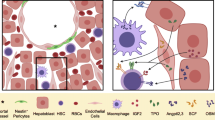
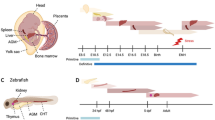
Abstract
Fetal liver (FL) is an intricate and highly vascularized hematopoietic organ, which can support the extensive expansion of hematopoietic stem cells (HSCs) without loss of stemness, as well as of the downstream lineages of HSCs. This powerful function of FL largely benefits from the niche (or microenvironment), which provides a residence for HSC expansion. Numerous studies have demonstrated that the FL niche consists of heterogeneous cell populations that associate with HSCs spatially and regulate HSCs functionally. At the molecular level, a complex of cell extrinsic and intrinsic signaling network within the FL niche cells maintains HSC expansion. Here, we summarize recent studies on the analysis of the FL HSCs and their niche, and specifically on the molecular regulatory network for HSC expansion. Based on these studies, we hypothesize a strategy to obtain a large number of functional HSCs via 3D reconstruction of FL organoid ex vivo for clinical treatment in the future.
Similar content being viewed by others
References
Acar, M., Kocherlakota, K.S., Murphy, M.M., Peyer, J.G., Oguro, H., Inra, C.N., Jaiyeola, C., Zhao, Z., Luby-Phelps, K., and Morrison, S.J. (2015). Deep imaging of bone marrow shows non-dividing stem cells are mainly perisinusoidal. Nature 526, 126–130
Arora, N., Wenzel, P.L., McKinney-Freeman, S.L., Ross, S.J., Kim, P.G., Chou, S.S., Yoshimoto, M., Yoder, M.C., and Daley, G.Q. (2014). Effect of developmental stage of HSC and recipient on transplant outcomes. Dev Cell 29, 621–628
Badylak, S.F. (2002). The extracellular matrix as a scaffold for tissue reconstruction. Semin Cell Dev Biol 13, 377–383
Baptista, P.M., Siddiqui, M.M., Lozier, G., Rodriguez, S.R., Atala, A., and Soker, S. (2011). The use of whole organ decellularization for the generation of a vascularized liver organoid. Hepatology 53, 604–617
Baumann, C.I., Bailey, A.S., Li, W., Ferkowicz, M.J., Yoder, M.C., and Fleming, W.H. (2004). PECAM-1 is expressed on hematopoietic stem cells throughout ontogeny and identifies a population of erythroid progenitors. Blood 104, 1010–1016
Bertrand, J.Y., Chi, N.C., Santoso, B., Teng, S., Stainier, D.Y.R., and Traver, D. (2010). Haematopoietic stem cells derive directly from aortic endothelium during development. Nature 464, 108–111
Boisset, J.C., van Cappellen, W., Andrieu-Soler, C., Galjart, N., Dzierzak, E., and Robin, C. (2010). In vivo imaging of haematopoietic cells emerging from the mouse aortic endothelium. Nature 464, 116–120
Boulais, P.E., and Frenette, P.S. (2015). Making sense of hematopoietic stem cell niches. Blood 125, 2621–2629
Chen, J., Suo, S., Tam, P.P., Han, J.D.J., Peng, G., and Jing, N. (2017). Spatial transcriptomic analysis of cryosectioned tissue samples with Geo-seq. Nat Protoc 12, 566–580
Chen, Y., Haviernik, P., Bunting, K.D., and Yang, Y.C. (2007). Cited2 is required for normal hematopoiesis in the murine fetal liver. Blood 110, 2889–2898
Chou, S., and Lodish, H.F. (2010). Fetal liver hepatic progenitors are supportive stromal cells for hematopoietic stem cells. Proc Natl Acad Sci USA 107, 7799–7804
Coskun, S., and Hirschi, K.K. (2010). Establishment and regulation of the HSC niche: roles of osteoblastic and vascular compartments. Birth Defects Res Part C Embryo Today Rev 90, 229–242
Crane, G.M., Jeffery, E., and Morrison, S.J. (2017). Adult haematopoietic stem cell niches. Nat Rev Immunol 17, 573–590.
de Bruijn, M.F.T.R., Speck, N.A., Peeters, M.C.E., and Dzierzak, E. (2000). Definitive hematopoietic stem cells first develop within the major arterial regions of the mouse embryo. EMBO J 19, 2465–2474
Ding, L., Saunders, T.L., Enikolopov, G., and Morrison, S.J. (2012). Endothelial and perivascular cells maintain haematopoietic stem cells. Nature 481, 457–462
Dzierzak, E., and Robin, C. (2010). Placenta as a source of hematopoietic stem cells. Trends Mol Med 16, 361–367
Edling, C.E., and Hallberg, B. (2007). c-Kit—A hematopoietic cell essential receptor tyrosine kinase. Int J Biochem Cell Biol 39, 1995–1998
Ema, H., and Nakauchi, H. (2000). Expansion of hematopoietic stem cells in the developing liver of a mouse embryo. Blood 95, 2284–2288
Felfly, H., and Haddad, G.G. (2014). Hematopoietic stem cells: potential new applications for translational medicine. J Stem Cells 9, 163–197
Flanagan, J.G., and Leder, P. (1990). The kit ligand: A cell surface molecule altered in steel mutant fibroblasts. Cell 63, 185–194
Gao, X., Xu, C., Asada, N., and Frenette, P.S. (2018). The hematopoietic stem cell niche: from embryo to adult. Development 145, dev139691
Gekas, C., Dieterlen-Lièvre, F., Orkin, S.H., and Mikkola, H.K.A. (2005). The placenta is a niche for hematopoietic stem cells. Dev Cell 8, 365–375
Gerhardt, D.M., Pajcini, K.V., D’altri, T., Tu, L., Jain, R., Xu, L., Chen, M. J., Rentschler, S., Shestova, O., Wertheim, G.B., et al. (2014). The Notch1 transcriptional activation domain is required for development and reveals a novel role for Notch1 signaling in fetal hematopoietic stem cells. Genes Dev 28, 576–593
Grün, D., Kester, L., and van Oudenaarden, A. (2014). Validation of noise models for single-cell transcriptomics. Nat Methods 11, 637–640
Grün, D., and van Oudenaarden, A. (2015). Design and analysis of singlecell sequencing experiments. Cell 163, 799–810
Hackney, J.A., Charbord, P., Brunk, B.P., Stoeckert, C.J., Lemischka, I.R., and Moore, K.A. (2002). A molecular profile of a hematopoietic stem cell niche. Proc Natl Acad Sci USA 99, 13061–13066
Han, X., Wang, R., Zhou, Y., Fei, L., Sun, H., Lai, S., Saadatpour, A., Zhou, Z., Chen, H., Ye, F., et al. (2018). Mapping the mouse cell atlas by microwell-Seq. Cell 172, 1091–1107.e17
Harrison, D.E., Zhong, R.K., Jordan, C.T., Lemischka, I.R., and Astle, C. M. (1997). Relative to adult marrow, fetal liver repopulates nearly five times more effectively long-term than short-term. Exp Hematol 25, 293–297
He, Q., Gao, S., Lv, J., Li, W., and Liu, F. (2017). BLOS2 maintains hematopoietic stem cells in the fetal liver via repressing Notch signaling. Exp Hematol 51, 1–6.e2
Hoeffel, G., Chen, J., Lavin, Y., Low, D., Almeida, F.F., See, P., Beaudin, A.E., Lum, J., Low, I., Forsberg, E.C., et al. (2015). C-Myb+ erythromyeloid progenitor-derived fetal monocytes give rise to adult tissueresident macrophages. Immunity 42, 665–678
Hsu, H.C., Ema, H., Osawa, M., Nakamura, Y., Suda, T., and Nakauchi, H. (2000). Hematopoietic stem cells express Tie-2 receptor in the murine fetal liver. Blood 96, 3757–3762
Ikuta, K., Kina, T., MacNeil, I., Uchida, N., Peault, B., Chien, Y., and Weissman, I.L. (1990). A developmental switch in thymic lymphocyte maturation potential occurs at the level of hematopoietic stem cells. Cell 62, 863–874
Ikuta, K., and Weissman, I.L. (1992). Evidence that hematopoietic stem cells express mouse c-kit but do not depend on steel factor for their generation.. Proc Natl Acad Sci USA 89, 1502–1506
Iwasaki, H., Arai, F., Kubota, Y., Dahl, M., and Suda, T. (2010). Endothelial protein C receptor-expressing hematopoietic stem cells reside in the perisinusoidal niche in fetal liver. Blood 116, 544–553
Jassinskaja, M., Johansson, E., Kristiansen, T.A., Åkerstrand, H., Sjöholm, K., Hauri, S., Malmström, J., Yuan, J., and Hansson, J. (2017). Comprehensive proteomic characterization of ontogenic changes in hematopoietic stem and progenitor cells. Cell Rep 21, 3285–3297
Jordan, C.T., McKearn, J.P., and Lemischka, I.R. (1990). Cellular and developmental properties of fetal hematopoietic stem cells. Cell 61, 953–963
Khan, J.A., Mendelson, A., Kunisaki, Y., Birbrair, A., Kou, Y., Arnal-Estapé, A., Pinho, S., Ciero, P., Nakahara, F., Ma’ayan, A., et al. (2016). Fetal liver hematopoietic stem cell niches associate with portal vessels. Science 351, 176–180
Kieusseian, A., Brunet de la Grange, P., Burlen-Defranoux, O., Godin, I., and Cumano, A. (2012). Immature hematopoietic stem cells undergo maturation in the fetal liver. Development 139, 3521–3530
Kim, I., He, S., Yilmaz, O.H., Kiel, M.J., and Morrison, S.J. (2006). Enhanced purification of fetal liver hematopoietic stem cells using SLAM family receptors. Blood 108, 737–744
Kim, I., Yilmaz, O.H., and Morrison, S.J. (2005). CD144 (VE-cadherin) is transiently expressed by fetal liver hematopoietic stem cells. Blood 106, 903–905
Kissa, K., and Herbomel, P. (2010). Blood stem cells emerge from aortic endothelium by a novel type of cell transition. Nature 464, 112–115
Krosl, J., Mamo, A., Chagraoui, J., Wilhelm, B.T., Girard, S., Louis, I., Lessard, J., Perreault, C., and Sauvageau, G. (2010). A mutant allele of the Swi/Snf member BAF250a determines the pool size of fetal liver hemopoietic stem cell populations. Blood 116, 1678–1684
Kubanek, B., Rencricca, N., Porcellini, A., Howard, D., and Stohlman, F. (1969). The pattern of recovery of erythropoiesis in heavily irradiated mice receiving transplants of fetal liver. Exp Biol Med 131, 831–834
Kunisaki, Y., Bruns, I., Scheiermann, C., Ahmed, J., Pinho, S., Zhang, D., Mizoguchi, T., Wei, Q., Lucas, D., Ito, K., et al. (2013). Arteriolar niches maintain haematopoietic stem cell quiescence. Nature 502, 637–643
Li, C.L., and Johnson, G.R. (1994). Stem cell factor enhances the survival but not the self-renewal of murine hematopoietic long-term repopulating cells. Blood 84, 408–414
Li, T., Huang, J., Jiang, Y., Zeng, Y., He, F., Zhang, M.Q., Han, Z., and Zhang, X. (2009). Multi-stage analysis of gene expression and transcription regulation in C57/B6 mouse liver development. Genomics 93, 235–242
Li, Z., Lan, Y., He, W., Chen, D., Wang, J., Zhou, F., Wang, Y., Sun, H., Chen, X., Xu, C., et al. (2012). Mouse embryonic head as a site for hematopoietic stem cell development. Cell Stem Cell 11, 663–675
Lin, M.I., Price, E.N., Boatman, S., Hagedorn, E.J., Trompouki, E., Satishchandran, S., Carspecken, C.W., Uong, A., DiBiase, A., Yang, S., et al. (2015). Angiopoietin-like proteins stimulate HSPC development through interaction with notch receptor signaling. eLife 4, e05544
Macosko, E.Z., Basu, A., Satija, R., Nemesh, J., Shekhar, K., Goldman, M., Tirosh, I., Bialas, A.R., Kamitaki, N., Martersteck, E.M., et al. (2015). Highly parallel genome-wide expression profiling of individual cells using nanoliter droplets. Cell 161, 1202–1214
Manesia, J.K., Xu, Z., Broekaert, D., Boon, R., van Vliet, A., Eelen, G., Vanwelden, T., Stegen, S., Van Gastel, N., Pascual-Montano, A., et al. (2015). Highly proliferative primitive fetal liver hematopoietic stem cells are fueled by oxidative metabolic pathways. Stem Cell Res 15, 715–721
McKinney-Freeman, S., Cahan, P., Li, H., Lacadie, S.A., Huang, H.T., Curran, M., Loewer, S., Naveiras, O., Kathrein, K.L., Konantz, M., et al. (2012). The transcriptional landscape of hematopoietic stem cell ontogeny. Cell Stem Cell 11, 701–714
Medvinsky, A., and Dzierzak, E. (1996). Definitive hematopoiesis is autonomously initiated by the AGM region. Cell 86, 897–906
Metcalf, D. (2008). Hematopoietic cytokines. Blood 111, 485–491
Mikkola, H.K.A., and Orkin, S.H. (2006). The journey of developing hematopoietic stem cells. Development 133, 3733–3744
Mochizuki-Kashio, M., Mishima, Y., Miyagi, S., Negishi, M., Saraya, A., Konuma, T., Shinga, J., Koseki, H., and Iwama, A. (2011). Dependency on the polycomb gene Ezh2 distinguishes fetal from adult hematopoietic stem cells. Blood 118, 6553–6561
Morrison, S.J., Hemmati, H.D., Wandycz, A.M., and Weissman, I.L. (1995). The purification and characterization of fetal liver hematopoietic stem cells. Proc Natl Acad Sci USA 92, 10302–10306
Morrison, S.J., and Scadden, D.T. (2014). The bone marrow niche for haematopoietic stem cells. Nature 505, 327–334
Munugalavadla, V., Dore, L.C., Tan, B.L., Hong, L., Vishnu, M., Weiss, M. J., and Kapur, R. (2005). Repression of c-kit and its downstream substrates by GATA-1 inhibits cell proliferation during erythroid maturation. Mol Cellular Biol 25, 6747–6759
Orkin, S.H., and Zon, L.I. (2008). Hematopoiesis: an evolving paradigm for stem cell biology. Cell 132, 631–644
Peng, G., Suo, S., Chen, J., Chen, W., Liu, C., Yu, F., Wang, R., Chen, S., Sun, N., Cui, G., et al. (2016). Spatial transcriptome for the molecular annotation of lineage fates and cell identity in Mid-gastrula mouse embryo. Dev Cell 36, 681–697
Porayette, P., and Paulson, R.F. (2008). BMP4/Smad5 dependent stress erythropoiesis is required for the expansion of erythroid progenitors during fetal development. Dev Biol 317, 24–35
Rybtsov, S., Ivanovs, A., Zhao, S., and Medvinsky, A. (2016). Concealed expansion of immature precursors underpins acute burst of adult HSC activity in foetal liver. Development 143, 1284–1289
Swain, A., Inoue, T., Tan, K.S., Nakanishi, Y., and Sugiyama, D. (2014). Intrinsic and extrinsic regulation of mammalian hematopoiesis in the fetal liver. Histol Histopathol 29, 1077–1082
Szilvassy, S.J., Meyerrose, T.E., Ragland, P.L., and Grimes, B. (2001). Differential homing and engraftment properties of hematopoietic progenitor cells from murine bone marrow, mobilized peripheral blood, and fetal liver. Blood 98, 2108–2115
Szpinda, M., Paruszewska-Achtel, M., Woźniak, A., Badura, M., Mila-Kierzenkowska, C., and Wiśniewski, M. (2015). Three-dimensional growth dynamics of the liver in the human fetus. Surg Radiol Anat 37, 439–448
Takebe, T., Sekine, K., Suzuki, Y., Enomura, M., Tanaka, S., Ueno, Y., Zheng, Y.W., and Taniguchi, H. (2012). Self-organization of human hepatic organoid by recapitulating organogenesis in vitro. Transplant Proc 44, 1018–1020
Tang, Y., Peitzsch, C., Charoudeh, H.N., Cheng, M., Chaves, P., Jacobsen, S.E.W., and Sitnicka, E. (2012). Emergence of NK-cell progenitors and functionally competent NK-cell lineage subsets in the early mouse embryo. Blood 120, 63–75
Treutlein, B., Brownfield, D.G., Wu, A.R., Neff, N.F., Mantalas, G.L., Espinoza, F.H., Desai, T.J., Krasnow, M.A., and Quake, S.R. (2014). Reconstructing lineage hierarchies of the distal lung epithelium using single-cell RNA-seq. Nature 509, 371–375
Williams, D.E., Eisenman, J., Baird, A., Rauch, C., Van Ness, K., March, C.J., Park, L.S., Martin, U., Mochizukl, D.Y., Boswell, H.S., et al. (1990). Identification of a ligand for the c-kit proto-oncogene. Cell 63, 167–174
Xie, Y., Li, Y., and Kong, Y. (2014). OPN induces FoxM1 expression and localization through ERK 1/2 AKT, and p38 signaling pathway in HEC-1A cells. Int J Mol Sci 15, 23345–23358
You, L., Li, L., Zou, J., Yan, K., Belle, J., Nijnik, A., Wang, E., and Yang, X.J. (2016). BRPF1 is essential for development of fetal hematopoietic stem cells. J Clin Investig 126, 3247–3262
Zayas, J., Spassov, D.S., Nachtman, R.G., and Jurecic, R. (2008). Murine hematopoietic stem cells and multipotent progenitors express truncated intracellular form of c-kit receptor. Stem Cells Dev 17, 343–354
Zeigler, F.C., Bennett, B.D., Jordan, C.T., Spencer, S.D., Baumhueter, S., Carroll, K.J., Hooley, J., Bauer, K., and Matthews, W. (1994). Cellular and molecular characterization of the role of the flk-2/flt-3 receptor tyrosine kinase in hematopoietic stem cells. Blood 84, 2422–2430
Zhang, C.C., Kaba, M., Ge, G., Xie, K., Tong, W., Hug, C., and Lodish, H. F. (2006). Angiopoietin-like proteins stimulate ex vivo expansion of hematopoietic stem cells. Nat Med 12, 240–245
Zhang, C.C., and Lodish, H.F. (2004). Insulin-like growth factor 2 expressed in a novel fetal liver cell population is a growth factor for hematopoietic stem cells. Blood 103, 2513–2521
Zhao, X., Gao, F., Zhang, X., Wang, Y., Xu, L., Liu, K., Zhao, X., Chang, Y., Wei, H., Chen, H., et al. (2016). Improved clinical outcomes of rhGCSF-mobilized blood and marrow haploidentical transplantation compared to propensity score-matched rhG-CSF-primed peripheral blood stem cell haploidentical transplantation: a multicenter study. Sci China Life Sci 59, 1139–1148
Zhao, Y., Zhou, J., Liu, D., Dong, F., Cheng, H., Wang, W., Pang, Y., Wang, Y., Mu, X., Ni, Y., et al. (2015). ATF4 plays a pivotal role in the development of functional hematopoietic stem cells in mouse fetal liver. Blood 126, 2383–2391
Zheng, J., Umikawa, M., Cui, C., Li, J., Chen, X., Zhang, C., Huynh, H.D., Hyunh, H., Kang, X., Silvany, R., et al. (2012). Inhibitory receptors bind ANGPTLs and support blood stem cells and leukaemia development. Nature 485, 656–660
Zhou, B.O., Yu, H., Yue, R., Zhao, Z., Rios, J.J., Naveiras, O., and Morrison, S.J. (2017). Bone marrow adipocytes promote the regeneration of stem cells and haematopoiesis by secreting SCF. Nat Cell Biol 19, 891–903.
Acknowledgements
We thank lab members for helpful discussions and critical reading of the paper. This work was supported by the National Natural Science Foundation of China (81530004, 31425016), the Ministry of Science and Technology of China (2016YFA0100500) and the Strategic Priority Research Program of the Chinese Academy of Sciences, China (XDA16010104).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gao, S., Liu, F. Fetal liver: an ideal niche for hematopoietic stem cell expansion. Sci. China Life Sci. 61, 885–892 (2018). https://doi.org/10.1007/s11427-018-9313-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-018-9313-4