Abstract
Background
Ramucirumab—alone or combined with paclitaxel—represents one of the main options for patients failing first-line treatment for advanced gastric cancer.
Objective
The RAMoss study aimed to evaluate the safety and efficacy profile of ramucirumab in the “real-life setting”.
Patients and Methods
Patients from 25 Italian hospitals started therapy consisting of ramucirumab 8 mg/kg i.v. d1,15q28 with or without paclitaxel 80 mg/m2 i.v. d1,8,15q28. The primary endpoint was safety, and secondary endpoints were overall response rate (ORR), progression-free survival (PFS), and overall survival (OS).
Results
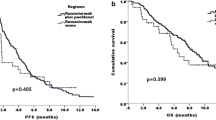
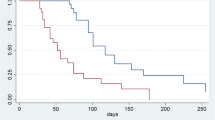
One hundred sixty-seven patients with disease progression on first-line therapy received ramucirumab as monotherapy (10%) or combined with paclitaxel (90%). Median treatment duration was 4 months (1–17 months). Global incidence of grade (G) 3–4 toxicity was 9.6%, and for neutropenia 5.4%; treatment was discontinued due to toxicity in 3% of patients. The most frequent adverse events (AE) were G1–2 fatigue (27.5%), G1–2 neuropathy (26.3%), and G1–2 neutropenia (14.9%). ORR was 20.2%. Stable disease was observed in 39.2% of patients, with a disease control rate of 59.4%. With a median follow-up of 11 months, median PFS was 4.3 months (95% confidence interval [CI] 4.1–4.7), whereas median OS was 8.0 months (95% CI: 7.09–8.9). In a multivariate analysis, ECOG performance status <1 or ≥1 (HR 1.13, 95% CI 1.0–1.27, p = 0.04) and the presence versus absence of peritoneal metastases (HR 1.57, 95% CI 1.63–2.39, p = 0.03) were independent poor prognostic factors.
Conclusions
These “real-life” efficacy data on ramucirumab treatment are in line with previous randomized trials. Ramucirumab is well tolerated in daily clinical practice.



Similar content being viewed by others
References
Ferro A, Peleteiro B, Malvezzi M, Bosetti C, Bertuccio P, Levi F, et al. Worldwide trends in gastric cancer mortality (1980-2011), with predictions to 2015, and incidence by subtype. Eur J Cancer. 2014;50(7):1330–44. https://doi.org/10.1016/j.ejca.2014.01.029.
Cunningham D, Allum WH, Stenning SP, Thompson JN, Van de Velde CJ, Nicolson M, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355(1):11–20. https://doi.org/10.1056/NEJMoa055531.
Macdonald JS, Smalley SR, Benedetti J, Hundahl SA, Estes NC, Stemmermann GN, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med. 2001;345(10):725–30. https://doi.org/10.1056/NEJMoa010187.
Ychou M, Boige V, Pignon JP, Conroy T, Bouche O, Lebreton G, et al. Perioperative chemotherapy compared with surgery alone for resectable gastroesophageal adenocarcinoma: an FNCLCC and FFCD multicenter phase III trial. J Clin Oncol. 2011;29(13):1715–21. https://doi.org/10.1200/JCO.2010.33.0597.
Van Cutsem E, Sagaert X, Topal B, Haustermans K, Prenen H. Gastric cancer. Lancet. 2016;388(10060):2654–64. https://doi.org/10.1016/S0140-6736(16)30354-3.
Wagner AD, Unverzagt S, Grothe W, Kleber G, Grothey A, Haerting J, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev. 2010;3:CD004064. https://doi.org/10.1002/14651858.CD004064.pub3.
Cho YH, Yoon SY, Kim SN. Irinotecan monotherapy versus irinotecan-based combination as second-line chemotherapy in advanced gastric cancer: a meta-analysis. Cancer Res Treat. 2017;49(1):255–62. https://doi.org/10.4143/crt.2015.452.
Ford H, Gounaris I. Docetaxel and its potential in the treatment of refractory esophagogastric adenocarcinoma. Therap Adv Gastroenterol. 2015;8(4):189–205. https://doi.org/10.1177/1756283X15585468.
Fuchs CS, Tomasek J, Yong CJ, Dumitru F, Passalacqua R, Goswami C, et al. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2014;383(9911):31–9. https://doi.org/10.1016/S0140-6736(13)61719-5.
Wilke H, Muro K, Van Cutsem E, Oh SC, Bodoky G, Shimada Y, et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol. 2014;15(11):1224–35. https://doi.org/10.1016/S1470-2045(14)70420-6.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–47. https://doi.org/10.1016/j.ejca.2008.10.026.
Shitara K, Muro K, Shimada Y, Hironaka S, Sugimoto N, Komatsu Y, et al. Subgroup analyses of the safety and efficacy of ramucirumab in Japanese and western patients in RAINBOW: a randomized clinical trial in second-line treatment of gastric cancer. Gastric Cancer. 2016;19(3):927–38. https://doi.org/10.1007/s10120-015-0559-z.
Janowitz T, Thuss-Patience P, Marshall A, Kang JH, Connell C, Cook N, et al. Chemotherapy vs supportive care alone for relapsed gastric, gastroesophageal junction, and oesophageal adenocarcinoma: a meta-analysis of patient-level data. Br J Cancer. 2016;114(4):381–7. https://doi.org/10.1038/bjc.2015.452.
Kang JH, Lee SI, Lim DH, Park KW, Oh SY, Kwon HC, et al. Salvage chemotherapy for pretreated gastric cancer: a randomized phase III trial comparing chemotherapy plus best supportive care with best supportive care alone. J Clin Oncol. 2012;30(13):1513–8. https://doi.org/10.1200/JCO.2011.39.4585.
Ford HE, Marshall A, Bridgewater JA, Janowitz T, Coxon FY, Wadsley J, et al. Docetaxel versus active symptom control for refractory oesophagogastric adenocarcinoma (COUGAR-02): an open-label, phase 3 randomised controlled trial. Lancet Oncol. 2014;15(1):78–86. https://doi.org/10.1016/S1470-2045(13)70549-7.
Thuss-Patience PC, Kretzschmar A, Bichev D, Deist T, Hinke A, Breithaupt K, et al. Survival advantage for irinotecan versus best supportive care as second-line chemotherapy in gastric cancer--a randomised phase III study of the Arbeitsgemeinschaft Internistische Onkologie (AIO). Eur J Cancer. 2011;47(15):2306–14. https://doi.org/10.1016/j.ejca.2011.06.002.
Hironaka S, Ueda S, Yasui H, Nishina T, Tsuda M, Tsumura T, et al. Randomized, open-label, phase III study comparing irinotecan with paclitaxel in patients with advanced gastric cancer without severe peritoneal metastasis after failure of prior combination chemotherapy using fluoropyrimidine plus platinum: WJOG 4007 trial. J Clin Oncol. 2013;31(35):4438–44. https://doi.org/10.1200/JCO.2012.48.5805.
Iacovelli R, Pietrantonio F, Farcomeni A, Maggi C, Palazzo A, Ricchini F, et al. Chemotherapy or targeted therapy as second-line treatment of advanced gastric cancer. A systematic review and meta-analysis of published studies. PLoS One. 2014;9(9):e108940. https://doi.org/10.1371/journal.pone.0108940.
Roviello G, Pacifico C, Polom K, Roviello F, Generali D. Different efficacy of ramucirumab in patients with metastatic gastric and gastroesophageal junction cancer according to ECOG performance status. Ann Oncol. 2017;28(3):665–6. https://doi.org/10.1093/annonc/mdw657.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No external funding was used in the preparation of this manuscript.
Conflict of Interest
Maria Di Bartolomeo, Monica Niger, Giuseppe Tirino, Angelica Petrillo, Rosa Berenato, Maria Maddalena Laterza, Filippo Pietrantonio, Federica Morano, Maria Antista, Sara Lonardi, Lorenzo Fornaro, Stefano Tamberi, Elisa Giommoni, Alberto Zaniboni, Lorenza Rimassa, Gianluca Tomasello, Teodoro Sava, Massimiliano Spada, Tiziana Latiano, Alessandro Bittoni, Alessandro Bertolini, Ilaria Proserpio, Katia Bruna Bencardino, Francesco Graziano, Giordano Beretta, Salvatore Galdy, Jole Ventriglia, Simone Scagnoli, Andrea Spallanzani, Raffaella Longarini, and Ferdinando De Vita declare that they have no conflict of interest that might be relevant to the content of this manuscript.
Ethical Approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
Rights and permissions
About this article
Cite this article
Di Bartolomeo, M., Niger, M., Tirino, G. et al. Ramucirumab as Second-Line Therapy in Metastatic Gastric Cancer: Real-World Data from the RAMoss Study. Targ Oncol 13, 227–234 (2018). https://doi.org/10.1007/s11523-018-0562-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-018-0562-5