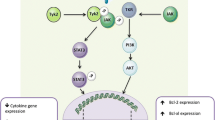
Abstract
An effective protective inflammatory response in the brain is crucial for the clearance of pathogens (e.g. microbes, amyloid fibrils, prionSC) and should be closely regulated. However, the CNS seems to have limited tissue resilience to withstand the detrimental effects of uncontrolled inflammation compromising functional recovery and tissue repair. Newly described neuro-immune-regulators (NIREGs) are functionally related proteins regulating the severity and duration of the host inflammatory response. NIREGs such as CD200, CD47 and CX3CL1 are vital for increasing tissue resilience and are constitutively expressed by neurons. The interaction with co-receptors (CD200R, CD172a, CX3CR1) will maintain microglia in the resting phenotype, directing aggressive microglia phenotype and limiting bystander injuries. Neurons can also express many of the complement NIREGs (CD55, CD46, CD59 and factor H). Neurons and glia also express suppressor of cytokine signaling proteins (SOCS) down regulating janus kinase–signal transducer and activator of transcription (JAK/STAT) pathway and to lead to the polarization of microglia towards anti-inflammatory phenotype. Other NIREGs such as serine protease inhibitors (serpins) and thrombomodulin (CD141) inhibit neurotoxic systemic coagulation proteins such as thrombin. The unfolded protein response (UPR) detects misfolded proteins and other stressors to prevent irreversible cell injury. Microglial pattern recognition receptors (PRR) (TREM-2, CR3, FcγR) are important to clear apoptotic cells and cellular debris but in non-phlogystic manner through inhibitory signaling pathways. The TYRO3, Axl, Mer (TAM) tyrosine receptor kinases activated by Gas 6 and PROS1 regulate inflammation by inhibiting Toll like receptors (TLR) /JAK-STAT activation and contribute to NIREG’s functions.




Similar content being viewed by others
Abbreviations
- Aβ:
-
fibrillary beta amyloid
- ATP:
-
adenosine triphosphate
- ACAMPS:
-
apoptotic- cell- associated molecular patterns
- AD:
-
alzheimer’s disease
- APP:
-
amyloid β precursor protein
- BBB:
-
blood brain barrier
- BNIP3:
-
Bcl-2 adenovirus E1B 19-kDa interacting protein 3
- C:
-
complement
- CNS:
-
central nervous system
- CP:
-
C pathway
- CR:
-
C receptors
- CREGs:
-
complement NIREGs
- CRRY:
-
complement receptor-related protein y
- CSF:
-
cerebro spinal fluid
- DAF:
-
decay accelerating factor
- DAMPs:
-
danger associated molecular patterns
- DC:
-
dendritic cell
- Dok2:
-
downstream tyrosine kinase 2
- EAE :
-
experimental autoimmune encephalitis
- EGF:
-
epidermal growth factor
- ER:
-
endoplasmic reticulum
- FH:
-
factor H
- Gas 6 :
-
growth arrest specific 6
- GBM:
-
Glioblastoma multiforme
- GSLC:
-
glioma stem like cells
- GPI :
-
glycosyl phosphatidyl inositol
- HMGB1:
-
high mobilty group box protein 1
- HSP:
-
heat shock proteins
- IFN:
-
interferon
- IgSF:
-
immunoglobulin superfamily
- ITAM:
-
immune receptor tyrosine based activation motif
- ITIM:
-
immune receptor tyrosine based inhibition motif
- JAK:
-
janus kinase
- LPS:
-
lipopolysaccharide
- MAC:
-
membrane attack complex
- MAPK:
-
p38 mitogen activated kinase
- MBL:
-
mannose binding lectin
- MCP:
-
membrane cofactor protein
- MF-EGF 8:
-
milk fat globule epidermal growth factor 8
- MMP:
-
matrix metalloproteinase
- MS :
-
multiple sclerosis
- MHC:
-
major histocompatibility complex
- MMR:
-
macrophage mannose receptor
- NGF:
-
nerve growth factor
- NMDA:
-
N-methyl-D-aspartate receptor
- NIREG:
-
neuroimmune regulatory protein
- NK:
-
natural killer
- NLR:
-
nucleotide-binding oligomerization domain-like receptors
- NLRP3:
-
nucleotide binding leucine rich protein 3
- NO:
-
nitric oxide
- NSP:
-
neuroserpin
- PAI:
-
plasminogen activator inhibitor
- PAMPs:
-
pathogen-associated molecular patterns
- PAR :
-
protease activated receptor
- PEDF:
-
pigment epithelium derived factor
- PI3-K:
-
phosphatidylinositol 3-kinase
- PN-1:
-
protease glial derived nexin −1
- PPAR-γ:
-
peroxisome proliferator activated receptor gamma
- PROS1:
-
protein S
- PRR:
-
pattern recognition receptor
- PS:
-
phosphatidyl serine
- PT:
-
prothrombin
- RAGE:
-
receptor for advanced glycated end products
- Ras GAP:
-
Ras p21 protein activator 1
- ROS:
-
reactive oxygen species
- SAMPs:
-
self associated molecular patterns
- Serpins:
-
serine protease inhibitors
- Siglecs:
-
sialic acid binding immunoglobulin like lectins
- SIRP:
-
signal regulatory protein
- SHP:
-
SH2 domain-containing phosphatase
- SOCS :
-
suppressor of cytokine signaling proteins
- SR:
-
scavenger receptor
- ssDNA:
-
single stranded DNA
- STAT:
-
signal transducers and activators of transcription
- TAM :
-
TYRO3,Axl,Mer
- TGF:
-
transforming growth factor
- TJ:
-
tight junctions
- TLR:
-
toll like receptors
- TM:
-
thrombomodulin
- TNF:
-
tumor necrosis factor
- tPA:
-
tissue plasminogen
- TREM:
-
triggering receptor of myeloid cells
- Tregs:
-
T regulatory cells
- TSP:
-
thrombospondins
- UPR:
-
unfolded protein response
References
Abbott NJ (1995) Morphology of nonmammalian glial cells: functional implications. In: Neuroglia. Oxford University Press, pp 97–116
Abeyama K, Stern DM, Ito Y et al (2005) The N-terminal domain of thrombomodulin sequesters high-mobility group-B1 protein, a novel antiinflammatory mechanism. J Clin Invest 115:1267–1274. https://doi.org/10.1172/JCI22782
Adams RA, Bauer J, Flick MJ et al (2007) The fibrin-derived γ377-395 peptide inhibits microglia activation and suppresses relapsing paralysis in central nervous system autoimmune disease. J Exp Med 204:571–582. https://doi.org/10.1084/jem.20061931
Akiyama H, McGeer PL (1990) Brain microglia constitutively express beta-2 integrins. J Neuroimmunol 30:81–93
Alblas J, Honing H, de Lavalette CR et al (2005) Signal regulatory protein alpha ligation induces macrophage nitric oxide production through JAK/STAT- and phosphatidylinositol 3-kinase/Rac1/NAPDH oxidase/H2O2-dependent pathways. Mol Cell Biol 25:7181–7192. https://doi.org/10.1128/MCB.25.16.7181-7192.2005
Angata T, Kerr SC, Greaves DR et al (2002) Cloning and characterization of human Siglec-11. A recently evolved signaling molecule that can interact with SHP-1 and SHP-2 and is expressed by tissue macrophages, including brain microglia. J Biol Chem 277:24466–24474. https://doi.org/10.1074/jbc.M202833200
Arvidsson A, Collin T, Kirik D et al (2002) Neuronal replacement from endogenous precursors in the adult brain after stroke. Nat Med 8:963–970. https://doi.org/10.1038/nm747
Asch AS, Leung LL, Shapiro J, Nachman RL (1986) Human brain glial cells synthesize thrombospondin. Proc Natl Acad Sci U S A 83:2904–2908
Bachstetter AD, Morganti JM, Jernberg J et al (2011) Fractalkine and CX 3 CR1 regulate hippocampal neurogenesis in adult and aged rats. Neurobiol Aging 32:2030–2044. https://doi.org/10.1016/j.neurobiolaging.2009.11.022
Baker BJ, Akhtar LN, Benveniste EN (2009) SOCS1 and SOCS3 in the control of CNS immunity. Trends Immunol 30:392–400. https://doi.org/10.1016/j.it.2009.07.001
Bamberger ME, Harris ME, McDonald DR et al (2003) A cell surface receptor complex for fibrillar beta-amyloid mediates microglial activation. J Neurosci 23:2665–2674
Barclay AN, Van den Berg TK (2014) The interaction between signal regulatory protein alpha (SIRPα) and CD47: structure, function, and therapeutic target. Annu Rev Immunol 32:25–50. https://doi.org/10.1146/annurev-immunol-032713-120142
Barclay AN, Ward HA (1982) Purification and chemical characterisation of membrane glycoproteins from rat thymocytes and brain, recognised by monoclonal antibody MRC OX 2. Eur J Biochem 129:447–458
Barnum S (1995) Complement biosynthesis in the central-nervous-system. Crit Rev Oral Biol Med 6:132–146. https://doi.org/10.1177/10454411950060020301
Bazan JF, Bacon KB, Hardiman G et al (1997) A new class of membrane-bound chemokine with a CX3C motif. Nature 385:640–644. https://doi.org/10.1038/385640a0
Belkin DA, Mitsui H, Wang CQF et al (2013) CD200 upregulation in vascular endothelium surrounding cutaneous squamous cell carcinoma. JAMA Dermatol 149:178–186. https://doi.org/10.1001/jamadermatol.2013.1609
Bianchi ME (2007) DAMPs, PAMPs and alarmins: all we need to know about danger. J Leukoc Biol 81:1–5. https://doi.org/10.1189/jlb.0306164
Bianchi ME (2009) HMGB1 loves company. J Leukoc Biol 86:573–576. https://doi.org/10.1189/jlb.1008585
Bilak MM, Becerra SP, Vincent AM et al (2002) Identification of the neuroprotective molecular region of pigment epithelium-derived factor and its binding sites on motor neurons. J Neurosci 22:9378–9386
Binder MD, Cate HS, Prieto AL et al (2008) Gas6 deficiency increases oligodendrocyte loss and microglial activation in response to cuprizone-induced demyelination. J Neurosci 28:5195–5206. https://doi.org/10.1523/JNEUROSCI.1180-08.2008
Birge RB, Ucker DS (2008) Innate apoptotic immunity: the calming touch of death. Cell Death Differ 15:1096–1102. https://doi.org/10.1038/cdd.2008.58
Bosurgi L, Bernink JH, Delgado Cuevas V et al (2013) Paradoxical role of the proto-oncogene Axl and Mer receptor tyrosine kinases in colon cancer. Proc Natl Acad Sci U S A 110:13091–13096. https://doi.org/10.1073/pnas.1302507110
Botto M, Dell’Agnola C, Bygrave AE et al (1998) Homozygous C1q deficiency causes glomerulonephritis associated with multiple apoptotic bodies. Nat Genet 19:56–59. https://doi.org/10.1038/ng0598-56
Boudakov I, Liu J, Fan N et al (2007) Mice lacking CD200R1 show absence of suppression of lipopolysaccharide-induced tumor necrosis factor-alpha and mixed leukocyte culture responses by CD200. Transplantation 84:251–257. https://doi.org/10.1097/01.tp.0000269795.04592.cc
Bsibsi M, Ravid R, Gveric D, van Noort JM (2002) Broad expression of Toll-like receptors in the human central nervous system. J Neuropathol Exp Neurol 61:1013–1021
Buisson A, Nicole O, Docagne F et al (1998) Up-regulation of a serine protease inhibitor in astrocytes mediates the neuroprotective activity of transforming growth factor beta1. FASEB J Off Publ Fed Am Soc Exp Biol 12:1683–1691
Buisson A, Lesne S, Docagne F et al (2003) Transforming growth factor-beta and ischemic brain injury. Cell Mol Neurobiol 23:539–550
Burudi EM, Riese S, Stahl PD, Régnier-Vigouroux A (1999) Identification and functional characterization of the mannose receptor in astrocytes. Glia 25:44–55
Camenisch TD, Koller BH, Earp HS, Matsushima GK (1999) A novel receptor tyrosine kinase, Mer, inhibits TNF-alpha production and lipopolysaccharide-induced endotoxic shock. J Immunol Baltim Md 1950 162:3498–3503
Canova C, Neal JW, Gasque P (2006) Expression of innate immune complement regulators on brain epithelial cells during human bacterial meningitis. J Neuroinflammation 3:22. https://doi.org/10.1186/1742-2094-3-22
Cardona AE, Pioro EP, Sasse ME et al (2006) Control of microglial neurotoxicity by the fractalkine receptor. Nat Neurosci 9:917–924. https://doi.org/10.1038/nn1715
Carson MJ, Doose JM, Melchior B et al (2006) CNS immune privilege: hiding in plain sight. Immunol Rev 213:48–65. https://doi.org/10.1111/j.1600-065X.2006.00441.x
Casadevall A, Pirofski LA (1999) Host-pathogen interactions: redefining the basic concepts of virulence and pathogenicity. Infect Immun 67:3703–3713
Castiglioni A, Canti V, Rovere-Querini P, Manfredi AA (2011) High-mobility group box 1 (HMGB1) as a master regulator of innate immunity. Cell Tissue Res 343:189–199. https://doi.org/10.1007/s00441-010-1033-1
Cavanaugh KP, Gurwitz D, Cunningham DD, Bradshaw RA (1990) Reciprocal modulation of astrocyte stellation by thrombin and protease nexin-1. J Neurochem 54:1735–1743
Cerwenka A, Lanier LL (2001) Natural killer cells, viruses and cancer. Nat Rev Immunol 1:41–49. https://doi.org/10.1038/35095564
Chan A, Magnus T, Gold R (2001) Phagocytosis of apoptotic inflammatory cells by microglia and modulation by different cytokines: mechanism for removal of apoptotic cells in the inflamed nervous system. Glia 33:87–95
Chang GH, Barbaro NM, Pieper RO (2000) Phosphatidylserine-dependent phagocytosis of apoptotic glioma cells by normal human microglia, astrocytes, and glioma cells. Neuro-Oncology 2:174–183
Chaudhari N, Talwar P, Parimisetty A et al (2014) A molecular web: endoplasmic reticulum stress, inflammation, and oxidative stress. Front Cell Neurosci 8:213. https://doi.org/10.3389/fncel.2014.00213
Chen D-X, He H, Gorczynski RM (2005) Synthetic peptides from the N-terminal regions of CD200 and CD200R1 modulate immunosuppressive and anti-inflammatory effects of CD200-CD200R1 interaction. Int Immunol 17:289–296. https://doi.org/10.1093/intimm/dxh208
Chen G-Y, Tang J, Zheng P, Liu Y (2009) CD24 and siglec-10 selectively repress tissue damage-induced immune responses. Science 323:1722–1725. https://doi.org/10.1126/science.1168988
Chen G-Y, Brown NK, Zheng P, Liu Y (2014) Siglec-G/10 in self-nonself discrimination of innate and adaptive immunity. Glycobiology 24:800–806. https://doi.org/10.1093/glycob/cwu068
Chung W-S, Clarke LE, Wang GX et al (2013) Astrocytes mediate synapse elimination through MEGF10 and MERTK pathways. Nature 504:394–400. https://doi.org/10.1038/nature12776
Clark IA, Vissel B (2015) Amyloid β: one of three danger-associated molecules that are secondary inducers of the proinflammatory cytokines that mediate Alzheimer’s disease. Br J Pharmacol 172:3714–3727. https://doi.org/10.1111/bph.13181
Claude J, Linnartz-Gerlach B, Kudin AP et al (2013) Microglial CD33-related Siglec-E inhibits neurotoxicity by preventing the phagocytosis-associated oxidative burst. J Neurosci 33:18270–18276. https://doi.org/10.1523/JNEUROSCI.2211-13.2013
Colonna M (2003) TREMs in the immune system and beyond. Nat Rev Immunol 3:445–453. https://doi.org/10.1038/nri1106
Coraci IS, Husemann J, Berman JW et al (2002) CD36, a class B scavenger receptor, is expressed on microglia in Alzheimer’s disease brains and can mediate production of reactive oxygen species in response to beta-amyloid fibrils. Am J Pathol 160:101–112
de Córdoba SR, de Jorge EG (2008) Translational mini-review series on complement factor H: genetics and disease associations of human complement factor H. Clin Exp Immunol 151:1–13. doi:https://doi.org/10.1111/j.1365-2249.2007.03552.x
Crocker PR (2005) Siglecs in innate immunity. Curr Opin Pharmacol 5:431–437. https://doi.org/10.1016/j.coph.2005.03.003
D’Cruz PM, Yasumura D, Weir J et al (2000) Mutation of the receptor tyrosine kinase gene Mertk in the retinal dystrophic RCS rat. Hum Mol Genet 9:645–651
Davalos D, Ryu JK, Merlini M et al (2012) Fibrinogen-induced perivascular microglial clustering is required for the development of axonal damage in neuroinflammation. Nat Commun 3:1227. https://doi.org/10.1038/ncomms2230
de Vries HE, Hendriks JJA, Honing H et al (2002) Signal-regulatory protein alpha-CD47 interactions are required for the transmigration of monocytes across cerebral endothelium. J Immunol Baltim Md 1950(168):5832–5839
Deczkowska A, Keren-Shaul H, Weiner A et al (2018) Disease-associated microglia: a universal immune sensor of neurodegeneration. Cell 173:1073–1081. https://doi.org/10.1016/j.cell.2018.05.003
Dentesano G, Serratosa J, Tusell JM et al (2014) CD200R1 and CD200 expression are regulated by PPAR-γ in activated glial cells. Glia 62:982–998. https://doi.org/10.1002/glia.22656
Docagne F, Nicole O, Marti HH et al (1999) Transforming growth factor-beta1 as a regulator of the serpins/t-PA axis in cerebral ischemia. FASEB J Off Publ Fed Am Soc Exp Biol 13:1315–1324
Donnelly DJ, Popovich PG (2008) Inflammation and its role in neuroprotection, axonal regeneration and functional recovery after spinal cord injury. Exp Neurol 209:378–388. https://doi.org/10.1016/j.expneurol.2007.06.009
Donnelly DJ, Longbrake EE, Shawler TM et al (2011) Deficient CX3CR1 signaling promotes recovery after mouse spinal cord injury by limiting the recruitment and activation of Ly6Clo/iNOS+ macrophages. J Neurosci 31:9910–9922. https://doi.org/10.1523/JNEUROSCI.2114-11.2011
Ehlers MR (2000) CR3: a general purpose adhesion-recognition receptor essential for innate immunity. Microbes Infect 2:289–294
El Khoury JB, Moore KJ, Means TK et al (2003) CD36 mediates the innate host response to beta-amyloid. J Exp Med 197:1657–1666. https://doi.org/10.1084/jem.20021546
Elward K, Gasque P (2003) “Eat me” and “don’t eat me” signals govern the innate immune response and tissue repair in the CNS: emphasis on the critical role of the complement system. Mol Immunol 40:85–94
Elward K, Griffiths M, Mizuno M et al (2005) CD46 plays a key role in tailoring innate immune recognition of apoptotic and necrotic cells. J Biol Chem 280:36342–36354
Esmon C (2005) Do-all receptor takes on coagulation, inflammation. Nat Med 11:475–477. https://doi.org/10.1038/nm0505-475
Fadok VA, Bratton DL, Konowal A et al (1998) Macrophages that have ingested apoptotic cells in vitro inhibit proinflammatory cytokine production through autocrine/paracrine mechanisms involving TGF-beta, PGE2, and PAF. J Clin Invest 101:890–898. https://doi.org/10.1172/JCI1112
Fadok VA, Bratton DL, Rose DM et al (2000) A receptor for phosphatidylserine-specific clearance of apoptotic cells. Nature 405:85–90. https://doi.org/10.1038/35011084
Fenner JE, Starr R, Cornish AL et al (2006) Suppressor of cytokine signaling 1 regulates the immune response to infection by a unique inhibition of type I interferon activity. Nat Immunol 7:33–39. https://doi.org/10.1038/ni1287
Ferreira VP, Pangburn MK, Cortés C (2010) Complement control protein factor H: the good, the bad, and the inadequate. Mol Immunol 47:2187–2197. https://doi.org/10.1016/j.molimm.2010.05.007
Floden AM, Combs CK (2011) Microglia demonstrate age-dependent interaction with amyloid-beta fibrils. J Alzheimers Dis 25:279–293. https://doi.org/10.3233/JAD-2011-101014
Flügel A, Schwaiger FW, Neumann H et al (2000) Neuronal FasL induces cell death of encephalitogenic T lymphocytes. Brain Pathol Zurich Switz 10:353–364
Fonseca MI, Zhou J, Botto M, Tenner AJ (2004) Absence of C1q leads to less neuropathology in transgenic mouse models of Alzheimer’s disease. J Neurosci 24:6457–6465. https://doi.org/10.1523/JNEUROSCI.0901-04.2004
Fourgeaud L, Través PG, Tufail Y et al (2016) TAM receptors regulate multiple features of microglial physiology. Nature 532:240–244. https://doi.org/10.1038/nature17630
Freria CM, Hall JCE, Wei P et al (2017) Deletion of the fractalkine receptor, CX3CR1, improves endogenous repair, axon sprouting, and synaptogenesis after spinal cord injury in mice. J Neurosci 37:3568–3587. https://doi.org/10.1523/JNEUROSCI.2841-16.2017
Frisullo G, Angelucci F, Caggiula M et al (2006) pSTAT1, pSTAT3, and T-bet expression in peripheral blood mononuclear cells from relapsing-remitting multiple sclerosis patients correlates with disease activity. J Neurosci Res 84:1027–1036. https://doi.org/10.1002/jnr.20995
Gaikwad S, Larionov S, Wang Y et al (2009) Signal regulatory protein-beta1: a microglial modulator of phagocytosis in Alzheimer’s disease. Am J Pathol 175:2528–2539. https://doi.org/10.2353/ajpath.2009.090147
Galea I, Palin K, Newman TA et al (2005) Mannose receptor expression specifically reveals perivascular macrophages in normal, injured, and diseased mouse brain. Glia 49:375–384. https://doi.org/10.1002/glia.20124
Gallucci S, Matzinger P (2001) Danger signals: SOS to the immune system. Curr Opin Immunol 13:114–119
Garcia J, Anderson ML (1990) Chapter 13: circulatory disorders and their effects on the brain. In: Davis RL, Robertson DM (eds) Textbook of neuropathology, subsequent edition. Williams & Wilkins, Baltimore, pp 621–719
Gasque P (2004) Complement: a unique innate immune sensor for danger signals. Mol Immunol 41:1089–1098. https://doi.org/10.1016/j.molimm.2004.06.011
Gasque P, Thomas A, Fontaine M, Morgan BP (1996) Complement activation on human neuroblastoma cell lines in vitro: route of activation and expression of functional complement regulatory proteins. J Neuroimmunol 66:29–40
Gasque P, Neal JW, Singhrao SK et al (2002) Roles of the complement system in human neurodegenerative disorders: pro-inflammatory and tissue remodeling activities. Mol Neurobiol 25:1–17
Gewurz H, Ying SC, Jiang H, Lint TF (1993) Nonimmune activation of the classical complement pathway. Behring Inst Mitt:138–147
Gingrich MB, Traynelis SF (2000) Serine proteases and brain damage - is there a link? Trends Neurosci 23:399–407
Gitik M, Liraz-Zaltsman S, Oldenborg P-A et al (2011) Myelin down-regulates myelin phagocytosis by microglia and macrophages through interactions between CD47 on myelin and SIRPα (signal regulatory protein-α) on phagocytes. J Neuroinflammation 8:24. https://doi.org/10.1186/1742-2094-8-24
Gold R, Hartung HP, Lassmann H (1997) T-cell apoptosis in autoimmune diseases: termination of inflammation in the nervous system and other sites with specialized immune-defense mechanisms. Trends Neurosci 20:399–404
Goldszmid RS, Trinchieri G (2012) The price of immunity. Nat Immunol 13:932–938. https://doi.org/10.1038/ni.2422
Graf R, Freyberg M, Kaiser D, Friedl P (2002) Mechanosensitive induction of apoptosis in fibroblasts is regulated by thrombospondin-1 and integrin associated protein (CD47). Apoptosis 7:493–498. https://doi.org/10.1023/A:1020634924760
Griffiths M, Neal JW, Gasque P (2007) Innate immunity and protective neuroinflammation: new emphasis on the role of neuroimmune regulatory proteins. Int Rev Neurobiol 82:29–55
Griffiths MR, Gasque P, Neal JW (2009a) The multiple roles of the innate immune system in the regulation of apoptosis and inflammation in the brain. J Neuropathol Exp Neurol 68:217–226
Griffiths MR, Neal JW, Fontaine M et al (1950) (2009b) Complement factor H, a marker of self protects against experimental autoimmune encephalomyelitis. J Immunol Baltim Md 182:4368–4377. https://doi.org/10.4049/jimmunol.0800205
Griffiths MR, Gasque P, Neal JW (2010) The regulation of the CNS innate immune response is vital for the restoration of tissue homeostasis (repair) after acute brain injury: a brief review. Int J Inflamm 2010:151097. https://doi.org/10.4061/2010/151097
Grimsley C, Ravichandran KS (2003) Cues for apoptotic cell engulfment: eat-me, don’t eat-me and come-get-me signals. Trends Cell Biol 13:648–656
Gu J-J, Chen J-B, Zhang J-H et al (2016) Recombinant human soluble thrombomodulin protects against brain injury in a CVST rat model, via downregulation of the HMGB1-RAGE axis. Mol Med Rep 14:5217–5222. https://doi.org/10.3892/mmr.2016.5891
Han HS, Suk K (2005) The function and integrity of the neurovascular unit rests upon the integration of the vascular and inflammatory cell systems. Curr Neurovasc Res 2:409–423
Han MH, Lundgren DH, Jaiswal S et al (2012) Janus-like opposing roles of CD47 in autoimmune brain inflammation in humans and mice. J Exp Med 209:1325–1334. https://doi.org/10.1084/jem.20101974
Hanada T, Tanaka K, Matsumura Y et al (2005) Induction of hyper Th1 cell-type immune responses by dendritic cells lacking the suppressor of cytokine signaling-1 gene. J Immunol Baltim Md 1950 174:4325–4332
Hanayama R, Tanaka M, Miwa K et al (2002) Identification of a factor that links apoptotic cells to phagocytes. Nature 417:182–187. https://doi.org/10.1038/417182a
Hanisch U-K, Kettenmann H (2007) Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat Neurosci 10:1387–1394. https://doi.org/10.1038/nn1997
Hastings GA, Coleman TA, Haudenschild CC et al (1997) Neuroserpin, a brain-associated inhibitor of tissue plasminogen activator is localized primarily in neurons. Implications for the regulation of motor learning and neuronal survival. J Biol Chem 272:33062–33067
Hauwel M, Furon E, Canova C et al (2005) Innate (inherent) control of brain infection, brain inflammation and brain repair: the role of microglia, astrocytes, “protective” glial stem cells and stromal ependymal cells. Brain Res Brain Res Rev 48:220–233
Helmy KY, Katschke KJ, Gorgani NN et al (2006) CRIg: a macrophage complement receptor required for phagocytosis of circulating pathogens. Cell 124:915–927. https://doi.org/10.1016/j.cell.2005.12.039
Hernangómez M, Mestre L, Correa FG et al (2012) CD200-CD200R1 interaction contributes to neuroprotective effects of anandamide on experimentally induced inflammation. Glia 60:1437–1450. https://doi.org/10.1002/glia.22366
Hoarau JJ, Krejbich-Trotot P, Jaffar-Bandjee MC et al (2011) Activation and control of CNS innate immune responses in health and diseases: a balancing act finely tuned by neuroimmune regulators (NIReg). CNS Neurol Disord Drug Targets 10:25–43
Hoek RM, Ruuls SR, Murphy CA et al (2000) Down-regulation of the macrophage lineage through interaction with OX2 (CD200). Science 290:1768–1771
Hoffmann MC, Nitsch C, Scotti AL et al (1992) The prolonged presence of glia-derived nexin, an endogenous protease inhibitor, in the hippocampus after ischemia-induced delayed neuronal death. Neuroscience 49:397–408
Hong S, Beja-Glasser VF, Nfonoyim BM et al (2016) Complement and microglia mediate early synapse loss in Alzheimer mouse models. Science 352:712–716. https://doi.org/10.1126/science.aad8373
Hotamisligil GS (2010) Endoplasmic reticulum stress and the inflammatory basis of metabolic disease. Cell 140:900–917. https://doi.org/10.1016/j.cell.2010.02.034
Huang C, Ma R, Sun S et al (2008) JAK2-STAT3 signaling pathway mediates thrombin-induced proinflammatory actions of microglia in vitro. J Neuroimmunol 204:118–125. https://doi.org/10.1016/j.jneuroim.2008.07.004
Husemann J, Loike JD, Anankov R et al (2002) Scavenger receptors in neurobiology and neuropathology: their role on microglia and other cells of the nervous system. Glia 40:195–205. https://doi.org/10.1002/glia.10148
Imler JL, Hoffmann JA (2001) Toll receptors in innate immunity. Trends Cell Biol 11:304–311
Jakus Z, Fodor S, Abram CL et al (2007) Immunoreceptor-like signaling by beta 2 and beta 3 integrins. Trends Cell Biol 17:493–501. https://doi.org/10.1016/j.tcb.2007.09.001
Janeway CA (1992) The immune system evolved to discriminate infectious nonself from noninfectious self. Immunol Today 13:11–16. https://doi.org/10.1016/0167-5699(92)90198-G
Janeway CA, Medzhitov R (2002) Innate immune recognition. Annu Rev Immunol 20:197–216. https://doi.org/10.1146/annurev.immunol.20.083001.084359
Keren-Shaul H, Spinrad A, Weiner A et al (2017) A unique microglia type associated with restricting development of Alzheimer’s disease. Cell 169:1276–1290.e17. https://doi.org/10.1016/j.cell.2017.05.018
Klune JR, Dhupar R, Cardinal J et al (2008) HMGB1: endogenous danger signaling. Mol Med Camb Mass 14:476–484. https://doi.org/10.2119/2008-00034.Klune
Koning N, Bö L, Hoek RM, Huitinga I (2007) Downregulation of macrophage inhibitory molecules in multiple sclerosis lesions. Ann Neurol 62:504–514. https://doi.org/10.1002/ana.21220
Koning N, Swaab DF, Hoek RM, Huitinga I (2009) Distribution of the immune inhibitory molecules CD200 and CD200R in the normal central nervous system and multiple sclerosis lesions suggests neuron-glia and glia-glia interactions. J Neuropathol Exp Neurol 68:159–167. https://doi.org/10.1097/NEN.0b013e3181964113
Krasemann S, Madore C, Cialic R et al (2017) The TREM2-APOE pathway drives the transcriptional phenotype of dysfunctional microglia in neurodegenerative diseases. Immunity 47:566–581.e9. https://doi.org/10.1016/j.immuni.2017.08.008
Lamy L, Foussat A, Brown EJ et al (2007) Interactions between CD47 and thrombospondin reduce inflammation. J Immunol Baltim Md 1950(178):5930–5939
Land WG (2015) The role of damage-associated molecular patterns in human diseases. Sultan Qaboos Univ Med J 15:e9–e21
Latour S, Tanaka H, Demeure C et al (2001) Bidirectional negative regulation of human T and dendritic cells by CD47 and its cognate receptor signal-regulator protein-alpha: down-regulation of IL-12 responsiveness and inhibition of dendritic cell activation. J Immunol Baltim Md 1950 167:2547–2554
Lawrence T, Natoli G (2011) Transcriptional regulation of macrophage polarization: enabling diversity with identity. Nat Rev Immunol 11:750–761. https://doi.org/10.1038/nri3088
Lee S, Varvel NH, Konerth ME et al (2010) CX3CR1 deficiency alters microglial activation and reduces beta-amyloid deposition in two Alzheimer’s disease mouse models. Am J Pathol 177:2549–2562. https://doi.org/10.2353/ajpath.2010.100265
Lemke G, Rothlin CV (2008) Immunobiology of the TAM receptors. Nat Rev Immunol 8:327–336. https://doi.org/10.1038/nri2303
Leonardi-Essmann F, Emig M, Kitamura Y et al (2005) Fractalkine-upregulated milk-fat globule EGF factor-8 protein in cultured rat microglia. J Neuroimmunol 160:92–101. https://doi.org/10.1016/j.jneuroim.2004.11.012
Leyns CEG, Ulrich JD, Finn MB et al (2017) TREM2 deficiency attenuates neuroinflammation and protects against neurodegeneration in a mouse model of tauopathy. Proc Natl Acad Sci U S A 114:11524–11529. https://doi.org/10.1073/pnas.1710311114
Li J, Wang JJ, Zhang SX (2011) Preconditioning with endoplasmic reticulum stress mitigates retinal endothelial inflammation via activation of X-box binding protein 1. J Biol Chem 286:4912–4921. https://doi.org/10.1074/jbc.M110.199729
Liang KJ, Lee JE, Wang YD et al (2009) Regulation of dynamic behavior of retinal microglia by CX3CR1 signaling. Invest Ophthalmol Vis Sci 50:4444–4451. https://doi.org/10.1167/iovs.08-3357
Linnartz B, Wang Y, Neumann H (2010) Microglial immunoreceptor tyrosine-based activation and inhibition motif signaling in neuroinflammation. Int J Alzheimers Dis 2010. https://doi.org/10.4061/2010/587463
Liu Y, Bühring H-J, Zen K et al (2002) Signal regulatory protein (SIRPalpha), a cellular ligand for CD47, regulates neutrophil transmigration. J Biol Chem 277:10028–10036. https://doi.org/10.1074/jbc.M109720200
Liu Y, Chen G-Y, Zheng P (2009) CD24-Siglec G/10 discriminates danger- from pathogen-associated molecular patterns. Trends Immunol 30:557–561. https://doi.org/10.1016/j.it.2009.09.006
Lotze MT, Tracey KJ (2005) High-mobility group box 1 protein (HMGB1): nuclear weapon in the immune arsenal. Nat Rev Immunol 5:331–342. https://doi.org/10.1038/nri1594
Lowenstein PR (2002) Immunology of viral-vector-mediated gene transfer into the brain: an evolutionary and developmental perspective. Trends Immunol 23:23–30
Lu Q, Lemke G (2001) Homeostatic regulation of the immune system by receptor tyrosine kinases of the Tyro 3 family. Science 293:306–311. https://doi.org/10.1126/science.1061663
Lucin KM, Wyss-Coray T (2009) Immune activation in brain aging and neurodegeneration: too much or too little? Neuron 64:110–122. https://doi.org/10.1016/j.neuron.2009.08.039
Lynch MA (2014) The impact of neuroimmune changes on development of amyloid pathology; relevance to Alzheimer’s disease. Immunology 141:292–301. https://doi.org/10.1111/imm.12156
Lyons A, Downer EJ, Crotty S et al (2007) CD200 ligand receptor interaction modulates microglial activation in vivo and in vitro: a role for IL-4. J Neurosci 27:8309–8313. https://doi.org/10.1523/JNEUROSCI.1781-07.2007
Lyons A, Downer EJ, Costello DA et al (2012) Dok2 mediates the CD200Fc attenuation of Aβ-induced changes in glia. J Neuroinflammation 9:107. https://doi.org/10.1186/1742-2094-9-107
Macauley MS, Crocker PR, Paulson JC (2014) Siglec-mediated regulation of immune cell function in disease. Nat Rev Immunol 14:653–666. https://doi.org/10.1038/nri3737
Magnus T, Chan A, Grauer O et al (2001) Microglial phagocytosis of apoptotic inflammatory T cells leads to down-regulation of microglial immune activation. J Immunol Baltim Md 1950 167:5004–5010
Manna PP, Dimitry J, Oldenborg P-A, Frazier WA (2005) CD47 augments Fas/CD95-mediated apoptosis. J Biol Chem 280:29637–29644. https://doi.org/10.1074/jbc.M500922200
Martinez FO, Gordon S (2014) The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000prime Rep 6:13. https://doi.org/10.12703/P6-13
Martinez FO, Helming L, Gordon S (2009) Alternative activation of macrophages: an immunologic functional perspective. Annu Rev Immunol 27:451–483. https://doi.org/10.1146/annurev.immunol.021908.132532
Marzolo MP, von Bernhardi R, Inestrosa NC (1999) Mannose receptor is present in a functional state in rat microglial cells. J Neurosci Res 58:387–395
Matzinger P (2007) Friendly and dangerous signals: is the tissue in control? Nat Immunol 8:11–13. https://doi.org/10.1038/ni0107-11
McGeer PL, McGeer EG (1995) The inflammatory response system of brain: Implications for therapy of Alzheimer and other neurodegenerative diseases. Brain Res Rev 21:195–218. https://doi.org/10.1016/0165-0173(95)00011-9
Mead RJ, Singhrao SK, Neal JW et al (2002) The membrane attack complex of complement causes severe demyelination associated with acute axonal injury. J Immunol Baltim Md 1950 168:458–465
Mead RJ, Neal JW, Griffiths MR et al (2004) Deficiency of the complement regulator CD59a enhances disease severity, demyelination and axonal injury in murine acute experimental allergic encephalomyelitis. Lab Investig J Tech Methods Pathol 84:21–28. https://doi.org/10.1038/sj.labinvest.3700015
Medzhitov R, Janeway C (2000) Innate immune recognition: mechanisms and pathways. Immunol Rev 173:89–97
Medzhitov R, Schneider DS, Soares MP (2012) Disease tolerance as a defense strategy. Science 335:936–941. https://doi.org/10.1126/science.1214935
Meuth SG, Simon OJ, Grimm A et al (2008) CNS inflammation and neuronal degeneration is aggravated by impaired CD200-CD200R-mediated macrophage silencing. J Neuroimmunol 194:62–69. https://doi.org/10.1016/j.jneuroim.2007.11.013
Mihrshahi R, Barclay AN, Brown MH (2009) Essential roles for Dok2 and RasGAP in CD200 receptor-mediated regulation of human myeloid cells. J Immunol Baltim Md 1950 183:4879–4886. https://doi.org/10.4049/jimmunol.0901531
Morgan BP, Gasque P (1996) Expression of complement in the brain: role in health and disease. Immunol Today 17:461–466
Morgan BP, Meri S (1994) Membrane proteins that protect against complement lysis. Springer Semin Immunopathol 15:369–396
Morganti JM, Riparip L-K, Rosi S (2016) Call Off the Dog(ma): M1/M2 polarization is concurrent following traumatic brain injury. PLoS One 11:e0148001. https://doi.org/10.1371/journal.pone.0148001
Mukhopadhyay S, Plüddemann A, Hoe JC et al (2010) Immune inhibitory ligand CD200 induction by TLRs and NLRs limits macrophage activation to protect the host from meningococcal septicemia. Cell Host Microbe 8:236–247. https://doi.org/10.1016/j.chom.2010.08.005
Murray PJ, Wynn TA (2011) Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol 11:723–737. https://doi.org/10.1038/nri3073
Murray PJ, Allen JE, Biswas SK et al (2014) Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41:14–20. https://doi.org/10.1016/j.immuni.2014.06.008
Muto J, Yamasaki K, Taylor KR, Gallo RL (2009) Engagement of CD44 by hyaluronan suppresses TLR4 signaling and the septic response to LPS. Mol Immunol 47:449–456. https://doi.org/10.1016/j.molimm.2009.08.026
Nagata K, Ohashi K, Nakano T et al (1996) Identification of the product of growth arrest-specific gene 6 as a common ligand for Axl, Sky, and Mer receptor tyrosine kinases. J Biol Chem 271:30022–30027
Nguyen MD, Julien J-P, Rivest S (2002) Innate immunity: the missing link in neuroprotection and neurodegeneration? Nat Rev Neurosci 3:216–227. https://doi.org/10.1038/nrn752
Niego B, Samson AL, Petersen K-U, Medcalf RL (2011) Thrombin-induced activation of astrocytes in mixed rat hippocampal cultures is inhibited by soluble thrombomodulin. Brain Res 1381:38–51. https://doi.org/10.1016/j.brainres.2011.01.016
O’Keefe GM, Nguyen VT, Ping Tang LL, Benveniste EN (2001) IFN-gamma regulation of class II transactivator promoter IV in macrophages and microglia: involvement of the suppressors of cytokine signaling-1 protein. J Immunol Baltim Md 1950 166:2260–2269
Oldenborg P-A (2013) CD47: A Cell Surface Glycoprotein Which Regulates Multiple Functions of Hematopoietic Cells in Health and Disease. ISRN Hematol 2013:614619. https://doi.org/10.1155/2013/614619
Oldenborg PA, Zheleznyak A, Fang YF et al (2000) Role of CD47 as a marker of self on red blood cells. Science 288:2051–2054
Pachter JS, de Vries HE, Fabry Z (2003) The blood-brain barrier and its role in immune privilege in the central nervous system. J Neuropathol Exp Neurol 62:593–604
Pangburn MK, Rawal N, Cortes C et al (1950) (2009) Polyanion-induced self-association of complement factor H. J Immunol Baltim Md 182:1061–1068
Pender MP, Rist MJ (2001) Apoptosis of inflammatory cells in immune control of the nervous system: role of glia. Glia 36:137–144
Pimenova AA, Marcora E, Goate AM (2017) A tale of two genes: microglial apoe and trem2. Immunity 47:398–400. https://doi.org/10.1016/j.immuni.2017.08.015
Popovich PG, Longbrake EE (2008) Can the immune system be harnessed to repair the CNS? Nat Rev Neurosci 9:481–493. https://doi.org/10.1038/nrn2398
Pradeu T, Cooper EL (2012) The danger theory: 20 years later. Front Immunol 3:287. https://doi.org/10.3389/fimmu.2012.00287
Qin H, Roberts KL, Niyongere SA et al (2007) Molecular mechanism of lipopolysaccharide-induced SOCS-3 gene expression in macrophages and microglia. J Immunol Baltim Md 1950 179:5966–5976
Qin H, Niyongere SA, Lee SJ et al (2008) Expression and functional significance of SOCS-1 and SOCS-3 in astrocytes. J Immunol Baltim Md 1950 181:3167–3176
Qin H, Holdbrooks AT, Liu Y et al (2012a) SOCS3 deficiency promotes M1 macrophage polarization and inflammation. J Immunol Baltim Md 1950 189:3439–3448. https://doi.org/10.4049/jimmunol.1201168
Qin H, Yeh W-I, De Sarno P et al (2012b) Signal transducer and activator of transcription-3/suppressor of cytokine signaling-3 (STAT3/SOCS3) axis in myeloid cells regulates neuroinflammation. Proc Natl Acad Sci U S A 109:5004–5009. https://doi.org/10.1073/pnas.1117218109
Råberg L, Sim D, Read AF (2007) Disentangling genetic variation for resistance and tolerance to infectious diseases in animals. Science 318:812–814. https://doi.org/10.1126/science.1148526
Rajput PS, Lyden PD, Chen B et al (2014) Protease activated receptor-1 mediates cytotoxicity during ischemia using in vivo and in vitro models. Neuroscience 281:229–240. https://doi.org/10.1016/j.neuroscience.2014.09.038
Ransohoff RM (2016) A polarizing question: do M1 and M2 microglia exist? Nat Neurosci 19:987–991. https://doi.org/10.1038/nn.4338
Ransohoff RM, Perry VH (2009) Microglial physiology: unique stimuli, specialized responses. Annu Rev Immunol 27:119–145. https://doi.org/10.1146/annurev.immunol.021908.132528
Reinhold MI, Lindberg FP, Plas D et al (1995) In vivo expression of alternatively spliced forms of integrin-associated protein (CD47). J Cell Sci 108(Pt 11):3419–3425
Rosenblum MD, Olasz E, Woodliff JE et al (2004) CD200 is a novel p53-target gene involved in apoptosis-associated immune tolerance. Blood 103:2691–2698. https://doi.org/10.1182/blood-2003-09-3184
Rothlin CV, Ghosh S, Zuniga EI et al (2007) TAM receptors are pleiotropic inhibitors of the innate immune response. Cell 131:1124–1136. https://doi.org/10.1016/j.cell.2007.10.034
Rothlin CV, Carrera-Silva EA, Bosurgi L, Ghosh S (2015) TAM receptor signaling in immune homeostasis. Annu Rev Immunol 33:355–391. https://doi.org/10.1146/annurev-immunol-032414-112103
Ryu JK, Davalos D, Akassoglou K (2009) Fibrinogen signal transduction in the nervous system. J Thromb Haemost JTH 7(Suppl 1):151–154. https://doi.org/10.1111/j.1538-7836.2009.03438.x
Ryu JK, Davalos D, Akassoglou K (2009) Fibrinogen signal transduction in the nervous system. J Thromb Haemost (Suppl 1):151–154. https://doi.org/10.1111/j.1538-7836.2009.03438.x
Sarfati M, Fortin G, Raymond M, Susin S (2008) CD47 in the immune response: role of thrombospondin and SIRP-alpha reverse signaling. Curr Drug Targets 9:842–850
Savill J, Fadok V (2000) Corpse clearance defines the meaning of cell death. Nature 407:784–788. https://doi.org/10.1038/35037722
Savill J, Dransfield I, Gregory C, Haslett C (2002) A blast from the past: clearance of apoptotic cells regulates immune responses. Nat Rev Immunol 2:965–975
Schachtrup C, Lu P, Jones LL et al (2007) Fibrinogen inhibits neurite outgrowth via beta 3 integrin-mediated phosphorylation of the EGF receptor. Proc Natl Acad Sci U S A 104:11814–11819. https://doi.org/10.1073/pnas.0704045104
Schafer DP, Lehrman EK, Kautzman AG et al (2012) Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron 74:691–705. https://doi.org/10.1016/j.neuron.2012.03.026
Seiffert M, Brossart P, Cant C et al (2001) Signal-regulatory protein alpha (SIRPalpha) but not SIRPbeta is involved in T-cell activation, binds to CD47 with high affinity, and is expressed on immature CD34(+)CD38(−) hematopoietic cells. Blood 97:2741–2749
Seitz HM, Camenisch TD, Lemke G et al (2007) Macrophages and dendritic cells use different Axl/Mertk/Tyro3 receptors in clearance of apoptotic cells. J Immunol Baltim Md 1950 178:5635–5642
Shrivastava K, Gonzalez P, Acarin L (2012) The immune inhibitory complex CD200/CD200R is developmentally regulated in the mouse brain. J Comp Neurol 520:2657–2675. https://doi.org/10.1002/cne.23062
Singhrao SK, Neal JW, Rushmere NK et al (1999) Differential expression of individual complement regulators in the brain and choroid plexus. Lab Investig J Tech Methods Pathol 79:1247–1259
Singhrao SK, Neal JW, Rushmere NK et al (2000) Spontaneous classical pathway activation and deficiency of membrane regulators render human neurons susceptible to complement lysis. Am J Pathol 157:905–918. https://doi.org/10.1016/S0002-9440(10)64604-4
Soane L, Cho HJ, Niculescu F et al (2001) C5b-9 terminal complement complex protects oligodendrocytes from death by regulating bad through phosphatidylinositol 3-kinase/Akt pathway. J Immunol 167:2305–2311. https://doi.org/10.4049/jimmunol.167.4.2305
Sofroniew MV (2015) Astrocyte barriers to neurotoxic inflammation. Nat Rev Neurosci 16:249–263. https://doi.org/10.1038/nrn3898
Striggow F, Riek M, Breder J et al (2000) The protease thrombin is an endogenous mediator of hippocampal neuroprotection against ischemia at low concentrations but causes degeneration at high concentrations. Proc Natl Acad Sci 97:2264–2269. https://doi.org/10.1073/pnas.040552897
Sugita Y, Becerra SP, Chader GJ, Schwartz JP (1997) Pigment epithelium-derived factor (PEDF) has direct effects on the metabolism and proliferation of microglia and indirect effects on astrocytes. J Neurosci Res 49:710–718. https://doi.org/10.1002/(SICI)1097-4547(19970915)49:6<710::AID-JNR5>3.0.CO;2-A
Takahashi K, Rochford CDP, Neumann H (2005) Clearance of apoptotic neurons without inflammation by microglial triggering receptor expressed on myeloid cells-2. J Exp Med 201:647–657. https://doi.org/10.1084/jem.20041611
Takeuchi O, Akira S (2010) Pattern recognition receptors and inflammation. Cell 140:805–820. https://doi.org/10.1016/j.cell.2010.01.022
Tarozzo G, Bortolazzi S, Crochemore C et al (2003) Fractalkine protein localization and gene expression in mouse brain. J Neurosci Res 73:81–88. https://doi.org/10.1002/jnr.10645
Tenner AJ (1999) Membrane receptors for soluble defense collagens. Curr Opin Immunol 11:34–41
Thurman JM, Holers VM (2006) The central role of the alternative complement pathway in human disease. J Immunol Baltim Md 1950 176:1305–1310
Tohidpour A, Morgun AV, Boitsova EB et al (2017) Neuroinflammation and Infection: Molecular Mechanisms Associated with Dysfunction of Neurovascular Unit. Front Cell Infect Microbiol 7:276. https://doi.org/10.3389/fcimb.2017.00276
Trouw LA, Bengtsson AA, Gelderman KA et al (2007) C4b-binding protein and factor H compensate for the loss of membrane-bound complement inhibitors to protect apoptotic cells against excessive complement attack. J Biol Chem 282:28540–28548. https://doi.org/10.1074/jbc.M704354200
van Kooyk Y, Geijtenbeek TBH (2003) DC-SIGN: escape mechanism for pathogens. Nat Rev Immunol 3:697–709. https://doi.org/10.1038/nri1182
Varki A (2009) Natural ligands for CD33-related Siglecs? Glycobiology 19:810–812. https://doi.org/10.1093/glycob/cwp063
Varki A (2011) Since there are PAMPs and DAMPs, there must be SAMPs? Glycan “self-associated molecular patterns” dampen innate immunity, but pathogens can mimic them. Glycobiology 21:1121–1124
Varnum MM, Kiyota T, Ingraham KL et al (2015) The anti-inflammatory glycoprotein, CD200, restores neurogenesis and enhances amyloid phagocytosis in a mouse model of Alzheimer’s disease. Neurobiol Aging 36:2995–3007. https://doi.org/10.1016/j.neurobiolaging.2015.07.027
Vernon-Wilson EF, Kee WJ, Willis AC, et al (2000) CD47 is a ligand for rat macrophage membrane signal regulatory protein SIRP (OX41) and human SIRPalpha 1. Eur J Immunol 30:2130–2137. doi: https://doi.org/10.1002/1521-4141(2000)30:8<2130::AID-IMMU2130>3.0.CO;2–8
Vivien D, Buisson A (2000) Serine protease inhibitors: novel therapeutic targets for stroke? J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 20:755–764. https://doi.org/10.1097/00004647-200005000-00001
Walker DG, Lue L-F (2013) Understanding the neurobiology of CD200 and the CD200 receptor: a therapeutic target for controlling inflammation in human brains? Future Neurol 8. https://doi.org/10.2217/fnl.13.14
Walker DG, Dalsing-Hernandez JE, Campbell NA, Lue L-F (2009) Decreased expression of CD200 and CD200 receptor in Alzheimer’s disease: a potential mechanism leading to chronic inflammation. Exp Neurol 215:5–19. https://doi.org/10.1016/j.expneurol.2008.09.003
Walker DG, Whetzel AM, Lue L-F (2015) Expression of suppressor of cytokine signaling genes in human elderly and Alzheimer’s disease brains and human microglia. Neuroscience 302:121–137. https://doi.org/10.1016/j.neuroscience.2014.09.052
Walker DG, Lue L-F, Tang TM et al (2017) Changes in CD200 and intercellular adhesion molecule-1 (ICAM-1) levels in brains of Lewy body disorder cases are associated with amounts of Alzheimer’s pathology not α-synuclein pathology. Neurobiol Aging 54:175–186. https://doi.org/10.1016/j.neurobiolaging.2017.03.007
Walter P, Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. Science 334:1081–1086. https://doi.org/10.1126/science.1209038
Wang J, Campbell IL (2002) Cytokine signaling in the brain: putting a SOCS in it? J Neurosci Res 67:423–427. https://doi.org/10.1002/jnr.10145
Wang Y, Neumann H (2010) Alleviation of neurotoxicity by microglial human Siglec-11. J Neurosci 30:3482–3488. https://doi.org/10.1523/JNEUROSCI.3940-09.2010
Wang H, Reiser G (2003) Thrombin signaling in the brain: the role of protease-activated receptors. Biol Chem 384:193–202. https://doi.org/10.1515/BC.2003.021
Wang L, Lu Y, Deng S et al (2012) SHPS-1 deficiency induces robust neuroprotection against experimental stroke by attenuating oxidative stress. J Neurochem 122:834–843. https://doi.org/10.1111/j.1471-4159.2012.07818.x
Wang N, Liang H, Zen K (2014) Molecular mechanisms that influence the macrophage m1-m2 polarization balance. Front Immunol 5:614. https://doi.org/10.3389/fimmu.2014.00614
Wang Y, Cella M, Mallinson K et al (2015) TREM2 lipid sensing sustains the microglial response in an Alzheimer’s disease model. Cell 160:1061–1071. https://doi.org/10.1016/j.cell.2015.01.049
Watkins LM, Neal JW, Loveless S et al (2016) Complement is activated in progressive multiple sclerosis cortical grey matter lesions. J Neuroinflammation 13:161. https://doi.org/10.1186/s12974-016-0611-x
Webb M, Barclay AN (1984) Localisation of the MRC OX-2 glycoprotein on the surfaces of neurones. J Neurochem 43:1061–1067
Webster SD, Yang AJ, Margol L et al (2000) Complement component C1q modulates the phagocytosis of Abeta by microglia. Exp Neurol 161:127–138. https://doi.org/10.1006/exnr.1999.7260
Weinger JG, Omari KM, Marsden K et al (2009) Up-regulation of soluble Axl and Mer receptor tyrosine kinases negatively correlates with Gas6 in established multiple sclerosis lesions. Am J Pathol 175:283–293. https://doi.org/10.2353/ajpath.2009.080807
Weinger JG, Brosnan CF, Loudig O et al (2011) Loss of the receptor tyrosine kinase Axl leads to enhanced inflammation in the CNS and delayed removal of myelin debris during experimental autoimmune encephalomyelitis. J Neuroinflammation 8:49. https://doi.org/10.1186/1742-2094-8-49
Witting A, Müller P, Herrmann A et al (2000) Phagocytic clearance of apoptotic neurons by Microglia/Brain macrophages in vitro: involvement of lectin-, integrin-, and phosphatidylserine-mediated recognition. J Neurochem 75:1060–1070
Wolf Y, Yona S, Kim K-W, Jung S (2013) Microglia, seen from the CX3CR1 angle. Front Cell Neurosci 7:26. https://doi.org/10.3389/fncel.2013.00026
Wright GJ, Jones M, Puklavec MJ et al (2001) The unusual distribution of the neuronal/lymphoid cell surface CD200 (OX2) glycoprotein is conserved in humans. Immunology 102:173–179
Wyss-Coray T, Mucke L (2002) Inflammation in neurodegenerative disease--a double-edged sword. Neuron 35:419–432
Wyss-Coray T, Yan F, Lin AH-T et al (2002) Prominent neurodegeneration and increased plaque formation in complement-inhibited Alzheimer’s mice. Proc Natl Acad Sci U S A 99:10837–10842. https://doi.org/10.1073/pnas.162350199
Xi G, Reiser G, Keep RF (2003) The role of thrombin and thrombin receptors in ischemic, hemorrhagic and traumatic brain injury: deleterious or protective? J Neurochem 84:3–9
Yeh FL, Wang Y, Tom I et al (2016) TREM2 binds to apolipoproteins, including APOE and CLU/APOJ, and thereby facilitates uptake of amyloid-beta by microglia. Neuron 91:328–340. https://doi.org/10.1016/j.neuron.2016.06.015
Yoshimura A, Naka T, Kubo M (2007) SOCS proteins, cytokine signalling and immune regulation. Nat Rev Immunol 7:454–465. https://doi.org/10.1038/nri2093
Zhang H, Li F, Yang Y et al (2015) SIRP/CD47 signaling in neurological disorders. Brain Res 1623:74–80. https://doi.org/10.1016/j.brainres.2015.03.012
Zipfel PF, Skerka C (2009) Complement regulators and inhibitory proteins. Nat Rev Immunol 9:729–740. https://doi.org/10.1038/nri2620
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Bedoui, Y., Neal, J.W. & Gasque, P. The Neuro-Immune-Regulators (NIREGs) Promote Tissue Resilience; a Vital Component of the Host’s Defense Strategy against Neuroinflammation. J Neuroimmune Pharmacol 13, 309–329 (2018). https://doi.org/10.1007/s11481-018-9793-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-018-9793-6