Abstract
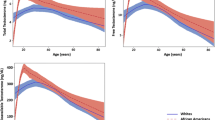
It is well established from previous cross-sectional studies that telomeres shorten with age. However, due to a considerable inter-individual variation in telomere length (TL), its relationship with biological aging is difficult to unpick. Longitudinal repeated assessments of TL changes within individuals should augment our understanding of TL dynamics in aging. This study disentangles within- and inter-individual effects of age on leukocyte telomere length (LTL) dynamics in a large population-based cohort of older adults. A total of 4053 subjects aged 50 and older from the WHO Study on global AGEing and adult health (SAGE) in Shanghai were studied. Relative LTL (T/S ratio) was measured at baseline (2009–2010) and follow-up (2017–2018) by quantitative real-time polymerase chain reaction. We used linear random slope models to analyze LTL dynamics in relation to age and sex and within-subject centering method to distinguish within- versus between-subject effects. We observed LTL shortening in 66.32%, maintenance in 11.23%, and elongation in 22.45% of the study participants. LTL declined significantly with age both cross-sectionally and longitudinally. More importantly, the longitudinal decline in LTL was much greater than the cross-sectional decline (− 0.017 (p < 0.001) versus − 0.002 (p < 0.001) per year). Furthermore, women had a lower within-subject LTL shortening rate than men (− 0.014 versus − 0.020 per year, p < 0.001). The within-individual longitudinal decline in LTL was much greater than the inter-individual cross-sectional decline, indicating that chronological age might impose a greater impact on LTL shortening than other influencing factors combined. Moreover, women showed a lower within-individual LTL shortening rate than men.
Similar content being viewed by others
References
Blackburn EH, Epel ES, Lin J. Human telomere biology: a contributory and interactive factor in aging, disease risks, and protection. Science. 2015;350(6265):1193–8.
Gilson E, Géli V. How telomeres are replicated. Nat Rev Mol Cell Biol. 2007;8(10):825–38.
Lingner J, Cooper JP, Cech TR. Telomerase and DNA end replication: no longer a lagging strand problem? Science. 1995;269(5230):1533–4.
Herrmann M, Pusceddu I, März W, Herrmann W. Telomere biology and age-related diseases. Clin Chem Lab Med. 2018;56(8):1210–22.
Hayflick L. Mortality and immortality at the cellular level. A review. Biochemistry (Mosc). 1997;62(11):1180–90.
Wong JMY, Collins K. Telomere maintenance and disease. Lancet. 2003;362(9388):983–8.
Sahin E, Depinho RA. Linking functional decline of telomeres, mitochondria and stem cells during ageing. Nature. 2010;464(7288):520–8.
Bendix L, Thinggaard M, Fenger M, Kolvraa S, Avlund K, Linneberg A, et al. Longitudinal changes in leukocyte telomere length and mortality in humans. J Gerontol A Biol Sci Med Sci. 2014;69(2):231–9.
Andrew T, Aviv A, Falchi M, Surdulescu GL, Gardner JP, Lu X, et al. Mapping genetic loci that determine leukocyte telomere length in a large sample of unselected female sibling pairs. Am J Hum Genet. 2006;78(3):480–6.
Vasa-Nicotera M, Brouilette S, Mangino M, Thompson JR, Braund P, Clemitson J-R, et al. Mapping of a major locus that determines telomere length in humans. Am J Hum Genet. 2005;76(1):147–51.
Bischoff C, Graakjaer J, Petersen HC, Hjelmborg JVB, Vaupel JW, Bohr V, et al. The heritability of telomere length among the elderly and oldest-old. Twin Res Hum Genet. 2005;8(5):433–9.
Slagboom PE, Droog S, Boomsma DI. Genetic determination of telomere size in humans: a twin study of three age groups. Am J Hum Genet. 1994;55(5):876–82.
Njajou OT, Cawthon RM, Damcott CM, Wu S-H, Ott S, Garant MJ, et al. Telomere length is paternally inherited and is associated with parental lifespan. Proc Natl Acad Sci U S A. 2007;104(29):12135–9.
Broer L, Codd V, Nyholt DR, Deelen J, Mangino M, Willemsen G, et al. Meta-analysis of telomere length in 19,713 subjects reveals high heritability, stronger maternal inheritance and a paternal age effect. Eur J Hum Genet. 2013;21(10):1163–8.
Hjelmborg JB, Dalgård C, Möller S, Steenstrup T, Kimura M, Christensen K, et al. The heritability of leucocyte telomere length dynamics. J Med Genet. 2015;52(5):297–302.
Gardner M, Bann D, Wiley L, Cooper R, Hardy R, Nitsch D, et al. Gender and telomere length: systematic review and meta-analysis. Exp Gerontol. 2014;51:15–27.
Hansen MEB, Hunt SC, Stone RC, Horvath K, Herbig U, Ranciaro A, et al. Shorter telomere length in Europeans than in Africans due to polygenetic adaptation. Hum Mol Genet. 2016;25(11):2324–30.
Lynch SM, Peek MK, Mitra N, Ravichandran K, Branas C, Spangler E, et al. Race, ethnicity, psychosocial factors, and telomere length in a multicenter setting. PLoS One. 2016;11(1):e0146723.
Aviv A, Chen W, Gardner JP, Kimura M, Brimacombe M, Cao X, et al. Leukocyte telomere dynamics: longitudinal findings among young adults in the Bogalusa Heart Study. Am J Epidemiol. 2009;169(3):323–9.
Nordfjäll K, Svenson U, Norrback K-F, Adolfsson R, Lenner P, Roos G. The individual blood cell telomere attrition rate is telomere length dependent. PLoS Genet. 2009;5(2):e1000375.
Müezzinler A, Zaineddin AK, Brenner H. A systematic review of leukocyte telomere length and age in adults. Ageing Res Rev. 2013;12(2):509–19.
Berglund K, Reynolds CA, Ploner A, Gerritsen L, Hovatta I, Pedersen NL, et al. Longitudinal decline of leukocyte telomere length in old age and the association with sex and genetic risk. Aging (Albany NY). 2016;8(7):1398–415.
Farzaneh-Far R, Lin J, Epel E, Lapham K, Blackburn E, Whooley MA. Telomere length trajectory and its determinants in persons with coronary artery disease: longitudinal findings from the heart and soul study. PLoS One. 2010;5(1):e8612.
Kowal P, Chatterji S, Naidoo N, Biritwum R, Fan W, Lopez Ridaura R, et al. Data resource profile: the World Health Organization Study on global AGEing and adult health (SAGE). Int J Epidemiol. 2012;41(6):1639–49.
Cawthon RM. Telomere measurement by quantitative PCR. Nucleic Acids Res. 2002;30(10):e47.
van de Pol M, Wright J. A simple method for distinguishing within- versus between-subject effects using mixed models. Anim Behav. 2009;77(3):753–8.
Turner KJ, Vasu V, Griffin DK. Telomere biology and human phenotype. Cells. 2019;8(1):73. https://doi.org/10.3390/cells8010073.
Sidorov I, Kimura M, Yashin A, Aviv A. Leukocyte telomere dynamics and human hematopoietic stem cell kinetics during somatic growth. Exp Hematol. 2009 Apr;37(4):514–24.
Aviv A. The epidemiology of human telomeres: faults and promises. J Gerontol A Biol Sci Med Sci. 2008;63(9):979–83.
Dugdale HL, Richardson DS. Heritability of telomere variation: it is all about the environment! Philos Trans R Soc Lond Ser B Biol Sci. 2018;373(1741):20160450. https://doi.org/10.1098/rstb.2016.0450.
Chen W, Kimura M, Kim S, Cao X, Srinivasan SR, Berenson GS, et al. Longitudinal versus cross-sectional evaluations of leukocyte telomere length dynamics: age-dependent telomere shortening is the rule. J Gerontol A Biol Sci Med Sci. 2011;66(3):312–9.
Guzzardi MA, Iozzo P, Salonen M, Kajantie E, Eriksson JG. Rate of telomere shortening and metabolic and cardiovascular risk factors: a longitudinal study in the 1934-44 Helsinki Birth Cohort Study. Ann Med. 2015;47(6):499–505.
Simons MJP, Stulp G, Nakagawa S. A statistical approach to distinguish telomere elongation from error in longitudinal datasets. Biogerontology. 2014;15(1):99–103.
Liu J, Wang L, Wang Z, Liu J-P. Roles of telomere biology in cell senescence, replicative and chronological ageing. Cells. 2019;8(1):54. https://doi.org/10.3390/cells8010054.
Steenstrup T, Hjelmborg JVB, Kark JD, Christensen K, Aviv A. The telomere lengthening conundrum--artifact or biology? Nucleic Acids Res. 2013 Jul;41(13):e131.
Cherif H, Tarry JL, Ozanne SE, Hales CN. Ageing and telomeres: a study into organ- and gender-specific telomere shortening. Nucleic Acids Res. 2003;31(5):1576–83.
Sanders JL, Newman AB. Telomere length in epidemiology: a biomarker of aging, age-related disease, both, or neither? Epidemiol Rev. 2013;35:112–31.
Bekaert S, De Meyer T, Rietzschel ER, De Buyzere ML, De Bacquer D, Langlois M, et al. Telomere length and cardiovascular risk factors in a middle-aged population free of overt cardiovascular disease. Aging Cell. 2007;6(5):639–47.
Dalgård C, Benetos A, Verhulst S, Labat C, Kark JD, Christensen K, et al. Leukocyte telomere length dynamics in women and men: menopause vs age effects. Int J Epidemiol. 2015;44(5):1688–95.
Harris SE, Marioni RE, Martin-Ruiz C, Pattie A, Gow AJ, Cox SR, et al. Longitudinal telomere length shortening and cognitive and physical decline in later life: the Lothian Birth Cohorts 1936 and 1921. Mech Ageing Dev. 2016;154:43–8.
Aviv A, Shay JW. Reflections on telomere dynamics and ageing-related diseases in humans. Philos Trans R Soc Lond Ser B Biol Sci. 2018;373(1741):20160436. https://doi.org/10.1098/rstb.2016.0436.
Kyo S, Takakura M, Kanaya T, Zhuo W, Fujimoto K, Nishio Y, et al. Estrogen activates telomerase. Cancer Res. 1999;59(23):5917–21.
Adams J, Martin-Ruiz C, Pearce MS, White M, Parker L, Zglinicki TV. No association between socio-economic status and white blood cell telomere length. Aging Cell. 2007;6(1):125–8.
Hunt SC, Chen W, Gardner JP, Kimura M, Srinivasan SR, Eckfeldt JH, et al. Leukocyte telomeres are longer in African Americans than in whites: the National Heart, Lung, and Blood Institute Family Heart Study and the Bogalusa Heart Study. Aging Cell. 2008;7(4):451–8.
Shiels PG, McGlynn LM, MacIntyre A, Johnson PCD, Batty GD, Burns H, et al. Accelerated telomere attrition is associated with relative household income, diet and inflammation in the pSoBid cohort. PLoS One. 2011;6(7):e22521.
Acknowledgments
The authors thank the participants and the investigators who participated in this study.
Funding
This work was supported by the WHO and the US National Institute on Aging through Interagency Agreements (OGHA 04034785, YA1323-08-CN-0020, Y1-AG-1005-01) and through a research grant (R01-AG034479); the Program for Outstanding Medical Academic Leader, Shanghai, China (2019LJ24); and Shanghai Municipal Health Commission (20204Y0196, 2020YJZX0113, 201840118).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Figure 1
Correlations of LTL change and LTL at Wave 3 with baseline LTL (PNG 5883 kb)
Supplementary Figure 2
Predicted mean LTL in relation to age (by sex) from standard random slope model including interaction between age and sex (PNG 6131 kb)
About this article
Cite this article
Huang, Z., Liu, C., Ruan, Y. et al. Dynamics of leukocyte telomere length in adults aged 50 and older: a longitudinal population-based cohort study. GeroScience 43, 645–654 (2021). https://doi.org/10.1007/s11357-020-00320-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-020-00320-y