Abstract
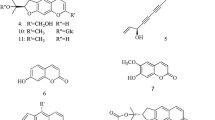
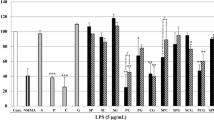
Phellodendri Cortex (Obaku in Japanese) and Coptidis Rhizoma (Oren), both of which contain berberine, have been used to prepare the kampo formula orengedokuto to treat inflammatory diseases, including dermatitis, gastric ulcers, and gastritis. These drugs are blended differently in other formulas, such as the use of Phellodendri Cortex in shichimotsukokato to treat hypertension and Coptidis Rhizoma in hangeshashinto to treat diarrhea and stomatitis. However, the differences in their medicinal properties are not well characterized. We prepared extracts from Phellodendron amurense bark (PAB) and Coptis chinensis rhizome (CCR) and separated them into alkaloid and non-alkaloid fractions. Anti-inflammatory effects were examined by monitoring the production of nitric oxide (NO), which is a pro-inflammatory mediator. A non-alkaloid fraction of the PAB extract suppressed NO production in hepatocytes more efficiently than that of the CCR extract. When each non-alkaloid fraction of the PAB and CCR extracts was administered to mice, the fractions of both extracts decreased the levels of mRNAs encoding inducible NO synthase and molecules in the interleukin-1β signaling pathway. Limonin and obakunone identified in the PAB non-alkaloid fraction suppressed NO production, exhibiting IC50 values of 16 and 2.6 μM, respectively, whereas berberine and coptisine displayed IC50 values of 12 and 14 μM, respectively. Limonin and obakunone reduced the expression of the iNOS gene, probably through the transcription factor nuclear factor-κB. Therefore, both limonoids and alkaloids may be responsible for the anti-inflammatory effects of the PAB extract, whereas alkaloids may be primarily responsible for those of the CCR extract. The different composition of the constituents may modulate the anti-inflammatory effects of Phellodendri Cortex and Coptidis Rhizoma.




Similar content being viewed by others
Abbreviations
- HPLC:
-
High-performance liquid chromatography
- UV:
-
Ultraviolet
- NMR:
-
Nuclear magnetic resonance
- TLC:
-
Thin-layer chromatography
- IC50 :
-
Half-maximal inhibitory concentration
- IL-1β:
-
Interleukin 1β
- LDH:
-
Lactate dehydrogenase
- NO:
-
Nitric oxide
- iNOS:
-
Inducible nitric oxide synthase
- RT:
-
Reverse transcription
- PCR:
-
Polymerase chain reaction
- EMSA:
-
Electrophoretic mobility shift assay
- asRNA:
-
Antisense transcripts
- PAB:
-
Phellodendron amurense bark
- CCR:
-
Coptis chinensis rhizomes
References
The Committee on the Japanese Pharmacopoeia (2016) The Japanese pharmacopoeia, 17th edn. The Minister of Health, Labour and Welfare, Japan
Ma Y, Fujimoto M, Watari H, Kimura M, Shimada Y (2016) The renoprotective effect of shichimotsukokato on hypertension-induced renal dysfunction in spontaneously hypertensive rats. J Nat Med 70:152–162
Yamashita T, Araki K, Tomifuji M, Kamide D, Tanaka Y, Shiotani A (2015) A traditional Japanese medicine—Hangeshashinto (TJ-14)—alleviates chemoradiation-induced mucositis and improves rates of treatment completion. Support Care Cancer 23:29–35
Anetai M, Hayashi T, Yamagishi T (1987) Determination of berberine alkaloids in Phellodendri Cortex. Rep Hokkaido Inst Public Health 37:18–21
Anetai M (1994) Chemical evaluation of Coptis rhizome—determination of berberine alkaloids by HPLC and spectrophotometry. Rep Hokkaido Inst Public Health 44:1–6
Fan G, Zhang MY, Zhou XD, Lai XR, Yue QH, Tang C, Luo WZ, Zhang Y (2012) Quality evaluation and species differentiation of Rhizoma coptidis by using proton nuclear magnetic resonance spectroscopy. Anal Chim Acta 747:76–83
Remppis A, Bea F, Greten HJ, Buttler A, Wang H, Zhou Q, Preusch MR, Enk R, Ehehalt R, Katus H, Blessing E (2010) Rhizoma Coptidis inhibits LPS-induced MCP-1/CCL2 production in murine macrophages via an AP-1 and NF-κB-dependent pathway. Mediat Inflamm 2010:194896
Wu J, Zhang H, Hu B, Yang L, Wang P, Wang F, Meng X (2016) Coptisine from Coptis chinensis inhibits production of inflammatory mediators in lipopolysaccharide-stimulated RAW 264.7 murine macrophage cells. Eur J Pharmacol 780:106–114
Lawrence T (2009) The nuclear factor NF-κB pathway in inflammation. Cold Spring Harb Perspect Biol 1:a001651
Min YD, Kwon HC, Yang MC, Lee KH, Choi SU, Lee KR (2007) Isolation of limonoids and alkaloids from Phellodendron amurense and their multidrug resistance (MDR) reversal activity. Arch Pharm Res 30:58–63
Leu CH, Li CY, Yao X, Wu TS (2006) Constituents from the leaves of Phellodendron amurense and their antioxidant activity. Chem Pharm Bull (Tokyo) 54:1308–1311
Zhou HY, Wang D, Cui Z (2008) Ferulates, amurenlactone A and amurenamide A from traditional Chinese medicine cortex Phellodendri amurensis. J Asian Nat Prod Res 10:409–413
Chen L, Wang L, Zhang Q, Zhang S, Ye W (2012) Non-alkaloid chemical constituents from Coptis chinensis. Zhongguo Zhong Yao Za Zhi 37:1241–1244
Colasanti M, Suzuki H (2000) The dual personality of NO. Trends Pharmacol Sci 21:249–252
Kitade H, Sakitani K, Inoue K, Masu Y, Kawada N, Hiramatsu Y, Kamiyama Y, Okumura T, Ito S (1996) Interleukin-1β markedly stimulates nitric oxide formation in the absence of other cytokines or lipopolysaccharide in primary cultured rat hepatocytes but not in Kupffer cells. Hepatology 23:797–802
Ohno N, Yoshigai E, Okuyama T, Yamamoto Y, Okumura T, Ikeya Y, Nishizawa M (2012) Chlorogenic acid from the Japanese herbal medicine Kinginka (Flos Lonicerae japonicae) suppresses the expression of inducible nitric oxide synthase in rat hepatocytes. HOAJ Biol 1:2. doi:10.7243/2050-0874-1-2
Matsuura T, Kaibori M, Araki Y, Matsumiya M, Yamamoto Y, Ikeya Y, Nishizawa M, Okumura T, Kwon AH (2012) Japanese herbal medicine, inchinkoto, inhibits inducible nitric oxide synthase induction in interleukin-1β-stimulated hepatocytes. Hepatol Res 42:76–90
Takimoto Y, Qian HY, Yoshigai E, Okumura T, Ikeya Y, Nishizawa M (2013) Hepatoprotective effects of gomisin N in the herbal drug gomishi (Schisandra chinensis) is mediated by C/EBPβ and NF-κB. Nitric Oxide 28:47–56
Miki H, Tokuhara K, Oishi M, Nakatake R, Tanaka Y, Kaibori M, Nishizawa M, Okumura T, Kon M (2016) Japanese kampo saireito has a liver-protective effect through the inhibition of inducible nitric oxide synthase induction in primary cultured rat hepatocytes. J Parenter Enter Nutr 40:1033–1041
Kamino T, Shimokura T, Morita Y, Tezuka Y, Nishizawa M, Tanaka K (2016) Comparative analysis of the constituents in Saposhnikoviae Radix and Glehniae Radix cum Rhizoma by monitoring inhibitory activity of nitric oxide production. J Nat Med 70:253–259
Nakano Y, Nasu M, Kano M, Kameoka H, Okuyama T, Nishizawa M, Ikeya Y (2017) Lignans from guaiac resin decrease nitric oxide production in interleukin 1β-treated hepatocytes. J Nat Med 71:190–197
Murayama Y, Shinozaki K (1926) On the alkaloids of Coptis root. On the alkaloids of the bark of Phellodendron amurense Rupr. Yakugaku Zasshi 46:299–302
Grycová L, Dostal J, Marek R (2007) Quaternary protoberberine alkaloids. Phytochemistry 68:150–175
Breksa AP III, Dragull K, Wong RY (2008) Isolation and identification of the first C-17 limonin epimer, epilimonin. J Agric Food Chem 56:5595–5598
Okuzaki D, Fukushima T, Tougan T, Ishii T, Kobayashi S, Yoshizaki K, Akita T, Nojima H (2010) Genopal™: a novel hollow fibre array for focused microarray analysis. DNA Res 17:369–379
Kanemaki T, Kitade H, Hiramatsu Y, Kamiyama Y, Okumura T (1993) Stimulation of glycogen degradation by prostaglandin E2 in primary cultured hepatocytes. Prostaglandins 45:459–474
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, and [15N]nitrate in biological fluids. Anal Biochem 126:131–138
Tanemoto R, Okuyama T, Matsuo H, Okumura T, Ikeya Y, Nishizawa M (2015) The constituents of licorice (Glycyrrhiza uralensis) differentially suppress nitric oxide production in interleukin-1β-treated hepatocytes. Biochem Biophys Rep 2:153–159. doi:10.1016/j.bbrep.2015.06.004
Yoshigai E, Hara T, Inaba H, Hashimoto I, Tanaka Y, Kaibori M, Kimura T, Okumura T, Kwon AH, Nishizawa M (2014) Interleukin-1β induces tumor necrosis factor-α secretion from rat hepatocytes. Hepatol Res 44:571–583
Matsui K, Nishizawa M, Ozaki T, Kimura T, Hashimoto I, Yamada M, Kaibori M, Kamiyama Y, Ito S, Okumura T (2008) Natural antisense transcript stabilizes inducible nitric oxide synthase messenger RNA in rat hepatocytes. Hepatology 47:686–697
Kawamura T, Yokoe M, Hisata Y, Okuda K, Noro Y, Yamaguchi S, Tanaka T, Nishibe S, Wada K (2002) Diversity of the quality of Phellodendron barks from different habitats. (2). Content variation of limonoids. Nat Med 56:198–199
Tetko IV, Tanchuk VY (2002) Application of associative neural networks for prediction of lipophilicity in ALOGPS 2.1 program. J Chem Inf Comput Sci 42:1136–1145
Acknowledgements
We thank Ms. Yuka Yamamoto, Ms. Megumi Hashima, and Mr. Kento Uemura for technical assistance; Dr. Takehito Miura for valuable advice; and Ms. Noriko Kanazawa for secretarial assistance; and Dr. Yuji Hasegawa (Central Equipment Room, Daiichi University of Pharmacy) for the MS measurements.
Author information
Authors and Affiliations
Contributions
MN and YI designed the experiments and wrote the manuscript. AF designed and performed the experiments as a graduate student. KW, TO, and TO performed the experiments and analyzed the data.
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest.
Rights and permissions
About this article
Cite this article
Fujii, A., Okuyama, T., Wakame, K. et al. Identification of anti-inflammatory constituents in Phellodendri Cortex and Coptidis Rhizoma by monitoring the suppression of nitric oxide production. J Nat Med 71, 745–756 (2017). https://doi.org/10.1007/s11418-017-1107-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-017-1107-4