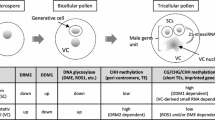
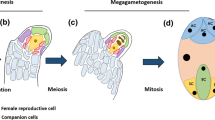
Abstract
Recent studies have indicated that a special type of small noncoding RNAs, phased small-interfering RNAs (phasiRNAs) play crucial roles in many cellular processes of plant development. PhasiRNAs are generated from long RNA precursors at intervals of 21 or 24 nt in plants, and they are produced from both protein-coding gene and long noncoding RNA genes. Different from those in eudicots, grass phasiRNAs include a special class of small RNAs that are specifically expressed in reproductive organs. These grass phasiRNAs are associated with gametogenesis, especially with anther development and male fertility. In this review, we summarized current knowledge on these small noncoding RNAs in male germ cells and their possible biological functions and mechanisms in grass species.
Similar content being viewed by others
References
Axtell, M.J. (2013). Classification and comparison of small RNAs from plants. Annu Rev Plant Biol 64, 137–159.
Barakat, A., Sriram, A., Park, J., Zhebentyayeva, T., Main, D., and Abbott, A. (2012). Genome wide identification of chilling responsive microRNAs in Prunus persica. BMC Genomics 13, 481.
Bohmert, K., Camus, I., Bellini, C., Bouchez, D., Caboche, M., and Benning, C. (1998). AGO1 defines a novel locus of Arabidopsis controlling leaf development. EMBO J 17, 170–180.
Borges, F., and Martienssen, R.A. (2015). The expanding world of small RNAs in plants. Nat Rev Mol Cell Biol 16, 727–741.
Chen, L., and Liu, Y.G. (2014). Male sterility and fertility restoration in crops. Annu Rev Plant Biol 65, 579–606.
Cui, J., You, C., and Chen, X. (2017). The evolution of microRNAs in plants. Curr Opin Plant Biol 35, 61–67.
Dukowic-Schulze, S., Sundararajan, A., Ramaraj, T., Kianian, S., Pawlowski, W.P., Mudge, J., and Chen, C. (2016). Novel meiotic miRNAs and indications for a role of phasiRNAs in meiosis. Front Plant Sci 7, 762.
Fan, Y., Yang, J., Mathioni, S.M., Yu, J., Shen, J., Yang, X., Wang, L., Zhang, Q., Cai, Z., Xu, C., Li, X., Xiao, J., Meyers, B.C., and Zhang, Q. (2016). PMS1T, producing phased small-interfering RNAs, regulates photoperiod-sensitive male sterility in rice. Proc Natl Acad Sci USA 113, 15144–15149.
Fei, Q., Xia, R., and Meyers, B.C. (2013). Phased, secondary, small interfering RNAs in posttranscriptional regulatory networks. Plant Cell 25, 2400–2415.
Fei, Q., Yang, L., Liang, W., Zhang, D., and Meyers, B.C. (2016). Dynamic changes of small RNAs in rice spikelet development reveal specialized reproductive phasiRNA pathways. J Exp Bot 67, 6037–6049.
Fu, Q., and Wang, P.J. (2014). Mammalian piRNAs. Spermatogenesis 4, e27889.
Gao, Z., Luo, X., Shi, T., Cai, B., Zhang, Z., Cheng, Z., and Zhuang, W. (2012). Identification and validation of potential conserved microRNAs and their targets in peach (Prunus persica). Mol Cells 34, 239–249.
Gou, L.T., Dai, P., Yang, J.H., Xue, Y., Hu, Y.P., Zhou, Y., Kang, J.Y., Wang, X., Li, H., Hua, M.M., Zhao, S., Hu, S.D., Wu, L.G., Shi, H.J., Li, Y., Fu, X.D., Qu, L.H., Wang, E.D., and Liu, M.F. (2014). Pachytene piRNAs instruct massive mRNA elimination during late spermiogenesis. Cell Res 24, 680–700.
Han, B.W., Wang, W., Li, C., Weng, Z., and Zamore, P.D. (2015). piRNAguided transposon cleavage initiates Zucchini-dependent, phased piRNA production. Science 348, 817–821.
He, H., Yang, T.Y., Wu, W.Y., and Zheng, B.L. (2015). Small RNAs in pollen. Sci China Life Sci 58, 246–252.
Itoh, J.I., Kitano, H., Matsuoka, M., and Nagato, Y. (2000). SHOOT ORGANIZATION genes regulate shoot apical meristem organization and the pattern of leaf primordium initiation in rice. Plant Cell 12, 2161–2174.
Jeong, D.H., Schmidt, S.A., Rymarquis, L.A., Park, S., Ganssmann, M., German, M.A., Accerbi, M., Zhai, J., Fahlgren, N., Fox, S.E., Garvin, D.F., Mockler, T.C., Carrington, J.C., Meyers, B.C., and Green, P.J. (2013). Parallel analysis of RNA ends enhances global investigation of microRNAs and target RNAs of Brachypodium distachyon. Genome Biol 14, R145.
Jia, J., Zhao, S., Kong, X., Li, Y., Zhao, G., He, W., Appels, R., Pfeifer, M., Tao, Y., Zhang, X., Jing, R., Zhang, C., Ma, Y., Gao, L., Gao, C., Spannagl, M., Mayer, K.F.X., Li, D., Pan, S., Zheng, F., Hu, Q., Xia, X., Li, J., Liang, Q., Chen, J., Wicker, T., Gou, C., Kuang, H., He, G., Luo, Y., Keller, B., Xia, Q., Lu, P., Wang, J., Zou, H., Zhang, R., Xu, J., Gao, J., Middleton, C., Quan, Z., Liu, G., Wang, J., Wang, J., Yang, H., Liu, X., He, Z., Mao, L., and Wang, J. (2013). Aegilops tauschii draft genome sequence reveals a gene repertoire for wheat adaptation. Nature 496, 91–95.
Johnson, C., Kasprzewska, A., Tennessen, K., Fernandes, J., Nan, G.L., Walbot, V., Sundaresan, V., Vance, V., and Bowman, L.H. (2009). Clusters and superclusters of phased small RNAs in the developing inflorescence of rice. Genome Res 19, 1429–1440.
Kantar, M., Lucas, S.J., and Budak, H. (2011). miRNA expression patterns of Triticum dicoccoides in response to shock drought stress. Planta 233, 471–484.
Kapoor, M., Arora, R., Lama, T., Nijhawan, A., Khurana, J.P., Tyagi, A.K., and Kapoor, S. (2008). Genome-wide identification, organization and phylogenetic analysis of Dicer-like, Argonaute and RNA-dependent RNA Polymerase gene families and their expression analysis during reproductive development and stress in rice. BMC Genomics 9, 451.
Kelliher, T., and Walbot, V. (2014). Maize germinal cell initials accommodate hypoxia and precociously express meiotic genes. Plant J 77, 639–652.
Komiya, R. (2017). Biogenesis of diverse plant phasiRNAs involves an miRNA-trigger and Dicer-processing. J Plant Res 130, 17–23.
Komiya, R., Ohyanagi, H., Niihama, M., Watanabe, T., Nakano, M., Kurata, N., and Nonomura, K.I. (2014). Rice germline-specific Argonaute MEL1 protein binds to phasiRNAs generated from more than 700 lincRNAs. Plant J 78, 385–397.
Kurihara, Y., and Watanabe, Y. (2004). Arabidopsis micro-RNA biogenesis through Dicer-like 1 protein functions. Proc Natl Acad Sci USA 101, 12753–12758.
Li, X., Shahid, M.Q., Xia, J., Lu, Z., Fang, N., Wang, L., Wu, J., Chen, Z., and Liu, X. (2017). Analysis of small RNAs revealed differential expressions during pollen and embryo sac development in autotetraploid rice. BMC Genomics 18, 129.
Liu, B., Chen, Z., Song, X., Liu, C., Cui, X., Zhao, X., Fang, J., Xu, W., Zhang, H., Wang, X., Chu, C., Deng, X., Xue, Y., and Cao, X. (2007). Oryza sativa Dicer-like4 reveals a key role for small interfering RNA silencing in plant development. Plant Cell 19, 2705–2718.
Liu, J., and Qu, L.J. (2008). Meiotic and mitotic cell cycle mutants involved in gametophyte development in Arabidopsis. Mol Plant 1, 564–574.
Liu, Y., Wang, Y., Zhu, Q.H., and Fan, L. (2013). Identification of phasiRNAs in wild rice (Oryza rufipogon). Plant Signal Behav 8, e25079.
Liu, X., Hao, L., Li, D., Zhu, L., and Hu, S. (2015). Long non-coding RNAs and their biological roles in plants. Genomics Proteomics Bioinformatics 13, 137–147.
Mohn, F., Handler, D., and Brennecke, J. (2015). piRNA-guided slicing specifies transcripts for Zucchini-dependent, phased piRNA biogenesis. Science 348, 812–817.
Nagasaki, H., Itoh, J., Hayashi, K., Hibara, K., Satoh-Nagasawa, N., Nosaka, M., Mukouhata, M., Ashikari, M., Kitano, H., Matsuoka, M., Nagato, Y., and Sato, Y. (2007). The small interfering RNA production pathway is required for shoot meristem initiation in rice. Proc Natl Acad Sci USA 104, 14867–14871.
Nonomura, K.I., Morohoshi, A., Nakano, M., Eiguchi, M., Miyao, A., Hirochika, H., and Kurata, N. (2007). A germ cell specific gene of the ARGONAUTE family is essential for the progression of premeiotic mitosis and meiosis during sporogenesis in rice. Plant Cell 19, 2583–2594.
Reinhart, B.J., Weinstein, E.G., Rhoades, M.W., Bartel, B., and Bartel, D.P. (2002). MicroRNAs in plants. Genes Dev 16, 1616–1626.
Satoh, N., Hong, S., Nishimura, A., Matsuoka, M., Kitano, H., and Nagato, Y. (1999). Initiation of shoot apical meristem in rice: characterization of four SHOOTLESS genes. Development 126, 3629–3636.
Shi, M. (1985). The discovery and study of the photosensitive recessive male-sterile rice (Oryza sativa l. Subsp. Japonica). Scient Agr Sin, 2, 006.
Song, X., Li, P., Zhai, J., Zhou, M., Ma, L., Liu, B., Jeong, D.H., Nakano, M., Cao, S., Liu, C., Chu, C., Wang, X.J., Green, P.J., Meyers, B.C., and Cao, X. (2012a). Roles of DCL4 and DCL3b in rice phased small RNA biogenesis. Plant J 69, 462–474.
Song, X., Wang, D., Ma, L., Chen, Z., Li, P., Cui, X., Liu, C., Cao, S., Chu, C., Tao, Y., and Cao, X. (2012b). Rice RNA-dependent RNA polymerase 6 acts in small RNA biogenesis and spikelet development. Plant J 71, 378–389.
Sosa-Valencia, G., Palomar, M., Covarrubias, A. A., and Reyes, J. L. (2017). The legume miR1514a modulates a NAC transcription factor transcript to trigger phasiRNA formation in response to drought. J Exp Bot 68, 2013–2026.
Su, C., Yang, X., Gao, S., Tang, Y., Zhao, C., and Li, L. (2014). Identification and characterization of a subset of microRNAs in wheat (Triticum aestivum L.). Genomics 103, 298–307.
Ta, K. N., Sabot, F., Adam, H., Vigouroux, Y., De Mita, S., Ghesquiere, A., Do, N. V., Gantet, P., and Jouannic, S. (2016). miR2118-triggered phased siRNAs are differentially expressed during the panicle development of wild and domesticated African rice species. Rice (N Y) 9, 10.
Walbot, V., and Egger, R.L. (2016). Pre-meiotic anther development: cell fate specification and differentiation. Annu Rev Plant Biol 67, 365–395.
Wang, L., and Wang, J.W. (2015). Coding function for non-coding RNA in plants—insights from miRNA encoded peptide (miPEP). Sci China Life Sci 58, 503–505.
Wu, J., Yang, Z., Wang, Y., Zheng, L., Ye, R., Ji, Y., Zhao S., Ji, S., Liu, R., Xu, L., Zheng, H., Zhou, Y., Zhang, X., Cao, X., Xie, L., Wu, Z., Qi, Y., Li, Y. (2015) Viral-inducible Argonaute18 confers broad-spectrum virus resistance in rice by sequestering a host microRNA. Elife 4, e05733.
Wu, L., Zhang, Q., Zhou, H., Ni, F., Wu, X., and Qi, Y. (2009). Rice microRNA effector complexes and targets. Plant Cell 21, 3421–3435.
Xie, Z., Allen, E., Wilken, A., and Carrington, J.C. (2005). DICER-LIKE 4 functions in trans-acting small interfering RNA biogenesis and vegetative phase change in Arabidopsis thaliana. Proc Natl Acad Sci USA 102, 12984–12989.
Xu, M., Li, Y., Zhang, Q., Xu, T., Qiu, L., Fan, Y., and Wang, L. (2014). Novel miRNA and phasiRNA biogenesis networks in soybean roots from two sister lines that are resistant and susceptible to SCN race 4. PLoS ONE 9, e110051.
Yoshikawa, M., Peragine, A., Park, M.Y., and Poethig, R.S. (2005). A pathway for the biogenesis of trans-acting siRNAs in Arabidopsis. Genes Dev 19, 2164–2175.
Zhai, J., Zhang, H., Arikit, S., Huang, K., Nan, G.L., Walbot, V., and Meyers, B.C. (2015). Spatiotemporally dynamic, cell-type-dependent premeiotic and meiotic phasiRNAs in maize anthers. Proc Natl Acad Sci USA 112, 3146–3151.
Zhang, H., Xia, R., Meyers, B.C., and Walbot, V. (2015a). Evolution, functions, and mysteries of plant ARGONAUTE proteins. Curr Opin Plant Biol 27, 84–90.
Zhang, P., Kang, J.Y., Gou, L.T., Wang, J., Xue, Y., Skogerboe, G., Dai, P., Huang, D.W., Chen, R., Fu, X.D., Liu, M.F., and He, S. (2015b). MIWI and piRNA-mediated cleavage of messenger RNAs in mouse testes. Cell Res 25, 193–207.
Zhang, Y.C., Liao, J.Y., Li, Z.Y., Yu, Y., Zhang, J.P., Li, Q.F., Qu, L.H., Shu, W.S., and Chen, Y.Q. (2014). Genome-wide screening and functional analysis identify a large number of long noncoding RNAs involved in the sexual reproduction of rice. Genome Biol 15, 512.
Zheng, Y., Wang, Y., Wu, J., Ding, B., and Fei, Z. (2015). A dynamic evolutionary and functional landscape of plant phased small interfering RNAs. BMC Biol 13, 32.
Zou, C., Wang, Q., Lu, C., Yang, W., Zhang, Y., Cheng, H., Feng, X., Prosper, M.A., and Song, G. (2016). Transcriptome analysis reveals long noncoding RNAs involved in fiber development in cotton (Gossypium arboreum). Sci China Life Sci 59, 164–171.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (91640202, 91335104) and the grants from Guangdong Province (2016A030308015) and Guangzhou (201707020018, 201710010029).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, Y., Zhou, Y., Zhang, Y. et al. Grass phasiRNAs and male fertility. Sci. China Life Sci. 61, 148–154 (2018). https://doi.org/10.1007/s11427-017-9166-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-017-9166-3