Abstract
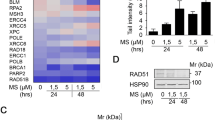
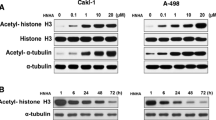
The anticancer therapies with the joint treatment of a histone deacetylase (HDAC) inhibitor and a DNA-damaging approach are actively under clinical investigations, but the underlying mechanism is unclear. Histone homeostasis is critical to genome stability, transcriptional accuracy, DNA repair process, senescence, and survival. We have previously demonstrated that the HDAC inhibitor, trichostatin A (TSA), could promote the degradation of the core histones induced by γ-radiation or the DNAalkylating agent methyl methanesulfonate (MMS) in non-cancer cells, including mouse spermatocyte and embryonic fibroblast cell lines. In this study, we found that the joint treatment by TSA and MMS induced the death of the cultured cancer cells with an additive effect, but induced degradation of the core histones synergistically in these cells. We then analyzed various combinations of other HDAC inhibitors, including suberoylanilide hydroxamic acid and valproate sodium, with MMS or other DNAdamaging agents, including etoposide and camptothecin. Most of these combined treatments induced cell death additively, but all the tested combinations induced degradation of the core histones synergistically. Meanwhile, we showed that cell cycle arrest might not be a primary consequence for the joint treatment of TSA and MMS. Given that clinic treatments of cancers jointly with an HDAC inhibitor and a DNA-damaging approach often show synergistic effects, histone degradation might more accurately underlie the synergistic effects of these joint treatments in clinic applications than other parameters, such as cell death and cell cycle arrest. Thus, our studies might suggest that the degradation of the core histones can serve as a new target for the development of cancer therapies.
Article PDF
Similar content being viewed by others
References
Altaf, M., Saksouk, N., and Côté, J. (2007). Histone modifications in response to DNA damage. Mutat Res/Fundamental Mol Mech Mutagenesis 618, 81–90.
Arnold, C., Stadler, P.F., and Prohaska, S.J. (2013). Chromatin computation: epigenetic inheritance as a pattern reconstruction problem. J Theor Biol 336, 61–74.
Blagosklonny, M.V., Robey, R., Sackett, D.L., Du, L., Traganos, F., Darzynkiewicz, Z., Fojo, T., and Bates, S.E. (2002). Histone deacetylase inhibitors all induce p21 but differentially cause tubulin acetylation, mitotic arrest, and cytotoxicity. Mol Cancer Ther 11, 937–941.
Campos, E.I., and Reinberg, D. (2009). Histones: annotating chromatin. Annu Rev Genet 43, 559–599.
Capparelli, C., Chiavarina, B., Whitaker-Menezes, D., Pestell, T.G., Pestell, R.G., Hulit, J., Andò, S., Howell, A., Martinez-Outschoorn, U.E., Sotgia, F., et al. (2012). CDK inhibitors (p16/p19/p21) induce senescence and autophagy in cancer-associated fibroblasts, “fueling” tumor growth via paracrine interactions, without an increase in neoangiogenesis. Cell Cycle 11, 3599–3610.
Carew, J.S., Giles, F.J., and Nawrocki, S.T. (2008). Histone deacetylase inhibitors: mechanisms of cell death and promise in combination cancer therapy. Cancer Lett 269, 7–17.
Chrun, E.S., Modolo, F., and Daniel, F.I. (2017). Histone modifications: a review about the presence of this epigenetic phenomenon in carcinogenesis. Pathol - Res Practice 213, 1329–1339.
Dahlin, J.L., Chen, X., Walters, M.A., and Zhang, Z. (2015). Histonemodifying enzymes, histone modifications and histone chaperones in nucleosome assembly: lessons learned from Rtt109 histone acetyltransferases. Critical Rev Biochem Mol Biol 50, 31–53.
Dokmanovic, M., and Marks, P.A. (2005). Prospects: histone deacetylase inhibitors. J Cell Biochem 96, 293–304.
Dovey, O.M., Foster, C.T., Conte, N., Edwards, S.A., Edwards, J.M., Singh, R., Vassiliou, G., Bradley, A., and Cowley, S.M. (2013). Histone deacetylase 1 and 2 are essential for normal T-cell development and genomic stability in mice. Blood 121, 1335–1344.
Downs, J.A., Allard, S., Jobin-Robitaille, O., Javaheri, A., Auger, A., Bouchard, N., Kron, S.J., Jackson, S.P., and Côté, J. (2004). Binding of chromatin-modifying activities to phosphorylated histone H2A at DNA damage sites. Mol Cell 16, 979–990.
Eckschlager, T., Plch, J., Stiborova, M., and Hrabeta, J. (2017). Histone deacetylase inhibitors as anticancer drugs. IJMS 18, 1414.
Feng, W., Cai, D., Zhang, B., Lou, G., and Zou, X. (2015). Combination of HDAC inhibitor TSA and silibinin induces cell cycle arrest and apoptosis by targeting survivin and cyclinB1/Cdk1 in pancreatic cancer cells. Biomed PharmacoTher 74, 257–264.
Ge, Z., Nair, D., Guan, X., Rastogi, N., Freitas, M.A., and Parthun, M.R. (2013). Sites of acetylation on newly synthesized histone H4 are required for chromatin assembly and DNA damage response signaling. Mol Cell Biol 33, 3286–3298.
Haberland, M., Montgomery, R.L., and Olson, E.N. (2009). The many roles of histone deacetylases in development and physiology: implications for disease and therapy. Nat Rev Genet 10, 32–42.
Khor, B., Bredemeyer, A.L., Huang, C.Y., Turnbull, I.R., Evans, R., Maggi, L.B., White, J.M., Walker, L.M., Carnes, K., Hess, R.A., et al. (2006). Proteasome activator PA200 is required for normal spermatogenesis. Mol Cell Biol 26, 2999–3007.
Kumar, V., Ara, G., Afzal, M., and Siddique, Y.H. (2011). Effect of methyl methanesulfonate on hsp70 expression and tissue damage in the third instar larvae of transgenic Drosophila melanogaster (hsp70-lacZ) Bg9. Interdisciplinary Toxicol 4, 159–165.
Lundin, C., North, M., Erixon, K., Walters, K., Jenssen, D., Goldman, A.S. H., and Helleday, T. (2005). Methyl methanesulfonate (MMS) produces heat-labile DNA damage but no detectable in vivo DNA double-strand breaks. Nucleic Acids Res 33, 3799–3811.
Meng, J., Zhang, H.H., Zhou, C.X., Li, C., Zhang, F., and Mei, Q.B. (2012) The histone deacetylase inhibitor trichostatin A induces cell cycle arrest and apoptosis in colorectal cancer cells via p53-dependent and -independent pathways. Oncol Rep.
Montecucco, A., Zanetta, F., and Biamonti, G. (2015). Molecular mechanisms of etoposide. EXCLI Journal 14, 95–108.
Murr, R., Loizou, J.I., Yang, Y.G., Cuenin, C., Li, H., Wang, Z.Q., and Herceg, Z. (2006). Histone acetylation by Trrap-Tip60 modulates loading of repair proteins and repair of DNA double-strand breaks. Nat Cell Biol 8, 91–99.
Pommier, Y., Leo, E., Zhang, H.L., and Marchand, C. (2010). DNA topoisomerases and their poisoning by anticancer and antibacterial drugs. Chem Biol 17, 421–433.
Qian, M.X., Pang, Y., Liu, C.H., Haratake, K., Du, B.Y., Ji, D.Y., Wang, G. F., Zhu, Q.Q., Song, W., Yu, Y., et al. (2013). Acetylation-mediated proteasomal degradation of core histones during DNA repair and spermatogenesis. Cell 153, 1012–1024.
Reinke, H., and Hörz, W. (2003). Histones are first hyperacetylated and then lose contact with the activated PHO5 promoter. Mol Cell 11, 1599–1607.
Robey, R.W., Chakraborty, A.R., Basseville, A., Luchenko, V., Bahr, J., Zhan, Z., and Bates, S.E. (2011). Histone deacetylase inhibitors: emerging mechanisms of resistance. Mol Pharm 8, 2021–2031.
Shabason, J.E., Tofilon, P.J., and Camphausen, K. (2011). Grand rounds at the National Institutes of Health: HDAC inhibitors as radiation modifiers, from bench to clinic. J Cellular Mol Med 15, 2735–2744.
Stiborova, M., Eckschlager, T., Poljakova, J., Hrabeta, J., Adam, V., Kizek, R., and Frei, E. (2012). The synergistic effects of DNA-targeted chemotherapeutics and histone deacetylase inhibitors as therapeutic strategies for cancer treatment. CMC 19, 4218–4238.
Suraweera, A., O’Byrne, K.J., and Richard, D.J. (2018). Combination therapy with histone deacetylase inhibitors (HDACi) for the treatment of cancer: achieving the full therapeutic potential of HDACi. Front Oncol 8, 92.
Turinetto, V., and Giachino, C. (2015). Multiple facets of histone variant H2AX: a DNA double-strand-break marker with several biological functions. Nucleic Acids Res 43, 2489–2498.
Ustrell, V., Hoffman, L., Pratt, G., and Rechsteiner, M. (2002). PA200, a nuclear proteasome activator involved in DNA repair. EMBO J 21, 3516–3525.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (31530014, 91319303 & 31600626).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yin, Y., Zhu, Q., Jiang, T. et al. Targeting histones for degradation in cancer cells as a novel strategy in cancer treatment. Sci. China Life Sci. 62, 1078–1086 (2019). https://doi.org/10.1007/s11427-018-9391-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-018-9391-7