Abstract
Metal-organic framework (MOF) materials have a topological structure, which helps the reactants to fully contact the active sites. This structure can effectively increase the number of active sites participating in the reaction and enhance their catalytic properties. Therefore, in this work, single metal MOF (Co-MOF), single metal MOF with Cu through electrodeposition (Cu/Co-MOF), and double metal MOF (CuCo-MOF) in the case of the same ligand and metal ion were prepared by a hydrothermal method and electrodeposition, which were then applied in the electrocatalytic oxygen evolution reaction (OER). A group of electrochemical tests showed that as-prepared CuCo-MOF exhibited lowest Rct value, highest electron transfer capability, and the best catalytic performance among the three catalysts. When the current density was 10 mA cm−2, the overpotential of CuCo-MOF was 0.38 V, and the slope of Tafel curve was 118 mV dec−1. Moreover, the CuCo-MOF had larger TOF values and better electrocatalytic property as compared with the other two MOF materials. Therefore, the self-assembled acicular CuCo-MOF material prepared in this work has the advantages of low overpotential, small initial potential, and good stability.

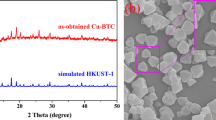
Scheme 1 Flowchart of CuCo-MOF fabrication and brief schematic of OER. CuCo-MOF were synthesized from Cu(Ac)2, Co(Ac)2, and terephthalic acid by hydrothermal method. Through optimal experiments, it was finally determined that the preparation of CuCo-MOF was set at 160 °C for 28 h. The as-prepared CuCo-MOF exhibited good OER performance.










Similar content being viewed by others
References
Xie Z, Wang Y (2018) Metal-organic framework-derived CoNi-embedded carbon nanocages as efficient electrocatalysts for oxygen evolution reaction. Ionics 24(6):1773–1780
Duan J, Chen S, Zhao C (2017) Ultrathin metal-organic framework array for efficient electrocatalytic water splitting. Nat Commun 8:15341–15342
Alia SM, Rasimick B, Ngo C, Neyerlin KC, Kocha SS, Pylypenko S, Xu H, Pivovara BS (2016) Activity and durability of iridium nanoparticles in the oxygen evolution reaction. J Electrochem Soc 163(11):F3105–F3112
Wang C, Liu X, Keser DN, Chen JP (2016) Applications of water stable metal-organic frameworks. Chem Soc Rev 45(18):5107–5108
Ghanem MA, Arunachalam P, Almayouf A, Wellerb MT (2016) Efficient bi-functional electrocatalysts of strontium iron oxy-halides for oxygen evolution and reduction reactions in alkaline media. J Electrochem Soc 163(6):H450–H458
Liu B, Li JZ, Gong XF, Zhang YL, Zhou QY, Cai JJ, Liu ZG, Sui XL, Wang ZB (2019) Facile synthesis of flower-like dual-metal (Co/Zn) MOF-derived 3D porous Co@Co-NPC as reversible oxygen electrocatalyst for rechargeable zinc-air batteries. Ionics:1–10
Wang Y, Zhang Z, Liu X, Ding F, Zou P, Wang X, Zhao Q, Rao H (2018) MOF-derived NiO/NiCo2O4 and NiO/NiCo2O4-rGO as highly efficient and stable electrocatalysts for oxygen evolution reaction. ACS Sustain Chem Eng 6(9):12511–12521
Jin YS, Wang HT, Li JJ, Yue X, Han YJ, Shen PK, Cui Y (2016) Porous MoO2 nanosheets as non-noble bifunctional electrocatalysts for overall water splitting. Adv Mater 28(19):3785–3790
Cao F, Gan M, Ma L, Li X, Yan F, Ye M, Zhai Y, Zhou Y (2017) Hierarchical sheet-like Ni–Co layered double hydroxide derived from a MOF template for high-performance supercapacitors. Synth Met 234:154–160
Zhi WS, Jakob K, Dickens CF, Ib C, Jens KN, Thomas FJ (2017) Combining theory and experiment in electrocatalysis: insights into materials design. Science 355(6321):4998
Li DJ, Li QH, Gu ZG, Zhang J (2012) Zero-dimensional, one-dimensional, two-dimensional and three-dimensional nanostructured materials for advanced electrochemical energy devices. Prog Mater Sci 57(4):724–803
Jitendra NT, Wang GL, Siraj S, Muhammad Y, Ahmad MH, Varun V, Kwang SK (2017) High-affinity-assisted nanoscale alloys as remarkable bifunctional catalyst for alcohol oxidation and oxygen reduction reactions. ACS Nano 11(8):7729–7735
Shen K, Chen XD, Chen JY, Li YW (2016) Development of MOF-derived carbon-based nanomaterials for efficient catalysis. ACS Catal 6:5887–5903
Li DJ, Li QH, Gua ZG, Jian ZG (2019) Surface-mounted MOF thin film with oriented nanosheet arrays for enhancing the oxygen evolution reaction. J Mater Chem A 7(31):18519–18528
Zheng J, Chen XL, Zhong X, Li SQ, Liu TZ, Zhuang GL, Li XN, Deng SW, Mei DH, Wang JG (2017) Hierarchical porous NC@CuCo nitride nanosheet networks: highly efficient bifunctional electrocatalyst for overall water splitting and selective electrooxidation of benzyl alcohol. Adv Funct Mater 27(46):1704169
Khurram SJ, Lutfan S, Lina GA, Chakr PJ, Hedhili MN, Bakr OM, Hussain I (2016) Atomically monodisperse nickel nanoclusters as highly active electrocatalysts for water oxidation. Nanoscale 8(18):9695–9703
Zheng J, Chen XL, Zhong X, Li SQ, Liu TZ, Zhuang GG, Li XN, Deng SG, Mei DH, Wang JG (2017) Hierarchical porous NC@ CuCo nitride nanosheet networks: highly efficient bifunctional electrocatalyst for overall water splitting and selective electrooxidation of benzyl alcohol. Adv Funct Mater 27(46):1704169
Zhao Y, Kamiya K, Hashimoto K, Nakanishi S (2015) Efficient bifunctional Fe/C/N electrocatalysts for oxygen reduction and evolution reaction. J Phys Chem C 119(5):2583–2588
Wang Z, Chen J, Bi R, Dou W, Wang K, Mao F, Wu H, Wang S (2020) Supercapacitor and oxygen evolution reaction performances based on morphology-dependent Co-MOFs. J Solid State Chem 283:121128
Abedini H, Shariati A, Khosravi-Nikou MR (2020) Adsorption of propane and propylene on M-MOF-74 (M=Cu, Co): equilibrium and kinetic study. Chem Eng Res Des 153:96–106
Wang J, Ni Y (2020) Ammonium molybdate-assisted shape-controlled synthesis of fluorescent Co (II)-based MOFs nanoflakes as highly-sensitive probes for selective detection of vanillin in milk powders. Mater Res Bull 123:110721
Lang XY, Han GF, Xiao BB, Gu L, Yang ZZ, Wen Z, Zhu YF, Zhao M, Li JC, Jiang Q (2015) Mesostructured intermetallic compounds of platinum and non-transition metals for enhanced electrocatalysis of oxygen reduction reaction. Adv Funct Mater 25(2):230–237
Yan WN, Yang ZR, Bian WY, Yang YY (2015) FeCo2O4/hollow graphene spheres hybrid with enhanced electrocatalytic activities for oxygen reduction and oxygen evolution reaction. Carbon 92(12):74–83
Zou Z, Wang T, Zhao X, Jiang W-J, Pan H, Gao D, Xu C (2019) Expediting in-situ electrochemical activation of two-dimensional metal–organic frameworks for enhanced OER intrinsic activity by Iron incorporation. ACS Catal 9(8):7356–7364
Du X, Su H, Zhang X (2019) Metal-organic framework-derived Cu-doped Co9S8 nanorod array with less low-valence co sites as highly efficient bifunctional electrodes for overall water splitting. ACS Sustain Chem Eng 7:16917–16926
Roy A, Ray A, Saha S, Ghosh M, Das T, Satpati B, Nandi M, Das S (2018) NiO-CNT composite for high performance supercapacitor electrode and oxygen evolution reaction. Electrochim Acta 283:327–337
Nakamoto (2006) Infrared and Raman spectra of inorganic and coordination compounds. In: Handbook of Vibrational Spectroscopy. https://doi.org/10.1002/9780470027325.s4104
Yan LT, Cao L, Dai PC, Gu X, Liu DD, Li LJ, Wang Y, Zhao XB (2017) Metal-organic frameworks derived nanotube of nickel–cobalt bimetal phosphides as highly efficient electrocatalysts for overall water splitting. Adv Funct Mater 27:1703455
Tullos GL, Powers JM, Jeskey SJ, Mathias LJ (1999) Thermal conversion of hydroxy-containing imides to benzoxazoles: polymer and model compound study. Macromolecules 32(11):3598–3612
He P, Yu XY, Lou XW (2017) Carbon-incorporated nickel-cobalt mixed metal phosphide nanoboxes with enhanced electrocatalytic activity for oxygen evolution. Angew Chem Int Ed 56(14):3897–3900
Yu X, Feng Y, Guan B, Lou XW, Pailk U (2016) Carbon coated nickel phosphides porous nanoplates for highly efficient oxygen evolution reaction. Energy Environ Sci 9(4):1246–1250
Xie SL, Li F, Xu SX, Li J, Zeng W (2019) Cobalt/iron bimetal-organic frameworks as efficient electrocatalysts for the oxygen evolution reaction. Chin J Catal 40:1205–1211
Shinagawa T, Garcia-Esparza AT, Takanabe K (2016) Insight on Tafel slopes from a microkinetic analysis of aqueous electrocatalysis for energy conversion. Sci Rep 5:13801
Taylor S, Fabbri E, Levecque P, Schmidt TJ, Conrad O (2016) The effect of platinum loading and surface morphology on oxygen reduction activity. Electrocatalysis 7(4):287–296
Sudha V, Annadurai K, Kumar S, Thangamuthu R (2019) CuCo2O4 nanobricks as electrode for enhanced electrochemical determination of hydroxylamine. Ionics 25(10):5023–5034
Wang J, Cui C, Lin R, Xu CH, Wang J, Li ZQ (2018) Hybrid cobalt-based electrocatalysts with adjustable compositions for electrochemical water splitting derived from Co2+-loaded MIL-53 (Fe) particles. Electrochim Acta 286:397–405
Swesi AT, Masud J, Nath M (2016) Enhancing electrocatalytic activity of bifunctional Ni3Se2 for overall water splitting through etching-induced surface nanostructuring. J Mater Res 31:2888–2896
Babar PT, Lokhande AC, Shima HJ, Gang MG, Pawar BS, Pawar SM, Kim JH (2019) SILAR deposited iron phosphate as a bifunctional electrocatalyst for efficient water splitting. J Colloid Interface Sci 534:350–356
Song ZS, Han XP, Deng YD, Zhao NQ, Hu WB, Zhong C (2017) Clarifying the controversial catalytic performance of Co (OH) 2 and Co3O4 for oxygen reduction/evolution reactions toward efficient Zn–air batteries. ACS Appl Mater Interfaces 9(27):22694–22703
Wang M, Wang P, Li C, Li H, Jin Y (2018) Boosting electrocatalytic oxygen evolution performance of ultrathin Co/Ni-MOF nanosheets via plasmon-induced hot carriers. ACS Appl Mater Interfaces 10(43):37095–37102
Peng L, Shah SSA, Wei Z (2018) Recent developments in metal phosphide and sulfide electrocatalysts for oxygen evolution reaction. Chin J Catal 39(10):1575–1593
Hany AE, Wei A, Olbrich LF, Putro GP, Gasteiger HA (2019) OER catalyst stability investigation using RDE technique: a stability measure or an artifact. J Electrochem Soc 166(8):F458–F464
Funding
This work was supported by the National Natural Science Foundation of China (No.21978026).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, J., Zhang, C., Duo, X. et al. Self-assembled acicular CuCo-MOF for enhancing oxygen evolution reaction. Ionics 26, 5123–5132 (2020). https://doi.org/10.1007/s11581-020-03651-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-020-03651-0