Abstract
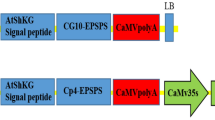
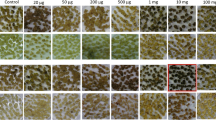
Soybean transformation is limited by the lack of multiple efficient selectable marker systems. Biolistic transformation of somatic proliferative embryogenic cultures, one of the commonly used soybean transformation methods, relies largely on hygromycin phosphotransferase II (hptII) selection. The purpose of the present study was to establish another efficient selectable marker system to facilitate multiple gene transformations of soybean. We tested neomycin phosphotransferase II (nptII) that has been used successfully in cotyledonary node transformation, but with limited success in transformation of embryogenic cultures. Transgenic events were obtained using nptII with improved G418 selection without generating escapes. G418 selection required longer recovery and selection periods, and resulted in a lower efficiency of initial transformants compared to hygromycin selection. Six independent fertile transgenic plants were recovered using nptII and G418, a frequency similar to that obtained with hygromycin selection. Soybean embryogenic cultures co-transformed with the hptII and nptII markers showed resistance to both hygromycin B and G418, while regeneration and plant fertility were not adversely affected. The nptII will be useful as a second selectable marker for multiple gene transformations in basic and applied soybean research.







Similar content being viewed by others
References
Aragão FJL, Sarokin L, Vianna GR, Rech EL (2000) Selection of transgenic meristematic cells utilizing a herbicidal molecule results in the recovery of fertile transgenic soybean [Glycine max (L.) Merril] plants at a high frequency. Theor Appl Genet 101:1–6
Bellini C, Guerche P, Spielmann A, Goujaud J, Lesaint C, Caboche M (1989) Genetic analysis of trasgenic tobacco plants obtained by liposome-mediated transformation: absence of evidence for the mutagenic effect of inserted sequences in sixty characterized transformants. J Hered 80(5):361–367. https://doi.org/10.1093/oxfordjournals.jhered.a110875
Carter T, Nelson R, Sneller C, Cui Z (2004) Genetic diversity in soybean. In: Boerma HR, Specht JE (eds) Soybean monograph. American Society of Agronomy, Madison, p 114
Chakraborty M, Sairam Reddy P, Laxmi Narasu M, Krishna G, Rana D (2016) Agrobacterium-mediated genetic transformation of commercially elite rice restorer line using nptII gene as a plant selection marker. Physiol Mol Biol Plants 22(1):51–60. https://doi.org/10.1007/s12298-015-0334-y
Dahmer M, Collins G, Hildebrand D (1991) Lipid content and composition of soybean somatic embryos. Crop Sci 31(3):741–746. https://doi.org/10.2135/cropsci1991.0011183X003100030041x
de Loubresse NG, Prokhorova I, Holtkamp W, Rodnina MV, Yusupova G, Yusupov M (2014) Structural basis for the inhibition of the eukaryotic ribosome. Nature 513(7519):517–522. https://doi.org/10.1038/nature13737
Droste A, Pasquali G, Bodanese-Zanettini MH (2002) Transgenic fertile plants of soybean [Glycine max (L.) Merrill] obtained from bombarded embryogenic tissue. Euphytica 127(3):367–376. https://doi.org/10.1023/A:1020370913140
Eustice DC, Wilhelm JM (1984) Mechanisms of action of aminoglycoside antibiotics in eucaryotic protein synthesis. Antimicrob Agents Chemother 26(1):53–60. https://doi.org/10.1128/AAC.26.1.53
Finer J, McMullen M (1991) Transformation of soybean via particle bombardment of embryogenic suspension culture tissue. In Vitro Cell Dev Biol Plant 27:175–182
Hadi MZ, McMullen MD, Finer JJ (1996) Transformation of 12 different plasmids into soybean via particle bombardment. Plant Cell Rep 15(7):500–505. https://doi.org/10.1007/BF00232982
Hinchee MA, Connor-Ward DV, Newell CA, McDonnell RE, Sato SJ, Gasser CS, Fischhoff DA, Re DB, Fraley RT, Horsch RB (1988) Production of transgenic soybean plants using Agrobacterium-mediated DNA transfer. Bio/Technology 6:915–922
Lee KY, Townsend J, Tepperman J, Black M, Chui CF, Mazur B, Dunsmuir P, Bedbrook J (1988) The molecular basis of sulfonylurea herbicide resistance in tobacco. EMBO J 7(5):1241–1248
Li R, Bancroft B, Hutcheon C, Zhao S, Zheng S, Blahut-Beatty L, De Rocher J, Simmonds D (2011) Multiple inserts of gene of interest and selectable marker gene are co-integrated and stably transmitted as a single genetic locus in transgenic soybean plants. In Vitro Cell Dev Biol Plant 47:274–281
Li Z, Xing A, Moon BP, McCardell RP, Mills K, Falco SC (2009) Site-specific integration of transgenes in soybean via recombinase-mediated DNA cassette exchange. Plant Physiol 151(3):1087–1095. https://doi.org/10.1104/pp.109.137612
Liu W, Torisky RS, McAllister KP, Avdiushko S, Hildebrand D, Collins GB (1996) Somatic embryo cycling: evaluation of a novel transformation and assay system for seed-specific gene expression in soybean. Plant Cell Tissue Organ Cult 47(1):33–42. https://doi.org/10.1007/BF02318963
Mazur BJ, Chui CF, Smith JK (1987) Isolation and characterization of plant genes coding for acetolactate synthase, the target enzyme for two classes of herbicides. Plant Physiol 85(4):1110–1117. https://doi.org/10.1104/pp.85.4.1110
McCabe DE, Swain WF, Martinell BJ, Christou P (1988) Stable transformation of soybean (Glycine max) by particle acceleration. Nat Biotech 6(8):923–926. https://doi.org/10.1038/nbt0888-923
Miki B, McHugh S (2004) Selectable marker genes in transgenic plants: applications, alternatives and biosafety. J Biotechnol 107(3):193–232. https://doi.org/10.1016/j.jbiotec.2003.10.011
Nitovska IO, Avilov ID, Morgun BV (2015) The positive effect of antibiotic paromomycin compared with kanamycin for selection of transgenic plants with NPTII gene on the example of Nicotiana tabacum. Фактори експериментальної еволюції організмів 17:270–273
Norelli JL, Aldwinckle HS (1993) The role of aminoglycoside antibiotics in the regeneration and selection of neomycin phosphotransferase-transgenic apple tissue. J Am Soc Hortic Sci 118:311–316
Olhoft PM, Flagel LE, Donovan CM, Somers DA (2003) Efficient soybean transformation using hygromycin B selection in the cotyledonary-node method. Planta 216(5):723–735. https://doi.org/10.1007/s00425-002-0922-2
Parrott W, Hoffman L, Hildebrand D, Williams E, Collins G (1989) Recovery of primary transformants of soybean. Plant Cell Rep 7(8):615–617. https://doi.org/10.1007/BF00272042
Parrott WA, All JN, Adang MJ, Bailey MA, Boerma HR, Stewart CN (1994) Recovery and evaluation of soybean plants transgenic for aBacillus thuringiensis var.Kurstaki insecticidal gene. In Vitro Cell Dev Biol Plant 30(3):144–149. https://doi.org/10.1007/BF02632204
Paz MM, Martinez JC, Kalvig AB, Fonger TM, Wang K (2006) Improved cotyledonary node method using an alternative explant derived from mature seed for efficient Agrobacterium-mediated soybean transformation. Plant Cell Rep 25(3):206–213. https://doi.org/10.1007/s00299-005-0048-7
Prakash NS, Prasad V, Chidambram TP, Cherian S, Jayaprakash TL, Dasgupta S, Wang Q, Mann MT, Spencer TM, Boddupalli RS (2008) Effect of promoter driving selectable marker on corn transformation. Transgenic Res 17(4):695–704. https://doi.org/10.1007/s11248-007-9149-0
Rech EL, Vianna GR, Aragao FJL (2008) High-efficiency transformation by biolistics of soybean, common bean and cotton transgenic plants. Nat Protocols 3(3):410–418. https://doi.org/10.1038/nprot.2008.9
Samoylov VM, Tucker DM, Thibaud-Nissen F, Parrott WA (1998) A liquid-medium-based protocol for rapid regeneration from embryogenic soybean cultures. Plant Cell Rep 18(1-2):49–54. https://doi.org/10.1007/s002990050530
Sathasivan K, Haughn GW, Murai N (1991) Molecular basis of imidazolinone herbicide resistance in Arabidopsis thaliana var Columbia. Plant Physiol 97(3):1044–1050. https://doi.org/10.1104/pp.97.3.1044
Sato S, Newell C, Kolacz K, Tredo L, Finer J, Hinchee M (1993) Stable transformation via particle bombardment in two different soybean regeneration systems. Plant Cell Rep 12(7-8):408–413. https://doi.org/10.1007/BF00234702
Shoemaker RC, Hammond EG (1988) Fatty acid composition of soybean (Glycine max (L.) Merr.) somatic embryos. In Vitro Cell Dev Biol Plant 24(8):829–832. https://doi.org/10.1007/BF02623655
Simmonds D (2003) Genetic transformation of soybean with biolistics. In: Jackson JF, Linskens HF (eds) Genetic transformation of plants. Springer Verlag, pp 159–174. https://doi.org/10.1007/978-3-662-07424-4_10
Simmonds DH, Donaldson PA (2000) Genotype screening for proliferative embryogenesis and biolistic transformation of short-season soybean genotypes. Plant Cell Rep 19(5):485–490. https://doi.org/10.1007/s002990050760
Stewart CN Jr, Adang MJ, All JN, Boerma HR, Cardineau G, Tucker D, Parrott WA (1996) Genetic transformation, recovery, and characterization of fertile soybean transgenic for a synthetic Bacillus thuringiensis cryIAc gene. Plant Physiol 112(1):121–129. https://doi.org/10.1104/pp.112.1.121
Taparia Y, Gallo M, Altpeter F (2012) Comparison of direct and indirect embryogenesis protocols, biolistic gene transfer and selection parameters for efficient genetic transformation of sugarcane. Plant Cell Tissue and Organ Culture (PCTOC) 111(2):131–141. https://doi.org/10.1007/s11240-012-0177-y
Timmermans MC, Maliga P, Vieira J, Messing J (1990) The pFF plasmids: cassettes utilising CaMV sequences for expression of foreign genes in plants. J Biotechnol 14(3-4):333–344. https://doi.org/10.1016/0168-1656(90)90117-T
Torbert KA, Rines HW, Somers DA (1995) Use of paromomycin as a selective agent for oat transformation. Plant Cell Rep 14(10):635–640. https://doi.org/10.1007/BF00232728
Trick HN, Finer JJ (1997) SAAT: sonication-assisted Agrobacterium-mediated transformation. Transgenic Res 6(5):329–336. https://doi.org/10.1023/A:1018470930944
Walter KL, Strachan SD, Ferry NM, Albert HH, Castle LA, Sebastian SA (2014) Molecular and phenotypic characterization of Als1 and Als2 mutations conferring tolerance to acetolactate synthase herbicides in soybean. Pest Manag Sci 70(12):1831–1839. https://doi.org/10.1002/ps.3725
Wang Q, Li P, Hanania U, Sahar N, Mawassi M, Gafny R, Sela I, Tanne E, Perl A (2005) Improvement of Agrobacterium-mediated transformation efficiency and transgenic plant regeneration of Vitis vinifera L. by optimizing selection regimes and utilizing cryopreserved cell suspensions. Plant Sci 168(2):565–571. https://doi.org/10.1016/j.plantsci.2004.09.033
Yamada T, Takagi K, Ishimoto M (2012) Recent advances in soybean transformation and their application to molecular breeding and genomic analysis. Breed Sci 61(5):480–494. https://doi.org/10.1270/jsbbs.61.480
Zhang Z, Xing A, Staswick P, Clemente TE (1999) The use of glufosinate as a selective agent in Agrobacterium-mediated transformation of soybean. Plant Cell Tissue Organ Cult 56(1):37–46. https://doi.org/10.1023/A:1006298622969
Acknowledgements
We thank Laureen Blahut-Beatty for excellent technical assistance. Funding from Agriculture and Agri-Food Canada is gratefully acknowledged. Ottawa Research and Development Centre Contribution number 17-012.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Editor: Ewen Mullins
Rights and permissions
About this article
Cite this article
Itaya, A., Zheng, S. & Simmonds, D. Establishment of neomycin phosphotransferase II (nptII) selection for transformation of soybean somatic embryogenic cultures. In Vitro Cell.Dev.Biol.-Plant 54, 184–194 (2018). https://doi.org/10.1007/s11627-017-9875-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-017-9875-9