Abstract
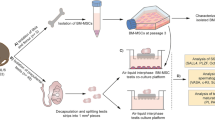
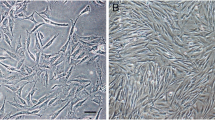
Human umbilical Wharton’s jelly-derived mesenchymal stem cells (HWJMSCs) are the best candidate to get plentiful stem cells and differentiate them to germ cells under appropriate conditions to treat infertility. We sought to determine under which conditions HWJMSCs could form male germ cells in vitro. So, HWJMSCs were differentiated to male germ cells under a mixture of bone morphogenetic protein-4 (BMP-4) and testicular and placental culture condition (TCC and PCC) medium followed by retinoic acid for 21 d. In the present study, the HWJMSCs were obtained from Wharton’s jelly of umbilical cords of male neonates delivered by cesarean section. At the third passage, mesenchymal stem cell markers and differentiation to osteocytes and adipocytes were investigated. Then, HWJMSCs were induced to differentiate into male germ cells in the presence of BMP-4, all-trans retinoic acid, PCC, and TCC for 21 d. The profile of c-Kit, DDX4, Piwil2, and Dazl gene expression was evaluated by qPCR and ICC. Data was analyzed by ANOVA test. After 3 wk of treatment with different reagents, the morphology of these spindle-like cells changed to shiny clusters and germ cell-specific markers in mRNA were upregulated in both TCC + retinoic acid (RA) and BMP-4 + RA. Induction of HWJMSCs with TCC in the presence of RA resulted in significant upregulation (P ≤ 0.05) of all germ cell-specific genes (c-Kit 2.6795 ± 0.75, DDX4 4.3188 ± 1.18, Piwil2 4.9962 ± 1.55, Dazl 6.1199 ± 0.78) compared to control and PCC + RA. Our results indicated that TCC and RA are involved in human germ cell development. Moreover, BMP signaling also induced differentiation. Our findings provide a novel effective approach for generation of germ cells in vitro and studying the interaction of germ cells with their niche. Our work represents an essential step toward gaining knowledge of the molecular properties of HWJMSCs in the field of cell therapy. We demonstrated that under a suitable situation, HWJMSCs have the ability to differentiate into germ cells and this provides an excellent pattern to study infertility cause and cure.






Similar content being viewed by others
References
Akmal KM, Dufour JM, Kim KH (1997) Retinoic acid receptor alpha gene expression in the rat testis: potential role during the prophase of meiosis and in the transition from round to elongating spermatids. Biol Reprod 56:549–556
Anzalone R, Iacono ML, Corrao S, Magno F, Loria T, Cappello F, Zummo G, Farina F, La Rocca G (2010) New emerging potentials for human Wharton’s jelly mesenchymal stem cells: immunological features and hepatocyte-like differentiative capacity. Stem Cells Dev 19:423–438
Castrillon DH, Quade BJ, Wang T, Quigley C, Crum CP (2000) The human VASA gene is specifically expressed in the germ cell lineage. Proc Natl Acad Sci 97:9585–9590
Chao KC, Chao KF, Fu YS, Liu SH (2008) Islet-like clusters derived from mesenchymal stem cells in Wharton’s Jelly of the human umbilical cord for transplantation to control type 1 diabetes. PLoS One 3:e1451
Chen MY, Lie PC, Li ZL, Wei X (2009) Endothelial differentiation of Wharton’s jelly-derived mesenchymal stem cells in comparison with bone marrow-derived mesenchymal stem cells. Exp Hematol 37:629–640
Clark AT, Bodnar MS, Fox M, Rodriquez RT, Abeyta MJ, Firpo MT, Pera RAR (2004) Spontaneous differentiation of germ cells from human embryonic stem cells in vitro. Human molecular genetics 13:727–739
Cowan G, Childs AJ, Anderson RA, Saunders PT (2010) Establishment of long-term monolayer cultures of somatic cells from human fetal testes and expansion of peritubular myoid cells in the presence of androgen. Reproduction 139:749–757
Drusenheimer N, Wulf G, Nolte J, Lee JH, Dev A, Dressel R, Gromoll J, Schmidtke J, Engel W, Nayernia K (2007) Putative human male germ cells from bone marrow stem cells. Soc Reprod Fertil Suppl 63:69–76
Eskild W, Ree AH, Levy FO, Jahnsen T, Hansson V (1991) Cellular localization of mRNAs for retinoic acid receptor-alpha, cellular retinol-binding protein, and cellular retinoic acid-binding protein in rat testis: evidence for germ cell-specific mRNAs. Biol Reprod 44:53–61
Fong CV, Subramanian A, Gauthaman K, Venugopal J, Biswas A, Ramakrishna S, Bongso A (2012) Human umbilical cord Wharton’s jelly stem cells undergo enhanced chondrogenic differentiation when grown on nanofibrous scaffolds and in a sequential two-stage culture medium environment. Stem Cell Rev Rep 8:195–209
Fu YS, Cheng YC, Lin MYA, Cheng H, Chu PM, Chou SC, Shin YH, Ko MH, Sung MS (2006) Conversion of human umbilical cord mesenchymal stem cells in Wharton’s jelly to dopaminergic neurons in vitro: potential therapeutic application for parkinsonism. Stem Cells 24:115–124
Gauthaman K, Venugopal JR, Yee FC, Biswas A, Ramakrishna S, Bongso A (2010) Osteogenic differentiation of human Wharton’s jelly stem cells on nanofibrous substrates in vitro. Tissue Eng A 17:71–81
Geijsen N, Horoschak M, Kim K, Gribnau J, Eggan K, Daley GQ (2004) Derivation of embryonic germ cells and male gametes from embryonic stem cells. Nature 427:148–154
Hollweck T, Hartmann I, Eblenkamp M, Wintermantel E, Reichart B, Überfuhr P, Eissner G (2011) Cardiac differentiation of human Wharton’s jelly stem cells—experimental comparison of protocols. Open Tissue Eng Regen Med J 4
Hu Y, Liang J, Cui H, Wang X, Rong H, Shao B, Cui H (2013) Wharton’s jelly mesenchymal stem cells differentiate into retinal progenitor cells. Neural Regen Res 8:1783
Huang P, Lin LM, Wu XY, Tang QL, Feng XY, Lin GY, Lin X, Wang HW, Huang TH, Ma L (2010) Differentiation of human umbilical cord Wharton’s jelly-derived mesenchymal stem cells into germ-like cells in vitro. J Cell Biochem 109:747–754
Hübner K, Fuhrmann G, Christenson LK, Kehler J, Reinbold R, De La Fuente R, Wood J, Strauss JF, Boiani M, Schöler HR (2003) Derivation of oocytes from mouse embryonic stem cells. Science 300:1251–1256
Kee K, Angeles VT, Flores M, Nguyen HN, Pera RAR (2009) Human DAZL, DAZ and BOULE genes modulate primordial germ-cell and haploid gamete formation. Nature 462:222–225
Kerkis A, Fonseca SA, Serafim RC, Lavagnolli TM, Abdelmassih S, Abdelmassih R, Kerkis I (2007) In vitro differentiation of male mouse embryonic stem cells into both presumptive sperm cells and oocytes. Cloning Stem Cells 9:535–548
Kerr CL, Cheng L (2010) The dazzle in germ cell differentiation. J Mol Cell Biol 2:26–29
Koubova J, Menke DB, Zhou Q, Capel B, Griswold MD, Page DC (2006) Retinoic acid regulates sex-specific timing of meiotic initiation in mice. Proc Natl Acad Sci USA 103:2474–2479
Lacham-Kaplan O, Chy H, Trounson A (2006) Testicular cell conditioned medium supports differentiation of embryonic stem cells into ovarian structures containing oocytes. Stem Cells 24:266–273
Lee J, Lako M, Armstrong L (2009) Derivation of human sperm from embryonic stem cells. http://www.ncbi.nlm.nih.gov/pubmed/19583494
Livera G, Rouiller-Fabre V, Pairault C, Levacher C, Habert R (2002) Regulation and perturbation of testicular functions by vitamin A. Reproduction 124:173–180
Mazaheri Z, Movahedin M, Rahbarizadeh F, Amanpour S (2012) Generation of in-vitro spermatogonial stem cells following genetic manipulation of primordial germ-like cells. Avicenna J Med Biotechnol 4:55
Mitchell KE, Weiss ML, Mitchell BM, Martin P, Davis D, Morales L, Helwig B, Beerenstrauch M, Abou-Easa K, Hildreth T (2003) Matrix cells from Wharton’s jelly form neurons and glia. Stem Cells 21:50–60
Nayernia K, Lee JH, Drusenheimer N, Nolte J, Wulf G, Dressel R, Gromoll J, Engel W (2006) Derivation of male germ cells from bone marrow stem cells. Lab Investig 86:654–663
Panula S, Medrano JV, Kee K, Bergstrom R, Nguyen HN, Byers B, Wilson KD, Wu JC, Simon C, Hovatta O (2011) Human germ cell differentiation from fetal- and adult-derived induced pluripotent stem cells. Hum Mol Genet 20:752–762
Peng J, Wang Y, Zhang L, Zhao B, Zhao Z, Chen J, Guo Q, Liu S, Sui X, Xu W (2011) Human umbilical cord Wharton’s jelly-derived mesenchymal stem cells differentiate into a Schwann-cell phenotype and promote neurite outgrowth< i>in vitro </i> Brain Res Bull 84:235–243
Pesce M, Wang X, Wolgemuth DJ, Scholer HR (1998) Differential expression of the Oct-4 transcription factor during mouse germ cell differentiation. Mech Dev 71:89–98
Ruggiu M, Saunders PT, Cooke HJ (2000) Dynamic subcellular distribution of the DAZL protein is confined to primate male germ cells. J Androl 21:470–477
Sandlow JI, Feng HL, Sandler A (1997) Localization and expression of the< i> c-Kit</i> receptor protein in human and rodent testis and sperm. Urology 49:494–500
Semenov O, Breymann C (2011) Mesenchymal stem cells derived from Wharton’s jelly and their potential for cardio-vascular tissue engineering. Open Tissue Eng Regen Med J 4
Toyooka Y, Tsunekawa N, Akasu R, Noce T (2003) Embryonic stem cells can form germ cells in vitro. Proc Natl Acad Sci 100:11457–11462
Venugopal P, Balasubramanian S, Majumdar AS, Ta M (2011) Isolation, characterization, and gene expression analysis of Wharton’s jelly-derived mesenchymal stem cells under xeno-free culture conditions. Stem Cells Cloning: Adv Appl 4:39
Wang HS, Hung SC, Peng ST, Huang CC, Wei HM, Guo YJ, Fu YS, Lai MC, Chen CC (2004) Mesenchymal stem cells in the Wharton’s jelly of the human umbilical cord. Stem Cells 22:1330–1337
Weiss M, Anderson C, Medicetty S, Sesharddy KB, Weiss RJ, Vanderwerff I, Troyer D, McIntosh KR (2008) Immune properties of human umbilical cord Wharton’s jelly-derived cells. Stem Cells 26:2865–2874
Xu C, Inokuma MS, Denham J, Golds K, Kundu P, Gold JD, Carpenter MK (2001) Feeder-free growth of undifferentiated human embryonic stem cells. Nat Biotechnol 19:971–974
Acknowledgments
We thank Mrs. Motlagh, Bahrami, Ramezani, Daneshvar, and Moghani for technical assistance. Gratitude is expressed to Dr. Akbari Asbagh for his critical comments. This study was supported by Tehran University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Tetsuji Okamoto
Rights and permissions
About this article
Cite this article
Amidi, F., Ataie Nejad, N., Agha Hoseini, M. et al. In vitro differentiation process of human Wharton’s jelly mesenchymal stem cells to male germ cells in the presence of gonadal and non-gonadal conditioned media with retinoic acid. In Vitro Cell.Dev.Biol.-Animal 51, 1093–1101 (2015). https://doi.org/10.1007/s11626-015-9929-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-015-9929-4