Abstract
Purpose
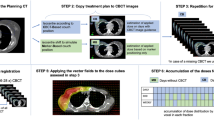
The aim of this study is to evaluate adaptive radiotherapy (ART) by use of replanning the tumor bed boost with repeated computed tomography (CT) simulation after whole breast irradiation (WBI) for breast cancer patients having clinically evident seroma.
Materials and methods
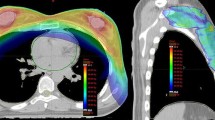
Forty-eight patients with clinically evident seroma at the time of planning CT simulation for WBI were included. Two RT treatment plannings were generated for each patient based on the initial CT simulation and tumor bed boost CT simulation to assess seroma and boost target volume (BTV) changes during WBI. Also, dosimetric impact of ART was analyzed by comparative evaluation of critical organ doses in both RT treatment plannings.
Results
Median time interval between the two CT simulations was 35 days. Statistically significant reduction was detected in seroma volume and BTV during the conventionally fractionated WBI course along with statistically significant reduction in critical organ doses with ART (p < 0.0001).
Conclusion
Our data suggest significant benefit of ART by use of replanning the tumor bed boost with repeated CT simulation after WBI for patients with clinically evident seroma.



Similar content being viewed by others
Abbreviations
- WBI:
-
Whole breast irradiation
- BCS:
-
Breast conserving surgery
- RT:
-
Radiotherapy
- CT:
-
Computed tomography
References
Coughlin SS, Ekwueme DU. Breast cancer as a global health concern. Cancer Epidemiol. 2009;33:315–8.
Bartelink H, Horiot JC, Poortmans PM, Struikmans H, Van den Bogaert W, Fourquet A, et al. Impact of a higher radiation dose on local control and survival in breast-conserving therapy of early breast cancer: 10-year results of the randomized boost versus no boost EORTC 22881-10882 trial. J Clin Oncol. 2007;25:3259–65.
Poortmans PM, Collette L, Horiot JC, Van den Bogaert WF, Fourquet A, Kuten A, et al. Impact of the boost dose of 10 Gy versus 26 Gy in patients with early stage breast cancer after a microscopically incomplete lumpectomy: 10-year results of the randomised EORTC boost trial. Radiother Oncol. 2009;90:80–5.
Poortmans PM, Collette L, Bartelink H, Struikmans H, Van den Bogaert WF, Fourquet A, et al. The addition of a boost dose on the primary tumour bed after lumpectomy in breast conserving treatment for breast cancer. A summary of the results of EORTC 22881-10882 “boost versus no boost” trial. Cancer Radiother. 2008;12:565–70.
Vrieling C, Collette L, Fourquet A, Hoogenraad WJ, Horiot JC, Jager JJ, et al. The influence of the boost in breast-conserving therapy on cosmetic outcome in the EORTC “boost versus no boost” trial. EORTC Radiotherapy and Breast Cancer Cooperative Groups. European Organization for Research and Treatment of Cancer. Int J Radiat Oncol Biol Phys. 1999;45:677–85.
Al-Ghazal SK, Fallowfield L, Blamey RW. Does cosmetic outcome from treatment of primary breast cancer influence psychosocial morbidity? Eur J Surg Oncol. 1999;25:571–3.
Major T, Gutiérrez C, Guix B, van Limbergen E, Strnad V, Polgár C, Breast Cancer Working Group of GEC-ESTRO. Recommendations from GEC ESTRO Breast Cancer Working Group (II): target definition and target delineation for accelerated or boost partial breast irradiation using multicatheter interstitial brachytherapy after breast conserving open cavity surgery. Radiother Oncol. 2016;118:199–204.
Goldberg H, Prosnitz RG, Olson JA, Marks LB. Definition of postlumpectomy tumor bed for radiotherapy boost field planning: CT versus surgical clips. Int J Radiat Oncol Biol Phys. 2005;63:209–13.
Benda RK, Yasuda G, Sethi A, Gabram SG, Hinerman RW, Mendenhall NP. Breast boost: are we missing the target? Cancer. 2003;97:905–9.
Perera F, Chisela F, Engel J, Venkatesan V. Method of localization and implantation of the lumpectomy site for high dose rate brachytherapy after conservative surgery for T1 and T2 breast cancer. Int J Radiat Oncol Biol Phys. 1995;31:959–65.
Srivastava V, Basu S, Shukla VK. Seroma formation after breast cancer surgery: what we have learned in the last two decades. J Breast Cancer. 2012;15:373–80.
Agrawal A, Ayantunde AA, Cheung KL. Concepts of seroma formation and prevention in breast cancer surgery. ANZ J Surg. 2006;76:1088–95.
Gonzalez EA, Saltzstein EC, Riedner CS, Nelson BK. Seroma formation following breast cancer surgery. Breast J. 2003;9:385–8.
Woodworth PA, McBoyle MF, Helmer SD, Beamer RL. Seroma formation after breast cancer surgery: incidence and predicting factors. Am Surg. 2000;66:444–50 (discussion 450–1).
Pierce LJ, Moughan J, White J, Winchester DP, Owen J, Wilson JF. 1998–1999 patterns of care study process survey of national practice patterns using breast-conserving surgery and radiotherapy in the management of stage I-II breast cancer. Int J Radiat Oncol Biol Phys. 2005;62:183–92.
Yang Z, Chen J, Xie J, Zhu C, Pan Z, Yu X, et al. Simultaneous integrated boost in breast conserving radiotherapy: is replanning necessary following tumor bed change? Technol Cancer Res Treat. 2013;12:115–22.
Cho H, Kim C. Volumetric changes in the lumpectomy cavity during whole breast irradiation after breast conserving surgery. Radiat Oncol J. 2011;29:277–82.
Hurkmans C, Admiraal M, van der Sangen M, Dijkmans I. Significance of breast boost volume changes during radiotherapy in relation to current clinical interobserver variations. Radiother Oncol. 2009;90:60–5.
Flannery TW, Nichols EM, Cheston SB, Marter KJ, Naqvi SA, Markham KM, et al. Repeat computed tomography simulation to assess lumpectomy cavity volume during whole-breast irradiation. Int J Radiat Oncol Biol Phys. 2009;75:751–6.
Sharma R, Spierer M, Mutyala S, Thawani N, Cohen HW, Hong L, et al. Change in seroma volume during whole-breast radiation therapy. Int J Radiat Oncol Biol Phys. 2009;75:89–93.
Tersteeg RJ, Roesink JM, Albregts M, Wárlám-Rodenhuis CC, van Asselen B. Changes in excision cavity volume: prediction of the reduction in absolute volume during breast irradiation. Int J Radiat Oncol Biol Phys. 2009;74:1181–5.
Prendergast B, Indelicato DJ, Grobmyer SR, Saito AI, Lightsey JL, Snead FE, et al. The dynamic tumor bed: volumetric changes in the lumpectomy cavity during breast-conserving therapy. Int J Radiat Oncol Biol Phys. 2009;74:695–701.
Oh KS, Kong FM, Griffith KA, Yanke B, Pierce LJ. Planning the breast tumor bed boost: changes in the excision cavity volume and surgical scar location after breast-conserving surgery and whole-breast irradiation. Int J Radiat Oncol Biol Phys. 2006;66:680–6.
Alderliesten T, den Hollander S, Yang TJ, Elkhuizen PH, van Mourik AM, Hurkmans C, et al. Dosimetric impact of post-operative seroma reduction during radiotherapy after breast-conserving surgery. Radiother Oncol. 2011;100:265–70.
Jeon SH, Shin KH, Park SY, Kim JI, Park JM, Kim JH, et al. Seroma change during magnetic resonance imaging-guided partial breast irradiation and its clinical applications. Radiat Oncol. 2017;12:103.
Mukesh M, Harris E, Jena R, Evans P, Coles C. Relationship between irradiated breast volume and late normal tissue complications: a systematic review. Radiother Oncol. 2012;104:1–10.
Borger JH, Kemperman H, Smitt HS, Hart A, van Dongen J, Lebesque J, et al. Dose and volume effects on fibrosis after breast conservation therapy. Int J Radiat Oncol Biol Phys. 1994;30:1073–81.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Informed consent was obtained from all individual participants included in the study. The study was performed in compliance with the Declaration of Helsinki principles.
About this article
Cite this article
Sager, O., Dincoglan, F., Uysal, B. et al. Evaluation of adaptive radiotherapy (ART) by use of replanning the tumor bed boost with repeated computed tomography (CT) simulation after whole breast irradiation (WBI) for breast cancer patients having clinically evident seroma. Jpn J Radiol 36, 401–406 (2018). https://doi.org/10.1007/s11604-018-0735-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11604-018-0735-2