Abstract
One of the commonly used techniques for drug delivery is to temporarily increase the permeability of tissue barriers. Acoustic energies such as ultrasound and shock waves are known to modulate tissue permeability. Recently, it was found that shock waves modulate the blood-brain barrier in the rat brain without injection of contrast agents such as microbubbles. This finding implies that modulation of other tissue barriers by shock wave exposure without contrast agents may be possible. To examine whether the modulation is also possible with other tissue barriers, we here investigated whether shock waves would modulate an in vitro tissue barrier model consisting of epithelial cells cultured on culture inserts. The permeability of the epithelium sheets evaluated by trans-epithelial electrical resistance (TEER) was increased following shock waves at a peak pressure of 11 MPa. The increased permeability recovered within 2 h. This enhancement was realized with one-shot low-energy shock waves having an acoustic energy of 0.013 mJ/mm2. Monitoring the peak pressures in every exposure revealed that the minimum peak pressure required for the enhancement is 2.9 MPa. These results indicate that shock wave exposure has the potential to temporarily increase the permeability of epithelium barriers to enhance drug delivery without contrast agents.

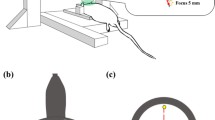
Enhancements of epithelial barrier permeability were evaluated by trans-epithelial electrical resistance (TEER) before and after shock wave exposures.



Similar content being viewed by others
References
Tscheik C, Blasig IE, Winkler L (2013) Trends in drug delivery through tissue barriers containing tight junctions. Tissue barriers 1:e24565. https://doi.org/10.4161/tisb.24565
Salama NN, Eddington ND, Fasano A (2006) Tight junction modulation and its relationship to drug delivery. Adv Drug Deliv Rev 58:15–28
Rosenthal R, Heydt MS, Amasheh M, Stein C, Fromm M, Amasheh S (2012) Analysis of absorption enhancers in epithelial cell models. Ann N Y Acad Sci 1258:86–92. https://doi.org/10.1111/j.1749-6632.2012.06562.x
Deli MA (2009) Potential use of tight junction modulators to reversibly open membranous barriers and improve drug delivery. Biochim Biophys Acta 1788:892–910. https://doi.org/10.1016/j.bbamem.2008.09.016
Izadifar Z, Babyn P, Chapman D (2017) Mechanical and biological effects of ultrasound: a review of present knowledge. Ultrasound Med Biol 43:1085–1104. https://doi.org/10.1016/j.ultrasmedbio.2017.01.023
Mainprize T, Lipsman N, Huang Y, Meng Y, Bethune A, Ironside S, Heyn C, Alkins R, Trudeau M, Sahgal A, Perry J, Hynynen K (2019) Blood-brain barrier opening in primary brain tumors with non-invasive MR-guided focused ultrasound: a clinical safety and feasibility study. Sci Rep 9:321. https://doi.org/10.1038/s41598-018-36340-0
Choi JJ, Pernot M, Small SA, Konofagou EE (2007) Noninvasive, transcranial and localized opening of the blood-brain barrier using focused ultrasound in mice. Ultrasound Med Biol 33:95–104. https://doi.org/10.1016/j.ultrasmedbio.2006.07.018
Downs ME, Buch A, Sierra C, Karakatsani ME, Teichert T, Chen S, Konofagou EE, Ferrera VP (2015) Long-term safety of repeated blood-brain barrier opening via focused ultrasound with microbubbles in non-human primates performing a cognitive task. PLoS One 10:e0125911. https://doi.org/10.1371/journal.pone.0125911
Kung Y, Lan C, Hsiao M-Y, Sun MK, Hsu YH, Huang APH, Liao WH, Liu HL, Inserra C, Chen WS (2018) Focused shockwave induced blood-brain barrier opening and transfection. Sci Rep 8:2218. https://doi.org/10.1038/s41598-018-20672-y
Nishida T, Shimokawa H, Oi K, Tatewaki H, Uwatoku T, Abe K, Matsumoto Y, Kajihara N, Eto M, Matsuda T, Yasui H, Takeshita A, Sunagawa K (2004) Extracorporeal cardiac shock wave therapy markedly ameliorates ischemia-induced myocardial dysfunction in pigs in vivo. Circulation 110:3055–3061. https://doi.org/10.1161/01.CIR.0000148849.51177.97
Odijk M, van der Meer AD, Levner D, Kim HJ, van der Helm MW, Segerink LI, Frimat JP, Hamilton GA, Ingber DE, van den Berg A (2015) Measuring direct current trans-epithelial electrical resistance in organ-on-a-chip microsystems. Lab Chip 15:745–752. https://doi.org/10.1039/C4LC01219D
Rasenat S, Hartung G, Winkler BL, Rehberg I (1989) The shadowgraph method in convection experiments. Exp Fluids 7:412–420. https://doi.org/10.1007/BF00193424
Miller DL, Smith NB, Bailey MR, Czarnota GJ, Hynynen K, Makin IRS, Bioeffects Committee of the American Institute of Ultrasound in Medicine (2012) Overview of therapeutic ultrasound applications and safety considerations. J Ultrasound Med 31:623–634
Na S, Collin O, Chowdhury F, Tay B, Ouyang M, Wang Y, Wang N (2008) Rapid signal transduction in living cells is a unique feature of mechanotransduction. Proc Natl Acad Sci 105:6626–6631. https://doi.org/10.1073/pnas.0711704105
Papachroni KK, Karatzas DN, Papavassiliou KA, Basdra EK, Papavassiliou AG (2009) Mechanotransduction in osteoblast regulation and bone disease. Trends Mol Med 15:208–216. https://doi.org/10.1016/j.molmed.2009.03.001
Dupont S (2016) Role of YAP/TAZ in cell-matrix adhesion-mediated signalling and mechanotransduction. Exp Cell Res 343:42–53. https://doi.org/10.1016/j.yexcr.2015.10.034
Samak G, Gangwar R, Crosby LM, Desai LP, Wilhelm K, Waters CM, Rao RK (2014) Cyclic stretch disrupts apical junctional complexes in Caco-2 cell monolayers by a JNK-2-, c-Src-, and MLCK-dependent mechanism. Am J Physiol Gastrointest Liver Physiol 306:G947–G958. https://doi.org/10.1152/ajpgi.00396.2013
Giusti S, Sbrana T, La Marca M et al (2014) A novel dual-flow bioreactor simulates increased fluorescein permeability in epithelial tissue barriers. Biotechnol J 9:1175–1184. https://doi.org/10.1002/biot.201400004
Duan Y, Gotoh N, Yan Q, du Z, Weinstein AM, Wang T, Weinbaum S (2008) Shear-induced reorganization of renal proximal tubule cell actin cytoskeleton and apical junctional complexes. Proc Natl Acad Sci U S A 105:11418–11423. https://doi.org/10.1073/pnas.0804954105
Chakrabarti S, Kobayashi KS, Flavell RA, Marks CB, Miyake K, Liston DR, Fowler KT, Gorelick FS, Andrews NW (2003) Impaired membrane resealing and autoimmune myositis in synaptotagmin VII-deficient mice. J Cell Biol 162:543–549. https://doi.org/10.1083/jcb.200305131
Keese CR, Wegener J, Walker SR, Giaever I (2004) Electrical wound-healing assay for cells in vitro. Proc Natl Acad Sci 101:1554–1559. https://doi.org/10.1073/pnas.0307588100
Moosavi-Nejad SF, Hosseini SHR, Satoh M, Takayama K (2006) Shock wave induced cytoskeletal and morphological deformations in a human renal carcinoma cell line. Cancer Sci 97:296–304. https://doi.org/10.1111/j.1349-7006.2006.00172.x
Lukes P, Sunka P, Hoffer P, Stelmashuk V, Pouckova P, Zadinova M, Zeman J, Dibdiak L, Kolarova H, Tomankova K, Binder S, Benes J (2014) Focused tandem shock waves in water and their potential application in cancer treatment. Shock Waves 24:51–57. https://doi.org/10.1007/s00193-013-0462-7
Hatano B, Matsumoto Y, Otani N et al (2011) Traumatic brain injury caused by laser-induced shock wave in rats: a novel laboratory model for studying blast-induced traumatic brain injury. In: Jansen ED, Thomas RJ (eds) International Society for Optics and Photonics, p 78971V
Funding
This study was supported, in part, by a grant for the Translational Systems Biology and Medicine Initiative (TSBMI) from the Ministry of Education, Culture, Sports, Science and Technology of Japan and JSPS KAKENHI Grant Number 23760107.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1.88 mb)
Rights and permissions
About this article
Cite this article
Monden, Y., Tsukamoto, A., Ushida, T. et al. An in vitro model of temporal enhancement of epithelium barrier permeability by low-energy shock waves without contrast agents. Med Biol Eng Comput 58, 1987–1993 (2020). https://doi.org/10.1007/s11517-020-02207-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11517-020-02207-8