Abstract
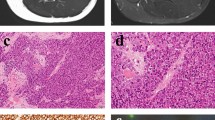
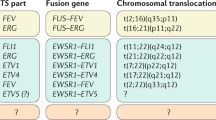
Ewing’s sarcoma is an aggressive mesenchymal tumor characterized by the presence of a unique EWSR1-FLI1 translocation. Ewing’s sarcoma primarily occurs in the bone and soft tissues. Cell lines enable researchers to investigate the molecular backgrounds of disease and the significance of genetic alterations in relevant cellular contexts. Here, we report the establishment and characterization of a novel Ewing’s sarcoma cell line following primary Ewing’s sarcoma tumor tissue culture. The established cell line was authenticated by DNA microsatellite short tandem repeat analysis, characterized by in vitro assays, and named NCC-ES1-C1. The NCC-ES1-C1 cell line grew well for 15 mo and was subcultured more than 50 times during this period. Characterization of the cells revealed that they were not adherent and showed floating features. In conclusion, we successfully established a novel Ewing’s sarcoma cell line, NCC-ES1-C1, from primary tumor tissue. The cell line has the characteristic EWSR1-FLI1 gene fusion and exhibits aggressive growth in vitro. Thus, the NCC-ES1-C1 cell line will be a useful tool for investigating the mechanisms of disease and the biological role of the EWSR1-FLI1 fusion gene.




Similar content being viewed by others
References
Adams J, Kauffman M (2004) Development of the proteasome inhibitor Velcade (Bortezomib). Cancer Investig 22(2):304–311
Alcindor T (2015) Response of refractory Ewing sarcoma to pazopanib. Acta Oncol 54(7):1063–1064. https://doi.org/10.3109/0284186x.2014.971938
Attia S, Okuno SH, Robinson SI, Webber NP, Indelicato DJ, Jones RL et al (2015) Clinical activity of pazopanib in metastatic extraosseous Ewing sarcoma. Rare Tumors 7(2):5992–5988. https://doi.org/10.4081/rt.2015.5992
Bailly RA, Bosselut R, Zucman J, Cormier F, Delattre O, Roussel M, Thomas G, Ghysdael J (1994) DNA-binding and transcriptional activation properties of the EWS-FLI-1 fusion protein resulting from the t(11;22) translocation in Ewing sarcoma. Mol Cell Biol 14(5):3230–3241
Bairoch, A (n.d.). The Cellosaurus: a cell line knowledge resource
Brohl AS, Solomon DA, Chang W, Wang J, Song Y, Sindiri S et al (2014) The genomic landscape of the Ewing sarcoma family of tumors reveals recurrent STAG2 mutation. PLoS Genet 10(7):e1004475. https://doi.org/10.1371/journal.pgen.1004475
Capes-Davis A, Dirks W, MacLeod R, Uphoff C (2014) Quality matters: cell lines and their use in research. GIT Lab J Eur 17:12–13
Carlomagno F, Anaganti S, Guida T, Salvatore G, Troncone G, Wilhelm SM, Santoro M (2006) BAY 43-9006 inhibition of oncogenic RET mutants. J Natl Cancer Inst 98(5):326–334. https://doi.org/10.1093/jnci/djj069
Crompton BD, Stewart C, Taylor-Weiner A, Alexe G, Kurek KC, Calicchio ML et al (2014) The genomic landscape of pediatric Ewing sarcoma. Cancer Discov 4(11):1326–1341. https://doi.org/10.1158/2159-8290.cd-13-1037
de Álava E, Lessnick SL, & Sorensen PHB (2013) Ewing sarcoma (C. D. M. Fletcher, J. A. Bridge, P. C. W. Hogendoorn, & F. Mertens Eds.)
Delattre O, Zucman J, Plougastel B, Desmaze C, Melot T, Peter M et al (1992) Gene fusion with an ETS DNA-binding domain caused by chromosome translocation in human tumours. Nature 359(6391):162–165. https://doi.org/10.1038/359162a0
Felgenhauer JL, Nieder ML, Krailo MD, Bernstein ML, Henry DW, Malkin D et al (2013) A pilot study of low-dose anti-angiogenic chemotherapy in combination with standard multiagent chemotherapy for patients with newly diagnosed metastatic Ewing sarcoma family of tumors: a Children’s Oncology Group (COG) phase II study NCT00061893. Pediatr Blood Cancer 60(3):409–414. https://doi.org/10.1002/pbc.24328
Gamberi G, Cocchi S, Benini S, Magagnoli G, Morandi L, Kreshak J et al (2011) Molecular diagnosis in Ewing family tumors: the Rizzoli experience--222 consecutive cases in four years. J Mol Diagn 13(3):313–324. https://doi.org/10.1016/j.jmoldx.2011.01.004
Grohar PJ, Helman LJ (2013) Prospects and challenges for the development of new therapies for Ewing sarcoma. Pharmacol Ther 137(2):216–224. https://doi.org/10.1016/j.pharmthera.2012.10.004
Jedlicka P (2010) Ewing sarcoma, an enigmatic malignancy of likely progenitor cell origin, driven by transcription factor oncogenic fusions. Int J Clin Exp Pathol 3(4):338–347
Juergens H, Daw NC, Geoerger B, Ferrari S, Villarroel M, Aerts I et al (2011) Preliminary efficacy of the anti-insulin-like growth factor type 1 receptor antibody figitumumab in patients with refractory Ewing sarcoma. J Clin Oncol 29(34):4534–4540. https://doi.org/10.1200/jco.2010.33.0670
Kasper B, Hohenberger P (2011) Pazopanib: a promising new agent in the treatment of soft tissue sarcomas. Future Oncol 7(12):1373–1383. https://doi.org/10.2217/fon.11.116
Kito F, Oyama R, Sakumoto M, Takahashi M, Shiozawa K, Qiao Z et al (2018) Establishment and characterization of novel patient-derived osteosarcoma xenograft and cell line. Vitro Cell Dev Biol Anim 54(7):528–536. https://doi.org/10.1007/s11626-018-0274-2
Kodama K, Doi O, Higashiyama M, Mori Y, Horai T, Tateishi R et al (1991) Establishment and characterization of a new Ewing’s sarcoma cell line. Cancer Genet Cytogenet 57(1):19–30
Lawlor ER, Scheel C, Irving J, Sorensen PH (2002) Anchorage-independent multi-cellular spheroids as an in vitro model of growth signaling in Ewing tumors. Oncogene 21(2):307–318. https://doi.org/10.1038/sj.onc.1205053
Lessnick SL, Ladanyi M (2012) Molecular pathogenesis of Ewing sarcoma: new therapeutic and transcriptional targets. Annu Rev Pathol 7:145–159. https://doi.org/10.1146/annurev-pathol-011110-130237
Moore JB t, Loeb DM, Hong KU, Sorensen PH, Triche TJ, Lee DW et al (2015) Epigenetic reprogramming and re-differentiation of a Ewing sarcoma cell line. Front Cell Dev Biol 3:15. https://doi.org/10.3389/fcell.2015.00015
Olmos D, Postel-Vinay S, Molife LR, Okuno SH, Schuetze SM, Paccagnella ML, Batzel GN, Yin D, Pritchard-Jones K, Judson I, Worden FP, Gualberto A, Scurr M, de Bono JS, Haluska P (2010) Safety, pharmacokinetics, and preliminary activity of the anti-IGF-1R antibody figitumumab (CP-751,871) in patients with sarcoma and Ewing's sarcoma: a phase 1 expansion cohort study. Lancet Oncol 11(2):129–135. https://doi.org/10.1016/s1470-2045(09)70354-7
Oyama R, Kito F, Sakumoto M, Shiozawa K, Toki S, Endo M et al (2018a) Establishment and proteomic characterization of a novel synovial sarcoma cell line, NCC-SS2-C1. Vitro Cell Dev Biol Anim 54(5):392–399. https://doi.org/10.1007/s11626-018-0237-7
Oyama R, Kito F, Sakumoto M, Shiozawa K, Toki S, Yoshida A et al (2018b) Establishment and proteomic characterization of a novel cell line, NCC-UPS2-C1, derived from a patient with undifferentiated pleomorphic sarcoma. Vitro Cell Dev Biol Anim 54(3):257–263. https://doi.org/10.1007/s11626-018-0229-7
Oyama R, Takahashi M, Kito F, Sakumoto M, Shiozawa K, Qiao Z et al (2018c) Establishment and characterization of patient-derived xenograft and its cell line of primary leiomyosarcoma of bone. Vitro Cell Dev Biol Anim 54(6):458–467. https://doi.org/10.1007/s11626-018-0258-2
Plumb JA, Finn PW, Williams RJ, Bandara MJ, Romero MR, Watkins CJ et al (2003) Pharmacodynamic response and inhibition of growth of human tumor xenografts by the novel histone deacetylase inhibitor PXD101. Mol Cancer Ther 2(8):721–728
Prieur A, Tirode F, Cohen P, Delattre O (2004) EWS/FLI-1 silencing and gene profiling of Ewing cells reveal downstream oncogenic pathways and a crucial role for repression of insulin-like growth factor binding protein 3. Mol Cell Biol 24(16):7275–7283. https://doi.org/10.1128/mcb.24.16.7275-7283.2004
Rodriguez-Galindo C, Liu T, Krasin MJ, Wu J, Billups CA, Daw NC et al (2007) Analysis of prognostic factors in Ewing sarcoma family of tumors: review of St. Jude Children’s Research Hospital studies. Cancer 110(2):375–384. https://doi.org/10.1002/cncr.22821
Sakumoto M, Oyama R, Takahashi M, Takai Y, Kito F, Shiozawa K et al (2018) Establishment and proteomic characterization of patient-derived clear cell sarcoma xenografts and cell lines. Vitro Cell Dev Biol Anim 54(2):163–176. https://doi.org/10.1007/s11626-017-0207-5
Santini MT, Rainaldi G (1999) Three-dimensional spheroid model in tumor biology. Pathobiology 67(3):148–157. https://doi.org/10.1159/000028065
Schwentner R, Papamarkou T, Kauer MO, Stathopoulos V, Yang F, Bilke S et al (2015) EWS-FLI1 employs an E2F switch to drive target gene expression. Nucleic Acids Res 43(5):2780–2789. https://doi.org/10.1093/nar/gkv123
Snijders AM, Nowak N, Segraves R, Blackwood S, Brown N, Conroy J et al (2001) Assembly of microarrays for genome-wide measurement of DNA copy number. Nat Genet 29(3):263–264. https://doi.org/10.1038/ng754
Staege MS, Hutter C, Neumann I, Foja S, Hattenhorst UE, Hansen G et al (2004) DNA microarrays reveal relationship of Ewing family tumors to both endothelial and fetal neural crest-derived cells and define novel targets. Cancer Res 64(22):8213–8221. https://doi.org/10.1158/0008-5472.can-03-4059
Tap WD, Demetri G, Barnette P, Desai J, Kavan P, Tozer R et al (2012) Phase II study of ganitumab, a fully human anti-type-1 insulin-like growth factor receptor antibody, in patients with metastatic Ewing family tumors or desmoplastic small round cell tumors. J Clin Oncol 30(15):1849–1856. https://doi.org/10.1200/jco.2011.37.2359
The International Cell Line Authentication Committee. Retrieved from http://standards.atcc.org/kwspub/home/the_international_cell_line_authentication_committee-iclac_/
Tirode F, Surdez D, Ma X, Parker M, Le Deley MC, Bahrami A et al (2014) Genomic landscape of Ewing sarcoma defines an aggressive subtype with co-association of STAG2 and TP53 mutations. Cancer Discov 4(11):1342–1353. https://doi.org/10.1158/2159-8290.cd-14-0622
Tomasz M (1995) Mitomycin C: small, fast and deadly (but very selective). Chem Biol 2(9):575–579
Toomey EC, Schiffman JD, Lessnick SL (2010) Recent advances in the molecular pathogenesis of Ewing's sarcoma. Oncogene 29(32):4504–4516. https://doi.org/10.1038/onc.2010.205
Ueda H, Nakajima H, Hori Y, Fujita T, Nishimura M, Goto T, Okuhara M (1994) FR901228, a novel antitumor bicyclic depsipeptide produced by Chromobacterium violaceum no. 968. I. Taxonomy, fermentation, isolation, physico-chemical and biological properties, and antitumor activity. J Antibiot (Tokyo) 47(3):301–310
Uren A, Toretsky JA (2005) Ewing’s sarcoma oncoprotein EWS-FLI1: the perfect target without a therapeutic agent. Future Oncol 1(4):521–528. https://doi.org/10.2217/14796694.1.4.521
Wilhelm SM, Carter C, Tang L, Wilkie D, McNabola A, Rong H et al (2004) BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res 64(19):7099–7109. https://doi.org/10.1158/0008-5472.can-04-1443
Womer RB, West DC, Krailo MD, Dickman PS, Pawel BR, Grier HE et al (2012) Randomized controlled trial of interval-compressed chemotherapy for the treatment of localized Ewing sarcoma: a report from the Children’s Oncology Group. J Clin Oncol 30(33):4148–4154. https://doi.org/10.1200/jco.2011.41.5703
Yamamoto Y, Nozawa M, Shimizu N, Minami T, Yoshimura K, Uemura H (2014) Pazopanib for recurrent extraosseous Ewing’s sarcoma of the retroperitoneum. Int J Urol 21(11):1183–1184. https://doi.org/10.1111/iju.12546
Acknowledgments
We appreciate Drs. M. Endo, Y. Minami, K. Shimizu, T. Mori, T. Uehara, M. Sugawara, Y. Araki, Ms. R. Nakano, and S. Zenitani, Division of Musculoskeletal Oncology, National Cancer Center Hospital, for sampling tumor-tissue specimens from surgically resected materials. We would also like to thank Editage (www.editage.jp) for English-language editing and their constructive comments on the manuscript.
Funding
This research was supported by the National Cancer Center Research and Development Fund (grant nos. 26-A-3, 26-A-9, and 29-A-2), and by the Fundamental Innovative Oncology Core in the National Cancer Center. We appreciate Dr. H. Sakamoto for SNP array experiments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Tetsuji Okamoto
Electronic supplementary material
Supplementary Table 1
(XLSX 22 kb)
Rights and permissions
About this article
Cite this article
Oyama, R., Kito, F., Qiao, Z. et al. Establishment of a novel patient-derived Ewing’s sarcoma cell line, NCC-ES1-C1. In Vitro Cell.Dev.Biol.-Animal 54, 770–778 (2018). https://doi.org/10.1007/s11626-018-0302-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-018-0302-2