Abstract
Introduction
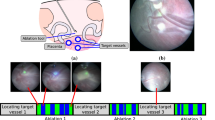
Twin-to-twin transfusion syndrome (TTTS) is a potentially lethal condition that affects pregnancies in which twins share a single placenta. The definitive treatment for TTTS is fetoscopic laser photocoagulation, a procedure in which placental blood vessels are selectively cauterized. Challenges in this procedure include difficulty in quickly identifying placental blood vessels due to the many artifacts in the endoscopic video that the surgeon uses for navigation. We propose using deep-learned segmentations of blood vessels to create masks that can be recombined with the original fetoscopic video frame in such a way that the location of placental blood vessels is discernable at a glance.
Methods
In a process approved by an institutional review board, intraoperative videos were acquired from ten fetoscopic laser photocoagulation surgeries performed at Yale New Haven Hospital. A total of 345 video frames were selected from these videos at regularly spaced time intervals. The video frames were segmented once by an expert human rater (a clinician) and once by a novice, but trained human rater (an undergraduate student). The segmentations were used to train a fully convolutional neural network of 25 layers.
Results
The neural network was able to produce segmentations with a high similarity to ground truth segmentations produced by an expert human rater (sensitivity = 92.15% ± 10.69%) and produced segmentations that were significantly more accurate than those produced by a novice human rater (sensitivity = 56.87% ± 21.64%; p < 0.01).
Conclusion
A convolutional neural network can be trained to segment placental blood vessels with near-human accuracy and can exceed the accuracy of novice human raters. Recombining these segmentations with the original fetoscopic video frames can produced enhanced frames in which blood vessels are easily detectable. This has significant implications for aiding fetoscopic surgeons—especially trainees who are not yet at an expert level.






Similar content being viewed by others
References
Cordero L, Franco A, Joy D, O’Shaughnessy W (2005) Monochorionic diamniotic infants without twin-to-twin transfusion syndrome. J Perinatol 25:753–758. https://doi.org/10.1038/sj.jp.7211405
Bahtiyar O, Emery P, Dashe S, Wilkins-Haug E, Johnson A, Paek W, Moon-Grady J, Skupski W, OʼBrien M, Harman R, Simpson L (2015) The North American Fetal Therapy Network consensus statement: prenatal surveillance of uncomplicated monochorionic gestations. Obstet Gynecol 125:118–123. https://doi.org/10.1097/AOG.0000000000000599
Faye-Petersen M, Crombleholme M (2008) Twin-to-twin transfusion syndrome. NeoReviews 9:370–379
Emery P, Bahtiyar O, Moise J (2015) The North American Fetal Therapy Network consensus statement: management of complicated monochorionic gestations. Obstet Gynecol 126:575–584. https://doi.org/10.1097/AOG.0000000000000994
Luks F (2009) Schematic illustration of endoscopic fetal surgery for twin-to-twin trans-fusion syndrome
Pratt R, Deprest J, Vercauteren T, Ourselin S, David L (2015) Computer-assisted surgical planning and intraoperative guidance in fetal surgery: a systematic review. Prenat Diagn 35:1159–1166. https://doi.org/10.1002/pd.4660
Miller R, Novotny J, Laidlaw H, Luks F, Merck D, Collins S (2016) Virtually visualizing vessels: a study of the annotation of placental vasculature from MRI in large-scale virtual reality for surgical planning. Brown University, Providence
Tella-Amo M, Daga P, Chadebecq F, Thompson S, Shakir I, Dwyer G, Wimalasundera R, Deprest J, Stoyanov D, Vercauteren T, Ourselin S (2016) A combined EM and visual tracking probabilistic model for robust mosaicking: application to fetoscopy. In: Proceedings of IEEE CVPR workshops, vol 31, pp 84–92. https://doi.org/10.1515/10.1109/cvprw.2016.72
Graves E, Harrison R, Padilla E (2017) Minimally invasive fetal surgery. Clin Perinatol 44:729–751. https://doi.org/10.1016/j.clp.2017.08.001
Tchirikov M, Oshovskyy V, Steetskamp J, Falkert A, Huber G, Entezami M (2011) Neonatal outcome using ultrathin fetoscope for laser coagulation in twin-to-twin-transfusion syndrome. J Perinat Med. https://doi.org/10.1515/jpm.2011.091
Olguner M, Akgür M, Özdemir T, Aktuğ T, Özer E (2000) Amniotic fluid exchange for the prevention of neural tissue damage in myelomeningocele: an alternative minimally invasive method to open in utero surgery. Pediatr Neurosurg 33:252–256. https://doi.org/10.1159/000055964
Yang L, Wang J, Ando T, Kubota A, Yamashita H, Sakuma I, Chiba T, Kobayashi E (2016) Towards scene adaptive image correspondence for placental vasculature mosaic in computer assisted fetoscopic procedures. Int J Med Robot Comput Assist Surg 12:375–386. https://doi.org/10.1002/rcs.1700
Gaisser F, Peeters S, Lenseigne B, Jonker P, Oepkes D (2018) Stable image registration for in vivo fetoscopic panorama reconstruction. J Imaging 4:24. https://doi.org/10.3390/jimaging4010024
Almoussa N, Dutra B, Lampe B, Getreuer P, Wittman T, Salafia C, Vese L (2011) Automated vasculature extraction from placenta images. In: Medical imaging 2011: image processing, vol 7962. International Society for Optics and Photonics
Park M, Yampolsky M, Shlakhter O, VanHorn S, Dygulska B, Kiryankova N, Salafia C (2013) Vessel enhancement with multiscale and curvilinear filter matching for placenta images. Placenta 34:A12
Chang JM, Huynh N, Vazquez M, Salafia C. (2013) Vessel enhancement with multiscale and curvilinear filter matching for placenta images. In: 2013 20th international conference on systems, signals and image processing (IWSSIP), pp 125–128
Perera Bel E (2017) Ultrasound segmentation for vascular network reconstruction in twin-to-twin transfusion syndrome. M.S. Thesis, Pompeu Fabra University, Barcelona, Spain. https://repositori.upf.edu/handle/10230/33180. Accessed 13 Nov 2018
Ronneberger O, Fischer P, Brox T (2015) U-Net: convolutional networks for biomedical image segmentation. MICCAI 18:234–241. https://doi.org/10.1007/978-3-319-24574-4_28
Panchapagesan S, Sun M, Khare A, Matsoukas S, Mandal A, Hoffmeister B, Vitaladevuni S (2016) Multi-task learning and weighted cross-entropy for DNN-based keyword spotting. In: Interspeech, pp. 760–764. https://doi.org/10.21437/Interspeech.2016-1485
Dice R (1945) Measures of the amount of ecologic association between species. Ecology 26:297–302. https://doi.org/10.2307/1932409
Frangi F, Niessen J, Vincken L, Viergever A (1998) Multiscale vessel enhancement filtering. MICCAI 1496:130–137
Srivastava R, Wong K, Duan L, Liu J, Wong TY (2015) Red lesion detection in retinal fundus images using Frangi-based filters. IEEE EMBC 37:5663–5666. https://doi.org/10.1109/EMBC.2015.7319677
Jiang Y, Zhuang W, Sinusas J, Staib H, Papademetris X (2011) Vessel connectivity using Murray’s hypothesis. MICCAI 14:528–536
Funding
This work was supported by the National Institutes of Health Grant Number T35DK104689 (NIDDK Medical Student Research Fellowship).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sadda, P., Imamoglu, M., Dombrowski, M. et al. Deep-learned placental vessel segmentation for intraoperative video enhancement in fetoscopic surgery. Int J CARS 14, 227–235 (2019). https://doi.org/10.1007/s11548-018-1886-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-018-1886-4