: involvement of the PKC/ROS signaling pathway. Redox Biol 2020;30:101411.
Baquer NZ, Kumar P, Taha A, Kale RK, Cowsik SM, McLean P. Metabolic and molecular action of Trigonella foenum-graecum (fenugreek) and trace metals in experimental diabetic tissues. J Biosci 2011;36:383–396.
Liu L, Du XH, Zhang Z, Zhou JY. Trigonelline inhibits caspase 3 to protect β cells apoptosis in streptozotocin-induced type 1 diabetic mice. Eur J Pharmacol 2018;2999:30474–30476.
Wang YF, Liu YN, Xiong W, Yan DM, Zhu Y, Gao XM, et al. A UPLC-MS/MS method for in vivo and in vitro pharmacokinetic studies of psoralenoside, isopsoralenoside, psoralen and isopsoralen from Psoralea corylifolia extract. J Ethnopharmacol 2014;151:609–617.
Seo E, Lee EK, Lee CS, Chun KH, Lee MY, Jun HS. Psoralea corylifolia L. seed extract ameliorates streptozotocin-induced diabetes in mice by inhibition of oxidative stress. Oxid Med Cell Longev 2014;2014:1–9.
Johnsen SG. Testicular biopsy score count—a method for registration of spermatogenesis in human testes: normal values and results in 335 hypogonadal males. Hormones 1970;1:2–25.
Thakur M, Bhargava S, Praznik W, Loeppert R, Dixit VK. Effect of Chlorophytum borivilianum Santapau and fernandes on sexual dysfunction in hyperglycemic male rats. Chin J Integr Med 2009;15:448–453.
Kilarkaje N, Al-Bader MM. Diabetes-induced oxidative DNA damage alters p53-p21CIP1/Waf1 signaling in the rat testis. Reprod Sci 2015;22:102–112.
Liu W, Liang X, Shi Y. Effects of hirudin on high glucose-induced oxidative stress and inflammatory pathway in rat dorsal root ganglion neurons. Chin J Integr Med 2020;26:197–204.
Li L, Li YL, Zhou YF, Ge ZY, Wang LL, Li ZQ, et al. Jiangtang Xiaozhi Recipe prevents diabetic retinopathy in streptozotocin-induced diabetic rats. Chin J Integr Med 2017;23:425–432.
Deshpande PO, Mohan V, Thakurdesai PA. Preclinical toxicological evaluation of IDM01: the botanical composition of 4-hydroxyisoleucine- and trigonelline-based standardized fenugreek seed extract. Pharmacognosy Res 2017;9:138–150.
Long LL, Wang JN, Lu XF, Xu YX, Zheng SH, Luo CQ, et al. Protective effects of scutellarin on type - diabetes mellitus-induced testicular damages related to reactive oxygen species/Bcl-2/Bax and reactive oxygen species/microcirculation/staving pathway in diabetic rat. J Diabetes Res 2015;2015:1–11.
Liu SM, Li XZ, Zhang SN, Yang ZM, Wang KX, Lu F, et al. Acanthopanax senticosus protects structure and function of mesencephalic mitochondria in a mouse model of Parkinson’s disease. Chin J Integr Med 2018;24:835–843.
Yang W, Zhang GW, Jiang F, Zeng YF, Zou P, An HH, et al. BPDE and B[a]P induce mitochondrial compromise by ROS-mediated suppression of the SIRT1/TERT/PGC-1α pathway in spermatogenic cells both in vitro and in vivo. Toxicol Appl Pharmacol 2019;376:17–37.
Samie A, Sedaghat R, Baluchnejadmojarad T, Roghani M. Hesperetin, a citrus flavonoid, attenuates testicular damage in diabetic rats via inhibition of oxidative stress, inflammation, and apoptosis. Life Sci 2018;210:132–139.
Jiao N, Chen YP, Zhu YH, Wang W, Liu MX, Ding WL, et al. Protective effects of catalpol on diabetes mellitus-induced male reproductive damage via suppression of the AGEs/RAGE/Nox4 signaling pathway. Life Sci 2020;256:116736.
Zhou LS, Dong H, Huang Y, Xu LJ, Zou X, Wang KF, et al. Hu-Lu-Ba-Wan attenuates diabetic nephropathy in type 2 diabetic rats through PKC-a/NADPH oxidase signaling pathway. Evid Based Complement Alternat Med 2013;2013:1–10.
Author information
Authors and Affiliations
Contributions
Jiang SJ carried out the experiments, collected references and drafted the manuscript. Fang K and Li JB assisted Jiang SJ to finish the experiments. Chen G contributed to the statistical analysis. Lu FE defined the ideas of this study. Dong H designed the study and revised the manuscript. Xu LJ and Zou X provided the major support to study. All authors have read and approved the final manuscript.
Corresponding author
Additional information
Conflict of Interest
The authors declare that there is no conflict of interests regarding the publication of this paper.
Supported by the National Natural Science Foundation of China (No. 81703886, 81273683)
Rights and permissions
About this article
Cite this article
Jiang, Sj., Dong, H., Fang, K. et al. Protective Effects of Hu-Lu-Ba-Wan (葫芦巴丸) against Oxidative Stress in Testis of Diabetic Rats through PKCα/NAPDH Oxidase Signaling Pathway. Chin. J. Integr. Med. 27, 432–439 (2021). https://doi.org/10.1007/s11655-021-2863-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-021-2863-2