Abstract
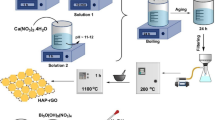
A facile, feasible, and green synthesis via an electrochemical exfoliation process was applied to synthesize nitrogen-doped MgO/graphene nanocomposite (N-MgO/G). The N-MgO/G nanocomposite was characterized by several analytical techniques including X-ray photoelectron spectroscopy, X-ray powder diffraction, transmission electron microscopy, field emission scanning electron microscopy, energy-dispersive X-ray spectroscopy, selected area electron diffraction, and elemental mapping analysis. N-MgO/G nanocomposite was then applied to adsorb lead metal ions (Pb2+) from aqueous solutions. The N-MgO/G nanocomposite demonstrated a remarkably high Langmuir maximum adsorption capacity (294.12 mg/g) for Pb2+ ions under the optimum experimental conditions at a pH of 5.13, time of 35 min, dose of 0.025 g, the concentration of 400 mg/L, and a temperature of 36 °C. Adsorption kinetics results fitted with a pseudo-second-order model and a thermodynamic study showed that Pb2+ adsorption is an endothermic process. The practical application of N-MgO/G was also investigated to test its applicability in real water samples collected from different sources such as deionized water, tap water, wastewater, and river water.




Similar content being viewed by others
References
Ablouh E, Essaghraoui A, Eladlani N, Rhazi M, Taourirte M (2019) Uptake of Pb(II) onto nanochitosan/sodium alginate hybrid beads: mechanism and kinetics study. Water Environ Res 91:239–249. https://doi.org/10.1002/wer.1050
Alghamdi AA, Al-Odayni A-B, Saeed WS et al (2019) Efficient adsorption of lead (II) from aqueous phase solutions using polypyrrole-based activated carbon. Materials (Basel) 12:2020. https://doi.org/10.3390/ma12122020
Alharbi NS, Hu B, Hayat T, Rabah SO, Alsaedi A, Zhuang L, Wang X (2020) Efficient elimination of environmental pollutants through sorption-reduction and photocatalytic degradation using nanomaterials. Front Chem Sci Eng 14:1124–1135. https://doi.org/10.1007/s11705-020-1923-z
Ansari MO, Khan MM, Ansari SA, Amal I, Lee J, Cho MH (2014) pTSA doped conducting graphene/polyaniline nanocomposite fibers: Thermoelectric behavior and electrode analysis. Chem Eng J 242:155–161. https://doi.org/10.1016/j.cej.2013.12.033
Baig U, Uddin MK, Gondal MA (2020a) Removal of hazardous azo dye from water using synthetic nano adsorbent: facile synthesis, characterization, adsorption, regeneration and design of experiments. Colloids Surf A Physicochem Eng Asp 584:124031. https://doi.org/10.1016/j.colsurfa.2019.124031
Baig U, Uddin MK, Sajid M (2020b) Surface modification of TiO2 nanoparticles using conducting polymer coating: spectroscopic, structural, morphological characterization and interaction with dye molecules. Mater Today Commun 25:101534. https://doi.org/10.1016/j.mtcomm.2020.101534
Cenic-Milosevic D, Mileusnic I, Kolak V, Pejanovic D, Ristic T, Jakovljevic A, Popovic M, Pesic D, Melih I (2013) Environmental lead pollution and its possible influence on tooth loss and hard dental tissue lesions. Vojnosanit Pregl 70:751–756. https://doi.org/10.2298/VSP1308751C
Choi H (2019) Assessment of the adsorption kinetics, equilibrium, and thermodynamic for Pb(II) removal using a low-cost hybrid biowaste adsorbent, eggshell/coffee ground/sericite. Water Environ Res 91:1600–1612. https://doi.org/10.1002/wer.1158
Çiğil AB, Urucu OA, Kahraman MV (2019) Nanodiamond-containing polyethyleneimine hybrid materials for lead adsorption from aqueous media. J Appl Polym Sci 136:48241. https://doi.org/10.1002/app.48241
Colantonio N, Kim Y (2016) Lead(II) Removal at the bioanode of microbial electrolysis cells. Chem Select 1:5743–5748. https://doi.org/10.1002/slct.201601539
Compton OC, Nguyen ST (2010) Graphene oxide, highly reduced graphene oxide, and graphene: versatile building blocks for carbon-based materials. Small 6:711–723. https://doi.org/10.1002/smll.200901934
Corneille JS, He J-W, Goodman DW (1994) XPS characterization of ultra-thin MgO films on a Mo(100) surface. Surf Sci 306:269–278. https://doi.org/10.1016/0039-6028(94)90071-X
Coroş M, Pogăcean F, Roşu M-C, Socaci C, Borodi G, Mageruşan L, Biriş AR, Pruneanu S (2016) Simple and cost-effective synthesis of graphene by electrochemical exfoliation of graphite rods. RSC Adv 6:2651–2661. https://doi.org/10.1039/C5RA19277C
Das B, Moumita S, Ghosh S, Khan MI, Indira D, Jayabalan R, Tripathy SK, Mishra A, Balasubramanian P (2018) Biosynthesis of magnesium oxide (MgO) nanoflakes by using leaf extract of Bauhinia purpurea and evaluation of its antibacterial property against Staphylococcus aureus. Mater Sci Eng C 91:436–444. https://doi.org/10.1016/j.msec.2018.05.059
El-Dib FI, Mohamed DE, El-Shamy OAA, Mishrif MR (2020) Study the adsorption properties of magnetite nanoparticles in the presence of different synthesized surfactants for heavy metal ions removal. Egypt J Pet 29:1–7. https://doi.org/10.1016/j.ejpe.2019.08.004
Esmaeili A, Eslami H (2019) Efficient removal of Pb(II) and Zn(II) ions from aqueous solutions by adsorption onto a native natural bentonite. MethodsX 6:1979–1985. https://doi.org/10.1016/j.mex.2019.09.005
Gao J, Guo X, Tao W, Chen D, Lu J, Chen Y (2019) Norepinephrine-functionalised nanoflower-like organic silica as a new adsorbent for effective Pb(II) removal from aqueous solutions. Sci Rep 9:293. https://doi.org/10.1038/s41598-018-36644-1
He Y, Wu P, Xiao W, Li G, Yi J, He Y, Chen C, Ding P, Duan Y (2019) Efficient removal of Pb(II) from aqueous solution by a novel ion imprinted magnetic biosorbent: Adsorption kinetics and mechanisms. PLoS One 14:e0213377. https://doi.org/10.1371/journal.pone.0213377
Husein DZ (2019) Facile one-pot synthesis of porous N-doped graphene based NiO composite for parabens removal from wastewater and its reusability. Desalin Water Treat 166:211–221. https://doi.org/10.5004/dwt.2019.24339
Huynh P-T, Nguyen N-T, Van HN et al (2020) Modeling and optimization of biosorption of lead (II) ions from aqueous solution onto pine leaves (Pinus kesiya) using response surface methodology. Desalin Water Treat 173:383–393. https://doi.org/10.5004/dwt.2020.24807
Ibupoto AS, Qureshi UA, Arain M, Ahmed F, Khatri Z, Brohi RZ, Kim IS, Ibupoto Z (2020) Zno/Carbon nanofibers for efficient adsorption of lead from aqueous solutions. Environ Technol 41:2731–2741. https://doi.org/10.1080/09593330.2019.1580774
Johra FT, Jung W-G (2015) Hydrothermally reduced graphene oxide as a supercapacitor. Appl Surf Sci 357:1911–1914. https://doi.org/10.1016/j.apsusc.2015.09.128
Jung JM, Peddinti R (2018) Lead toxicity in the pediatric patient with sickle cell disease: Unique risks and management. Pediatr Ann 47:e36–e40. https://doi.org/10.3928/19382359-20171218-01
Kartick B, Srivastava SK, Srivastava I (2013) Green synthesis of graphene. J Nanosci Nanotechnol 13:4320–4324. https://doi.org/10.1166/jnn.2013.7461
Kavand M, Eslami P, Razeh L (2020) The adsorption of cadmium and lead ions from the synthesis wastewater with the activated carbon: Optimization of the single and binary systems. J Water Process Eng 34:101151. https://doi.org/10.1016/j.jwpe.2020.101151
Khan MA, Uddin MK, Bushra R, Ahmad A, Nabi SA (2014) Synthesis and characterization of polyaniline Zr(IV) molybdophosphate for the adsorption of phenol from aqueous solution. React Kinet Mech Catal 113:499–517. https://doi.org/10.1007/s11144-014-0751-x
Khatoon A, Uddin MK, Rao RAK (2018) Adsorptive remediation of Pb(II) from aqueous media using Schleichera oleosa bark. Environ Technol Innov 11:1–14. https://doi.org/10.1016/j.eti.2018.04.004
Kovach AE (2008) Rubinʼs Pathology, Clinicopathologic Foundations of Medicine. Am J Surg Pathol 32:1427. https://doi.org/10.1097/pas.0b013e31816d7194
Kumar M, Chung JS, Hur SH (2019) Graphene Composites for Lead Ions Removal from Aqueous Solutions. Appl Sci 9:2925. https://doi.org/10.3390/app9142925
Lee M-E, Park JH, Chung JW (2019) Comparison of the lead and copper adsorption capacities of plant source materials and their biochars. J Environ Manag 236:118–124. https://doi.org/10.1016/j.jenvman.2019.01.100
Li W, Liao X, Wang L, Huang Z (2019) Adsorption of cadmium and lead in wastewater by four kinds of biomass xanthates. Water Sci Technol 79:1222–1230. https://doi.org/10.2166/wst.2019.124
Lisi N, Dikonimos T, Buonocore F, Pittori M, Mazzaro R, Rizzoli R, Marras S, Capasso A (2017) Contamination-free graphene by chemical vapor deposition in quartz furnaces. Sci Rep 7:9927. https://doi.org/10.1038/s41598-017-09811-z
Lu F, Astruc D (2020) Nanocatalysts and other nanomaterials for water remediation from organic pollutants. Coord Chem Rev 408:213180. https://doi.org/10.1016/j.ccr.2020.213180
Machida M, Mochimaru T, Tatsumoto H (2006) Lead(II) adsorption onto the graphene layer of carbonaceous materials in aqueous solution. Carbon N Y 44:2681–2688. https://doi.org/10.1016/j.carbon.2006.04.003
Mahdi Z, Yu QJ, El Hanandeh A (2018) Removal of lead(II) from aqueous solution using date seed-derived biochar: batch and column studies. Appl Water Sci 8:181. https://doi.org/10.1007/s13201-018-0829-0
Manzoor K, Ahmad M, Ahmad S, Ikram S (2019) Removal of Pb( <scp>ii</scp> ) and Cd( <scp>ii</scp> ) from wastewater using arginine cross-linked chitosan–carboxymethyl cellulose beads as green adsorbent. RSC Adv 9:7890–7902. https://doi.org/10.1039/C9RA00356H
Mashkoor F, Nasar A (2020) Magsorbents: Potential candidates in wastewater treatment technology – A review on the removal of methylene blue dye. J Magn Magn Mater 500:166408. https://doi.org/10.1016/j.jmmm.2020.166408
Mashkoor F, Nasar A, Inamuddin (2020) Carbon nanotube-based adsorbents for the removal of dyes from waters: A review. Environ Chem Lett 18:605–629. https://doi.org/10.1007/s10311-020-00970-6
Mason LH, Harp JP, Han DY (2014) Pb Neurotoxicity: Neuropsychological Effects of Lead Toxicity. Biomed Res Int 2014:1–8. https://doi.org/10.1155/2014/840547
Motlochová M, Slovák V, Pližingrová E, Lidin S, Šubrt J (2020) Highly-efficient removal of Pb(II), Cu(II) and Cd(II) from water by novel lithium, sodium and potassium titanate reusable microrods. RSC Adv 10:3694–3704. https://doi.org/10.1039/c9ra08737k
Needleman HL (2000) The Removal of lead from gasoline: historical and personal reflections. Environ Res 84:20–35. https://doi.org/10.1006/enrs.2000.4069
Nyairo WN, Eker YR, Kowenje C, Akin I, Bingol H, Tor A, Ongeri DM (2018) Efficient adsorption of lead (II) and copper (II) from aqueous phase using oxidized multiwalled carbon nanotubes/polypyrrole composite. Sep Sci Technol 53:1498–1510. https://doi.org/10.1080/01496395.2018.1424203
Pang FM, Kumar P, Teng TT, Mohd Omar AK, Wasewar KL (2011) Removal of lead, zinc and iron by coagulation–flocculation. J Taiwan Inst Chem Eng 42:809–815. https://doi.org/10.1016/j.jtice.2011.01.009
Park SK, O’Neill MS, Vokonas PS et al (2008) Air pollution and heart rate variability. Epidemiology 19:111–120. https://doi.org/10.1097/EDE.0b013e31815c408a
Qi J, Li Y, Majeed H, Goff HD, Rahman MRT, Zhong F (2019) Adsorption mechanism modeling using lead (Pb) sorption data on modified rice bran-insoluble fiber as universal approach to assess other metals toxicity. Int J Food Prop 22:1397–1410. https://doi.org/10.1080/10942912.2019.1650764
Ramdani A, Kadeche A, Adjdir M, Taleb Z, Ikhou D, Taleb S, Deratani A (2020) Lead and cadmium removal by adsorption process using hydroxyapatite porous materials. Water Pract Technol 15:130–141. https://doi.org/10.2166/wpt.2020.003
Rao RAK, Kashifuddin M (2012) Adsorption properties of coriander seed powder (Coriandrum sativum): Extraction and pre-concentration of Pb(II), Cu(II) and Zn(II) ions from aqueous solution. Adsorpt Sci Technol 30:127–146. https://doi.org/10.1260/0263-6174.30.2.127
Saini S, Kumar R, Chawla J, Kaur I (2019) Adsorption of bivalent lead ions from an aqueous phase system: Equilibrium, thermodynamic, kinetics, and optimization studies. Water Environ Res 91:1692–1704. https://doi.org/10.1002/wer.1180
Sayago UFC, Castro YP, Rivera LRC, Mariaca AG (2020) Estimation of equilibrium times and maximum capacity of adsorption of heavy metals by E. crassipes (review). Environ Monit Assess 192:141. https://doi.org/10.1007/s10661-019-8032-9
Shahabuddin S, Tashakori C, Kamboh MA, Sotoudehnia Korrani Z, Saidur R, Rashidi Nodeh H, Bidhendi ME (2018) Kinetic and equilibrium adsorption of lead from water using magnetic metformin-substituted SBA-15. Environ Sci Water Res Technol 4:549–558. https://doi.org/10.1039/C7EW00552K
Tu F, Liu S, Jin G, Yan G, Pan C (2013) Fabrication of graphene from graphene oxide by ultrasonication with high Li storage capability. Powder Technol 249:146–150. https://doi.org/10.1016/j.powtec.2013.08.006
Uddin MK (2017) A review on the adsorption of heavy metals by clay minerals, with special focus on the past decade. Chem Eng J 308:438–462. https://doi.org/10.1016/j.cej.2016.09.029
Uddin MK, Baig U (2019) Synthesis of Co3O4 nanoparticles and their performance toward methyl orange dye removal: Characterisation, adsorption and response surface methodology. J Clean Prod 211:1141–1153. https://doi.org/10.1016/j.jclepro.2018.11.232
Uddin MK, Nasar A (2020a) Decolorization of Basic Dyes Solution by Utilizing Fruit Seed Powder. KSCE J Civ Eng 24:345–355. https://doi.org/10.1007/s12205-020-0523-2
Uddin MK, Nasar A (2020b) Walnut shell powder as a low-cost adsorbent for methylene blue dye: isotherm, kinetics, thermodynamic, desorption and response surface methodology examinations. Sci Rep 10:7983. https://doi.org/10.1038/s41598-020-64745-3
Uddin MK, Ahmed SS, Naushad M (2019) A mini update on fluoride adsorption from aqueous medium using clay materials. Desalin Water Treat 145:232–248. https://doi.org/10.5004/dwt.2019.23509
Uddin MK, Rahaman PF (2017) A study on the potential applications of rice husk derivatives as useful adsorptive material. In: Inamuddin Mohammad A, Asiri AM (eds) Inorganic Pollutants in Wastewater. Methods of Analysis, Removal and Treatment, Materials Research Forum LLC. pp 149–186. https://doi.org/10.21741/9781945291357-4
Wadhawan S, Jain A, Nayyar J, Mehta SK (2020) Role of nanomaterials as adsorbents in heavy metal ion removal from waste water: A review. J Water Process Eng 33:101038. https://doi.org/10.1016/j.jwpe.2019.101038
Weisskopf MG, Weuve J, Nie H, Saint-Hilaire MH, Sudarsky L, Simon DK, Hersh B, Schwartz J, Wright RO, Hu H (2010) Association of cumulative lead exposure with Parkinson’s disease. Environ Health Perspect 118:1609–1613. https://doi.org/10.1289/ehp.1002339
Wu Y, Pang H, Liu Y, Wang X, Yu S, Fu D, Chen J, Wang X (2019) Environmental remediation of heavy metal ions by novel-nanomaterials: A review. Environ Pollut 246:608–620. https://doi.org/10.1016/j.envpol.2018.12.076
Zamani A, Poursattar Marjani A, Abedi Mehmandar M (2019) Synthesis of high surface area magnesia by using walnut shell as a template. Green Process Synth 8:199–206. https://doi.org/10.1515/gps-2018-0066
Zhang M, Yin Q, Ji X, Wang F, Gao X, Zhao M (2020) High and fast adsorption of Cd(II) and Pb(II) ions from aqueous solutions by a waste biomass based hydrogel. Sci Rep 10:3285. https://doi.org/10.1038/s41598-020-60160-w
Zhuo H, Zhang X, Wang L, Lu Q, Kaplan DL (2018) Sonication exfoliation of defect-free graphene in aqueous silk nanofiber solutions. ACS Sustain Chem Eng 6:12261–12267. https://doi.org/10.1021/acssuschemeng.8b02644
Acknowledgment
The authors extend their appreciation to the DSR at Majmaah University.
Funding
The authors extend their appreciation to the Deanship of Scientific Research (DSR) at Majmaah University for funding this work under project No (RGP-2019-14).
Author information
Authors and Affiliations
Contributions
DZH did the experiments, MKU analyzed the data and wrote the paper, OA performed the characterization and wrote this part, and SSA joined the team.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not Applicable
Consent for publication
Not Applicable
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Responsible Editor: Santiago V. Luis
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Husein, D.Z., Uddin, M.K., Ansari, M.O. et al. Green synthesis, characterization, application and functionality of nitrogen-doped MgO/graphene nanocomposite. Environ Sci Pollut Res 28, 28014–28023 (2021). https://doi.org/10.1007/s11356-021-12628-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-12628-z